Pediatric emergencies of the upper and lower airway
Whether one practices in a community hospital, a busy trauma center, or an independently standing pediatric hospital, emergencies of the pediatric airway can be uniquely challenging. In this article, we review upper airway anatomy and discuss inflammatory processes that present in the upper airway of children. We also discuss lower-airway emergencies, including pediatric chest trauma and common causes of cough and wheezing.
In the evaluation of upper airway abnormalities, the current standard for imaging relies initially on radiography, which may then be followed by contrast-enhanced computed tomography (CECT) of the neck. Lower airway abnormalities are most commonly evaluated by radiography, although traumatic injuries to the chest may warrant cross-sectional imaging with computed tomography (CT). The protocol is guided by the suspected pathology.
Upper airway anatomy
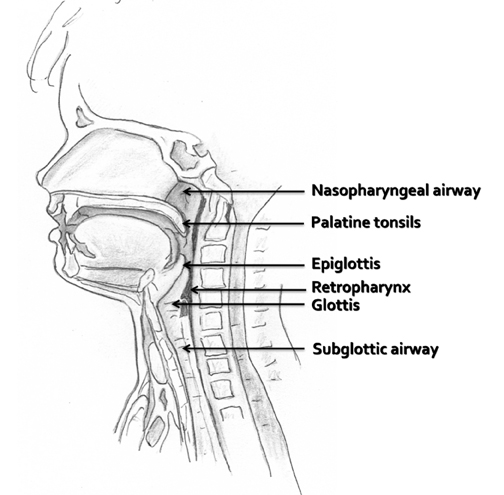
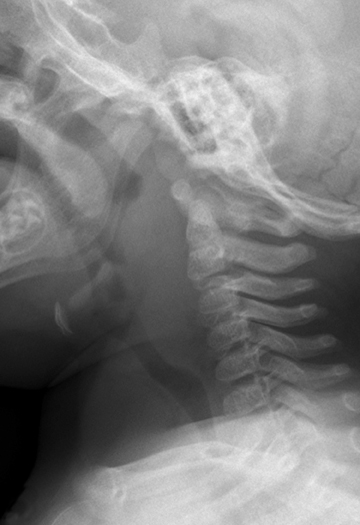
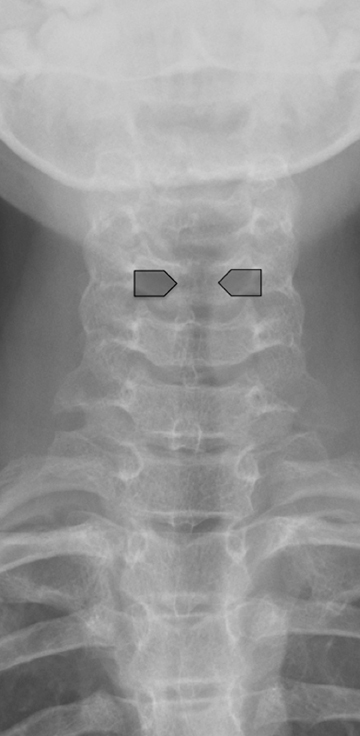
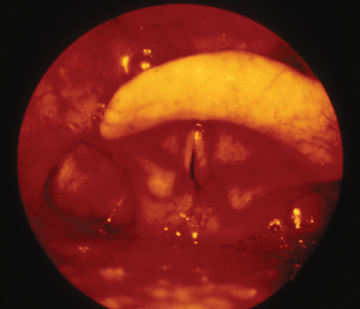
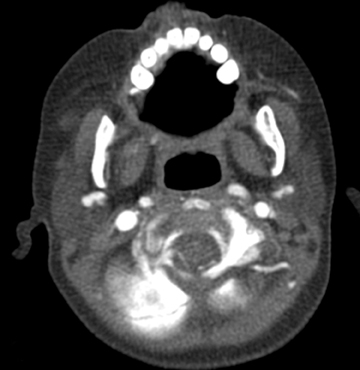
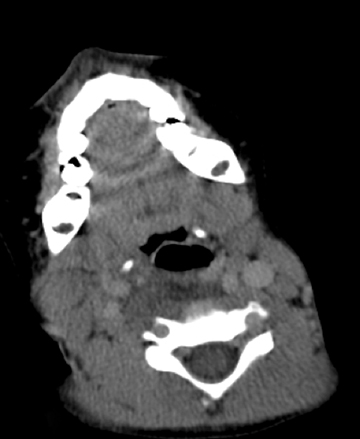
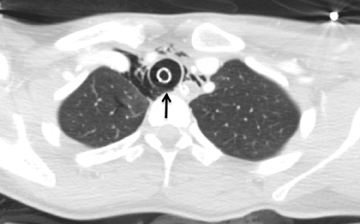
The upper airway includes the pharynx, the larynx, and the subglottic trachea to the level of the thoracic inlet. In children, upper airway pathology may be readily assessed by a lateral radiographic view of the neck to assess for soft-tissue thickening. Figure 1 illustrates the midline structures of the pediatric neck. The adenoids and tonsils are a collection of lymphoid tissues surrounding the pharyngeal airway and may become hypertrophied during upper airway infections. The adenoids, or pharyngeal tonsils, are seen along the posterior upper aspect of the nasopharynx, and when hypertrophied, they may partially efface the airway, as seen in Figure 2. The palatine tonsils, found at the posterior aspect of the soft palate, and the lingual tonsils, seen at the base of the tongue anterior to the epiglottis, often enlarge during upper respiratory infections as well.
