Congenital and Acquired Abnormalities of the Cardiac Valves: Spectrum of 64-Slice Multidetector CT Findings
Images






Dr. Colen is a third-year Radiology Resident at Massachusetts General Hospital (MGH) and Harvard Medical School (HMS), Boston, MA. Dr. Colen received her medical degree in 2003 from Ponce School of Medicine in Ponce, Puerto Rico, where she also completed her internship. In 2003, Dr. Colen became the first medical student from Puerto Rico to be accepted in the Radiology Residency program at MGH and the first female medical student from Puerto Rico to be accepted into a residency at MGH and HMS. When this article was written, Dr. Dodd was a Cardiac Fellow at MGH, Boston, MA. Dr. Roedl is a Research Fellow, MGH, Boston, MA. Dr. Cury is a Cardiac Radiologist, MRI and CT, and the Director of Clinical Cardiac MRI, MGH; and an Instructor of Radiology, HMS, Boston, MA. Dr. Abbara is the Director of Noninvasive Cardiac and Vascular Imaging, and the Director of Education, Cardiac MR and CT Program, MGH; and an Instructor, HMS, Boston, MA.
Multidetector computed tomography (MDCT) is expected to revolutionize the way the evaluation and work-up of the cardiac patient is performed. Although invasive coronary and cardiac angiography maintains a 0.2-mm spatial resolution versus 0.4-mm in 64-slice MDCT, the emergence of retrospectively electrocardiographic-gated 64-slice MDCT has resulted in high spatial resolutions that are not achievable with other noninvasive cardiac imaging modalities. The purpose of this article is to describe the MDCT qualitative techniques used to evaluate valvular lesions and to describe and illustrate a spectrum of congenital and acquired valvular disease with 64-slice MDCT.
Cardiac valvular disease is a cause of considerable morbidity and mortality. In 2006, the total estimated mortality attributable to valve disease in the United States was 19,989 deaths. Aortic valve disease accounted for 12,471 deaths, and mitral valve disease for 2759 deaths, with the remainder divided evenly between the tricuspid and pulmonary valves. 1
Transthoracic and transesophageal echocardiography are the primary imaging modalities used to evaluate valve disease, although in some patients it may be difficult to obtain accurate visualization and evaluation of valvular lesions. 2 User-operator dependency is an inherent disadvantage, and some patients have an inadequate acoustic window for scanning. Transesophageal echocardiography is an invasive procedure and is contraindicated in patients with recent esophageal surgery, recent oral ingestion, unstable cervical spine injuries, and unevaluated gastrointestinal bleeding. Although cardiac magnetic resonance imaging (MRI) is currently regarded as the noninvasive reference standard for valve evaluation, it is costly, time-consuming, and has limited availability. Contraindications for MRI include pacemakers, implantable cardiac defibrillators, intravascular stents <6 weeks after implantation, aneurysmal clips, heart valves installed before 1996, and claustrophobia.
Multidetector-row computed tomography (MDCT) is an emerging modality in noninvasive cardiac imaging. Using data acquired throughout the cardiac cycle, it is possible to reconstruct multiple reconstructions at 10% increments of the R-R interval. These can then be combined sequentially to provide functional imaging data sets in a cine loop that allows evaluation for valvular leaflet morphology and function. The purpose of this article is to describe the cardiac MDCT techniques that allow optimal depiction of the cardiac valves, describe the pathophysiology of a spectrum of valvular diseases, and illustrate their appearances on 64-slice MDCT.
MDCT scan protocol
For this manuscript, images have been acquired using a 64-slice MDCT scanner (Sensation 64, Siemens Medical Solutions, Erlangen, Germany). Our protocol involves intravenous beta-blocker administration (5 to 25 