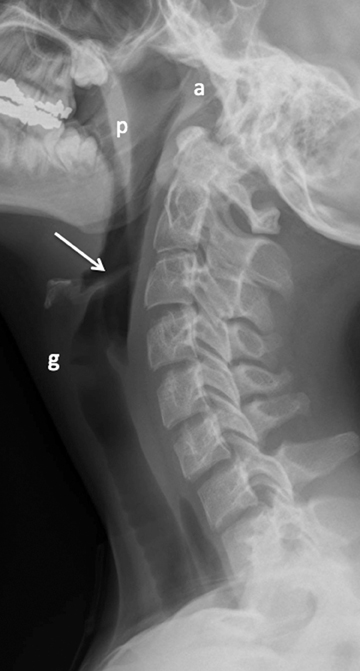
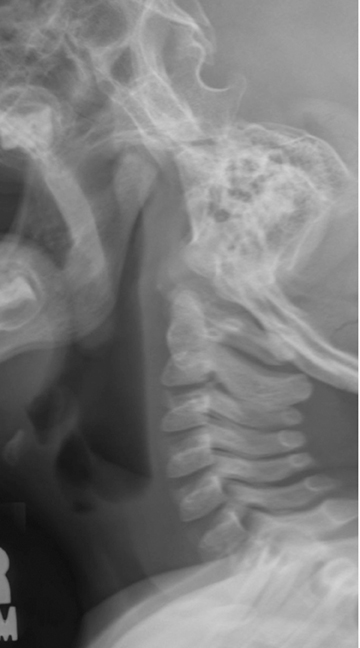
The esophagus begins at the lower aspect of the hypopharynx. To protect the airway from contents intended for the esophagus, the epiglottis is present as a flap-like roof that arises from the anterior larynx at the level of the hyoid bone. In the relaxed position, the epiglottis appears as a relatively thin density, angled cephalad and posteriorly (Figure 3). The aryepiglottic folds are thin walls of tissue that extend from the posterior upper larynx to the epiglottis, and these form the medial borders of the pyriform sinuses, which may be elucidated on a swallow study (not shown here). The epiglottis lowers to cover the glottis, which comprises the true vocal cords and the slightly higher vestibular folds, also known as the false cords. The glottis is at the level of the thyroid cartilage. Below the cords is the subglottic airway, supported by the cricoid cartilage and multiple cartilaginous rings continuing into the lower trachea.
Upper airway radiography
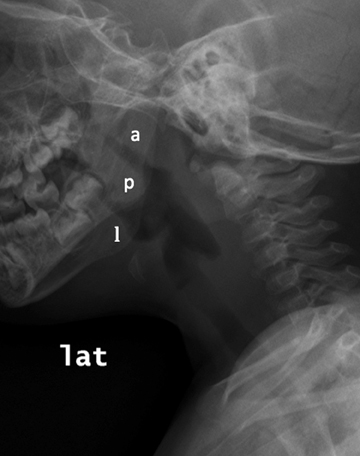
Radiographs of the airway above the thoracic inlet in the normal older child tend to be straightforward. The soft tissue at the posterior nasopharynx is the adenoidal tissue. The adenoids normally grow between ages 5 and 8 years and then begin to regress after that; they should be thinner by adolescence (Figure 3). The palatine tonsils are present at the posterior aspect of the soft palate, which divides the nasopharynx and oropharynx. The tongue is located at the anterior margin of the hypopharynx and the lingual tonsils reside at the base of the tongue. When enlarged, these tonsils will efface the vallecula, the space just anterior to the epiglottis. Below the level of the epiglottis and glottis, the subglottic airway should be well defined on the lateral view.
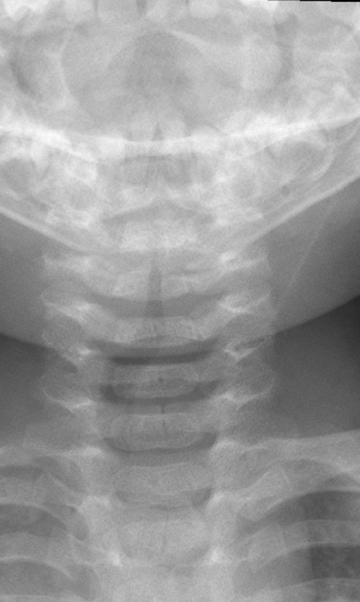
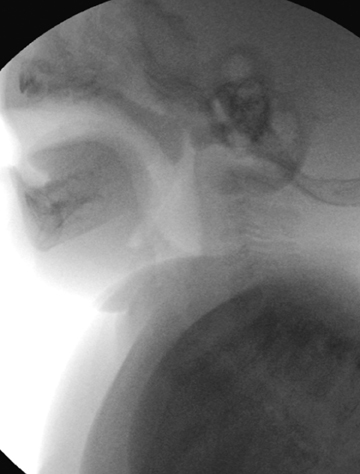
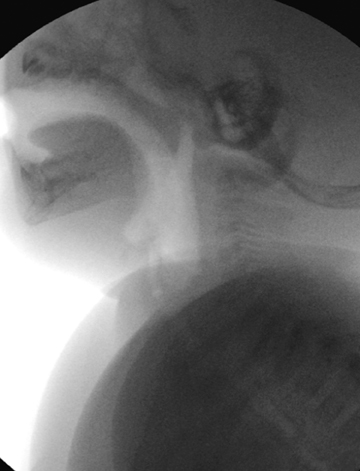
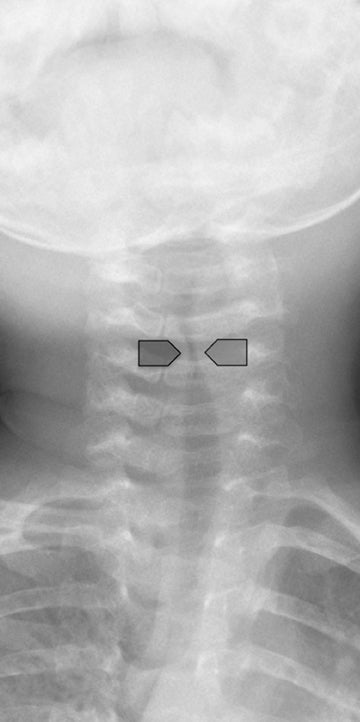
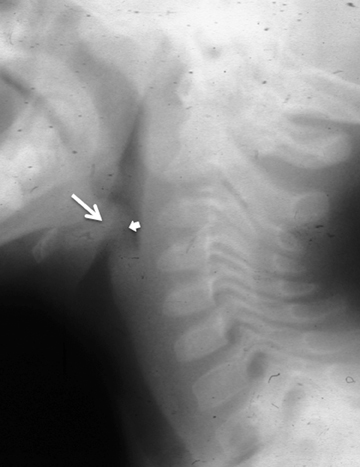
In infants and young children, evaluation of the soft tissues of the neck by radiography can be quite difficult, due in part to challenges in patient positioning and cooperation, and also in part to compliance of the soft tissues. A radiograph of the normal infant or young child’s neck captured during expiration or swallowing can appear remarkably abnormal and create concerns for abnormally thickened soft tissue. Figure 4 demonstrates just such a case in a normal toddler.
To further evaluate a case in which the radiograph is suspected to artificially augment the prevertebral soft tissues of the neck in a young child, fluoroscopy — ideally with the patient seated upright — can help to demonstrate the transient thickening of those soft tissues during normal respiration and swallowing (Figure 5). If fluoroscopy is not available, taking a repeat lateral radiograph while encouraging the child to exhale (perhaps, for instance, by having the child blow through a straw or blow bubbles) may clarify the findings.
Inflammation of the upper airway
Pediatric emergency physicians may rely on diagnostic imaging of a young child presenting with stridor, which is an inspiratory sound generated above the thoracic inlet due to a narrowed airway. Faced with a stridorous patient, the pediatrician will consider various causes of croup. Infectious croup may be viral, leading to laryngotracheobronchitis, or bacterial, leading to epiglottitis. Spasmodic croup is thought to be an allergic type of inflammation and is seen in 3% of children presenting with stridor.1
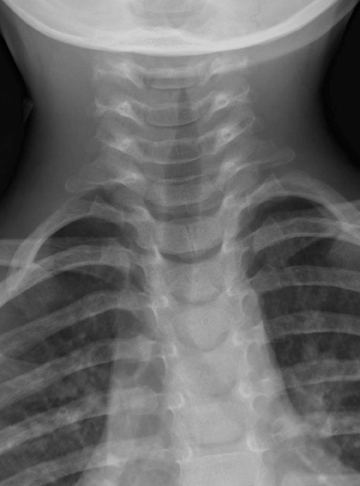
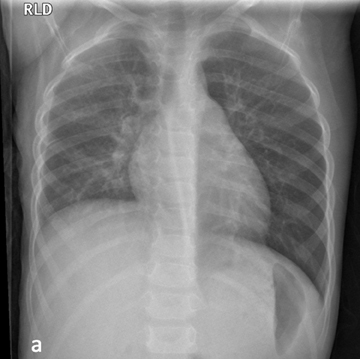
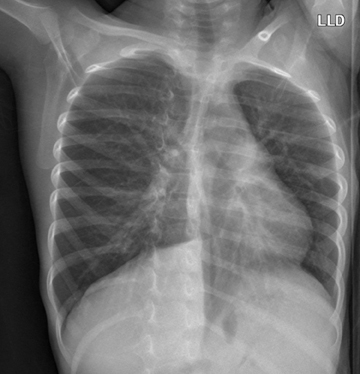
The clinical diagnosis of viral croup is usually readily made without imaging. Radiography will be pursued if there is any question of a more complex diagnosis, such as aspiration of a foreign body or retropharyngeal abscess. Viral croup usually presents with gradual onset in children between 6 months and 3-years-old. The most common cause is parainfluenza virus type 1, although many other viruses and Mycoplasma pneumoniae may also be an etiology.2,3 The illness causes a barking cough due to subglottic narrowing, which is appreciable on radiograph. It is important to note that approximately half of patients with laryngotracheobronchitis will have a normal neck radiograph.4,5 Figure 6 shows a classic example of viral croup radiographically with the subglottic narrowing reflected on the AP view by absence of the normal shouldering edges of the healthy subglottic airway and a tapered narrowing up to the glottis, called the “steeple sign.”6 On the lateral view, the normally sharp margins of the subglottic airway become hazy and ill-defined due to soft- tissue edema. The hypopharynx may be distended, related to inspiration against a fixed obstruction. Figure 7 demonstrates a normal AP film compared with that of viral croup.
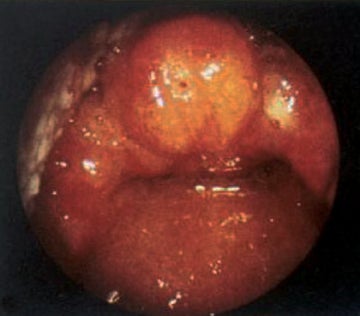
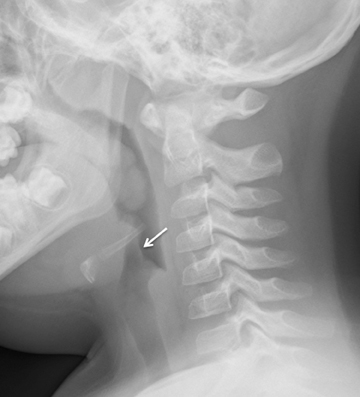
Bacterial croup, or epiglottitis, has a significantly lower incidence following routine immunization against Haemophilus influenzae type B,7 although numerous other causative agents, including streptococcus, staphylococci, pseudomonas, viruses, and candida8-11 may cause this life-threatening illness. The causative agent infects a slightly older age group than viral croup. Epiglottitis (or supraglottitis) is seen among children aged 2 to 8 years; it uncommonly produces the barking cough heard with laryngotracheobronchitis. The infection leads to cellulitis of the epiglottis, aryepiglottic folds, and surrounding soft tissues (Figure 8), and progresses rapidly within a matter of hours. The affected child will have difficulty handling the secretions, will appear toxic and anxious, and position himself in the tripod position (sitting upright and leaning forward, chin directed upward and mouth open) to optimize airway patency.
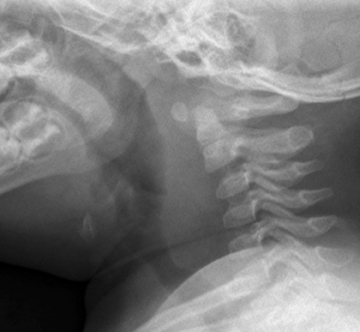
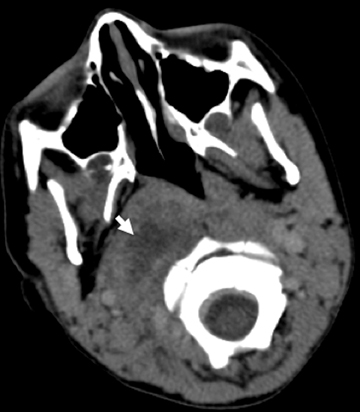
Radiography of the neck of a patient presenting in this way should be done gently and portably, with as little disturbance to the patient as possible. All precautions are taken to avoid agitating the patient in this degree of distress. A lateral view of the neck will show marked thickening of the epiglottis and the aryepiglottic folds (Figure 9). A particular entity, the omega-shaped epiglottis, may cause confusion on a radiograph. The omega-shaped epiglottis is a moderately uniformly thickened epiglottis that assumes a horseshoe shape and is associated with tracheomalacia. The aryepiglottic folds will be normal in this case (Figure 10).
Bacterial infection of the neck not involving the supraglottic airway may arise from lymphatic spread following tonsillitis or may be introduced by trauma or a foreign body and progress to a peritonsillar or retropharyngeal abscess. This process develops more gradually than supraglottitis and may be accompanied by drooling, voice change, and fever. The majority of these cases present in children under age 6.12,13 Radiography is an appropriate initial diagnostic tool in cases that are unclear clinically; CECT is usually diagnostic.
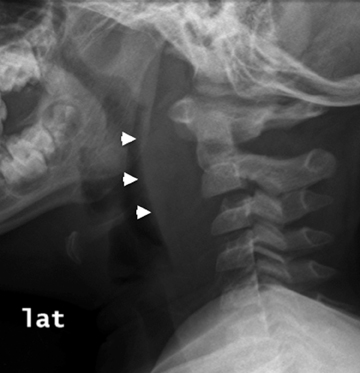
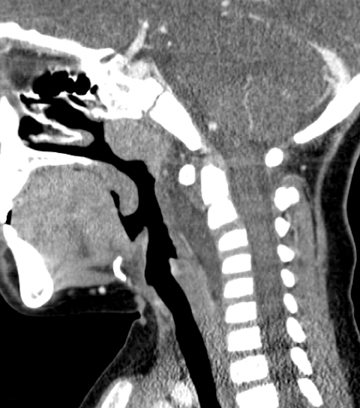
The peritonsillar or retropharyngeal abscess will create abnormal thickening of the prevertebral soft tissues. As discussed above, it is important to consider the possibility that the radiograph is abnormal although the child may not have an abscess. Figure 11 demonstrates a comparison of radiographs taken of 2 toddlers, each with prevertebral soft-tissue swelling, to illustrate how the compliance of a young child’s soft tissues may lead to the appearance of thickened tissues in the normal child (Figures 11). A retropharyngeal abscess or phlegmon will create soft tissue swelling of the prevertebral soft tissues extending from the adenoids to the lower cervical spine, with an outwardly convex margin displacing the airway anteriorly (Figures 11). Cross-sectional imaging will show a discrete fluid collection with enhancing margins if an abscess is present and will show ill-defined hypoattenuation in the parapharyngeal and retropharyngeal spaces in the setting of phlegmon. The retropharynx (see Figure 1) is a space that potentially can transmit infection into the mediastinum. Therefore, if retropharyngeal fluid is noted (Figure 12), it is important to make certain that the acquired images include the lower margin of any retropharyngeal inflammation to exclude mediastinal infection.
Foreign body aspiration or ingestion
Mobile infants and toddlers are at risk for foreign-body aspiration and ingestion. These patients present with a history of choking, while eating or playing, a persistent cough, or wheezing that does not respond to bronchodilators. The aspiration or ingestion is often unwitnessed.
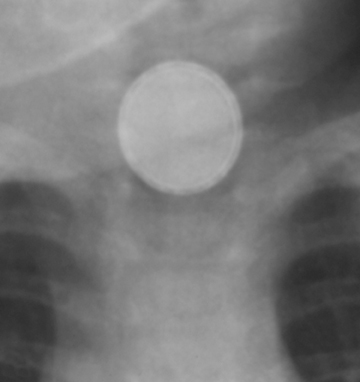
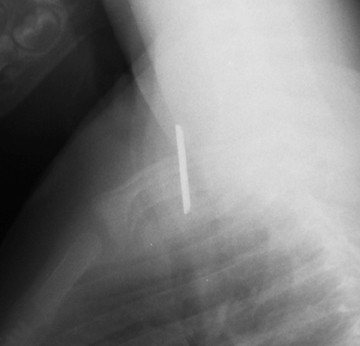
Although this review regards the airway and lungs, it is appropriate to mention ingested foreign body, since a high esophageal foreign body may present with respiratory symptoms. Two radiographic views are required to verify position of a foreign body. Of note, an esophageal foreign body may be long-standing and go unrecognized for many months. Of particular concern is ingestion of a button-shaped battery, which will rapidly result in a caustic injury to the esophageal mucosa and result in erosion, perforation and mediastinitis (Figure 13). Both the size of the battery and its composition influence whether a disc battery will become impacted and lead to injury.14 A small round lithium battery is radiographically distinct from a coin by its beveled margins and peripheral radiolucent rim (Figure 13).
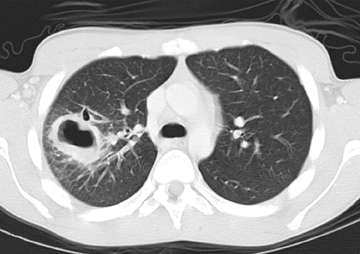
Diagnosing airway foreign bodies, most commonly seen in toddlers aged 1 to 3 years, can be challenging. When a foreign body enters an airway, it most commonly lodges in the right mainstem bronchus.15 Most (approximately 80%) aspirated foreign bodies will cause obstructive emphysema and the remaining cases will cause obstructive atelectasis. A minority will have no effect on aeration.16 Standard frontal and lateral chest radiographs are utilized, as well as special views such as forced expiratory view or bilateral decubitus views, to detect air trapping (Figure 14). Interestingly, a recent study intended to establish the diagnostic value of these special views17 showed that adding decubitus views to standard views increased false positives without increasing true positives. The test accuracy of expiratory views was found to be low. The study’s conclusion was that the clinical benefit of these additional radiographs is unclear and must be weighed against cost, time, and radiation exposure.
Wheezing in the pediatric patient
Aspiration of foreign bodies is just one of many causes of wheezing in infants and toddlers. Other causes that might be encountered in the emergency department include asthma, viral bronchitis, congenital airway abnormalities, cystic fibrosis, immunodeficiency diseases, gastrointestinal reflux, left-to-right shunts (“cardiac asthma”), and mediastinal masses, such as lymphoma, teratoma, foregut duplication cyst, and Morgagni hernia.
Vascular rings may present with wheezing, stridor, cough, or difficulty with feedings. The double aortic arch is the most common symptomatic ring. The right aortic arch with an aberrant left subclavian artery is also a vascular ring. Esophagrams are useful to exclude a vascular ring by showing a normal contour of the posterior esophageal margin on lateral view, without a posterior impression.18
Pneumonia in infants and children
Causative organisms of pneumonia are somewhat predictable based on the patient’s age and may be supported by classic radiologic findings.
In the newborn, pneumonia is typically caused by a bacterial organism, such as Group B Streptococcus, acquired during delivery. In the infant, pneumonias tend to be viral (respiratory syncitial virus, influenza type A and B, adenovirus, parainfluenzae virus types A and B, and rhinovirus). School-age children may have atypical pneumonias caused by viruses or Mycoplasma pneumoniae. Bacterial agents leading to pulmonary infection in the school age children may be Streptococcus pneumoniae, Staphylococcus aureus, Hemophilus influenzae, or Mycobacterium tuberculosis.19
Pediatric blunt chest trauma
Blunt chest trauma is an uncommon reason for admission in the pediatric population, recorded in only 5 to 12% of admissions for trauma, but it carries significant associated mortality.20 In children with multiple injuries, the presence of chest trauma increases mortality rate by 20 times compared to those without chest trauma.21 Approximately 85% of pediatric chest trauma is due to blunt force,22 from mechanisms such as motor vehicle accident, car versus pedestrian accident, abuse, and falls.20
Pulmonary parenchymal injuries
An important difference between children and adults with regard to blunt chest injury is that children have greater chest wall compliance than adults due to greater cartilage content and incomplete ossification of the ribs.22 This increased pliability makes rib fractures less common in children than adults, even in the setting of significant trauma.23 Additionally, this pliability results in increased transmission of traumatic forces to the pulmonary parenchyma and thus a relative increase in pediatric pulmonary parenchymal injuries compared with adults.21
Pulmonary parenchymal injury from blunt chest trauma can arise from several mechanisms, including direct compression, contrecoup injury, shearing forces, or laceration from fractured ribs.23 The resultant spectrum of injuries includes pulmonary contusion and pulmonary laceration.
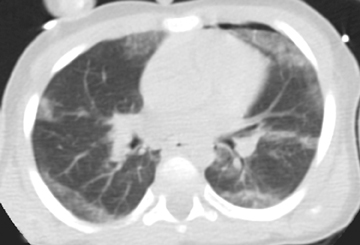
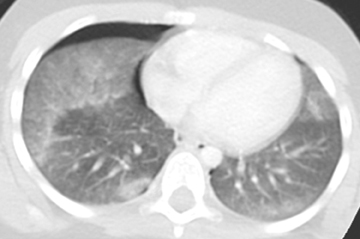
Pulmonary contusions are characterized by areas of alveolar and interstitial hemorrhage that do not follow anatomic boundaries (Figure 15).23 Contusions can be visible on chest radiographs, with a delay of up to 6 hours following trauma.24 On CT, Donnelly et al further characterized this form of injury by the typical sparing of the subpleural lung in 95% of reported cases.25
Parenchymal laceration is a more severe type of injury than contusion and is characterized by torn alveoli or bronchioles.23 Lacerations can be identified as cavities filled by fluid or air within an area of lung injury (Figure 16). Both contusions and lacerations typically require only conservative management, but are associated with increased mortality. Contusions greater than one third of the lung may require mechanical ventilation.22
Pleural injuries
Pneumothoraces in the setting of blunt chest trauma may result from leakage of air from damaged alveoli or small airways into the pleural space, or less commonly in conjunction with a central air leak from the tracheobronchial tree with pneumomediastinum.24 Radiographic findings include hyperlucency in the lower chest, depression of the ipsilateral diaphragm and deepening of the lateral costophrenic sulcus.23 Non-dependent crescentic lucency in the pleural space is seen on CT (deep sulcus sign).
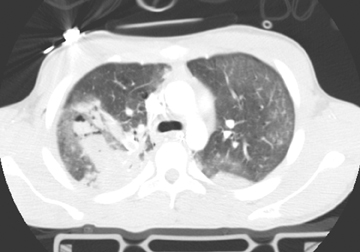
Hemothoraces are most commonly secondary to venous injury.21 On supine chest radiographs, pleural fluid creates a generalized increased opacity over the affected hemithorax.23 Crescentic-dependent fluid collection in the pleural space is seen on CT (Figure 17).
Due to the increased mobility of the pediatric mediastinum, large pneumothoraces or hemothoraces are at greater risk of causing respiratory or cardiovascular compromise in children.21
Tracheobronchial injuries
Tracheobronchial rupture occurs due to shear forces and traction when the chest is compressed against a closed glottis.26 This injury occurs in <3% of pediatric chest injuries, but carries a 30% mortality rate.22 As previously described, the increased elasticity of the pediatric chest wall compared to adults allows for transmission of large external forces to the child’s mediastinum without disturbing the chest wall structure. Accordingly, rib fractures are less common with tracheobronchial injury in children than in adults (24% versus 90%).27 Absence of rib fractures does not rule out major intrathoracic injury in children.26
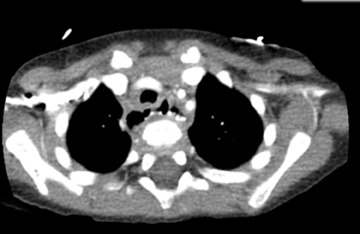
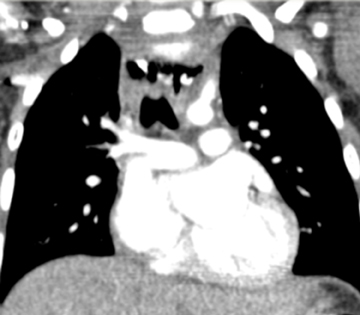
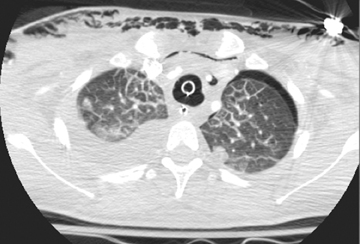
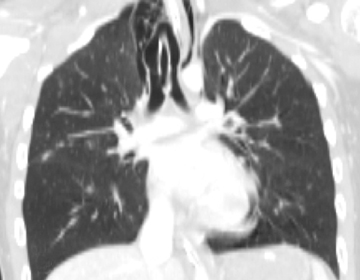
The most common location for tracheobronchial injury is the mainstem bronchi (80%), with most occurring within 2 cm of the carina. These are usually associated with pneumothorax or pneumomediastinum and should be suspected with continued air leak or despite chest tube decompression.26 Multidetector CT with multiplanar reformations (Figure 18) and virtual bronchoscopy are excellent techniques for demonstrating defects and resultant air leaks.24
Conclusion
Radiography is most commonly the initial modality for the evaluation of upper and lower airway abnormalities in the pediatric patient and may be followed by CT studies to further evaluate inflammatory processes and extent of trauma. An understanding of the expected radiographic appearance of the pediatric neck will assist the radiologist in determining whether cross-sectional imaging is necessary. Compliance of both the soft tissues of the neck and the chest wall in young patients influence imaging and susceptibility to traumatic insults, as reviewed in this article.
References
- Arthurton MW. Stridor in a pediatric department. Proc R Soc Med. 1970;63:712-714.
- Denny FW, Murphy TF, Clyde WA, et al. Croup: An 11-year study in a pediatric practice. Pediatrics. 1983;71:871-876.
- Baugh R, Gilmore BB. Infectious croup: A critical review. Otolaryngol Head Neck Surg. 1986;95:40-46.
- Skolnik NS. Treatment of croup: A critical review. Am J Dis Child. 1989;143:1045-1049.
- Cunningham MJ. The old and new of acute laryngotracheal infections. Clin Pediatr. 1992;31:56-64.
- Salour M. The steeple sign. Radiology. 2000;216:428-429.
- Progress toward eliminating Haemophilus influenzae type b disease among infants and children – United States, 1987-1997. MMRW Morb Mortal Wkly Rep. 1998;47:993-998.
- Glenn GM, Schofield T, Krober M. Group A streptococcal supraglottits. Clin Pediatr. 1990;29:674-676.
- Crysdale WS, Sendi K. Evolution in the management of acute epiglottitis: A 10-year experience with 242 children. Int Anesthesiol Clin. 1988;26:32-38.
- Lacroix J, Gauthier M, Lapointe N, et al. Pseudomonas aeruginosa supraglottitis in a six-month-old child with severe combined immunodeficiency syndrome. Pediatr Infect Dis J. 1988;7:739-741.
- Grattan-Smith T, Forer M, Kilham H, Gillis J. Viral supraglottitis. J Pediatr. 1987;110:434-435.
- Broughton RA. Nonsurgical management of deep neck infections in children. Pediatr Infect Dis J. 1992;11:14-18.
- Coulthard M, Isaacs D. Retropharyngeal abscess. Arch Dis Child. 1991;66:1227-1230.
- Slamon NB, Hertzog JH, Penfil SH, et al. An unusual case of button battery-induced traumatic tracheoesophageal fistula. Pediatr Emerg Care. 2008;24:313-316.
- Black R, Johnson D. Bronchospcopic removal of aspirated foreign bodies in children. J Pediatr Surg. 1994;29:682-684.
- Hedlund GL, Griscom NT, Celeveland RH, Kirks DR. Respiratory system. In: Practical pediatric imaging: diagnostic radiology of infants and children, 3rd edn. 1997 Philadelphia, PA: Willams & Wilkins.
- Brown JC, Chapman T, Chisholm SL, et al. The utility of adding expiratory and decubitus chest radiographs to the radiographic evaluation of suspected pediatric tracheobronchial foreign bodies. Scientific Abstract, Pediatric Academic Societies’ Annual Meeting, Vancouver, BC, Canada, 2010.
- Hernanz-Schulman M. Vascular rings: A practical approach to imaging diagnosis. Pediatr Radiol. 2005; 35:961-979.
- McIntosh K. Community-acquired pneumonia in children. NEJM. 2002;346:429-437.
- Ventura, AM, Goes PF, Otoch JP, Fenandes JC. Traumatic airway disruption in a child: A diagnostic challenge. J Pediatr. (Rio J) 2005;81:179-182.
- Donnelly LF. Imaging issues in CT of blunt trauma to the chest and abdomen. Pediatr Radiol. 2009;39:S406-413.
- Sartorelli KH, Vane DW. The diagnosis and management of children with blunt injury of the chest. Sem Pediatr Surg. 2004;13:2:98-105.
- Sivit CJ. Pediatric thoracic trauma: Imaging considerations. Emerg Radiol. 2002;9:21-25.
- Westra, SJ, Wallace, EC. Imaging evaluation of pediatric chest trauma. Radiol Clin N Am. 2005;43:267-281.
- Donnelly LF, Klosterman LA. Subpleural sparing: A CT finding of lung contusion in children. Radiology. 1997;204:385-387.
- Mandell DL. Traumatic emergencies involving the pediatric airway. Clin Ped Emerg Med. 2005;6:41-48.
- Grant WJ, Meyers RL, Jaffe RL, Johnson DG. Tracheobronchial injuries after blunt chest trauma in children--hidden pathology. J Pediatr Surg. 1998;33:1707-1711.
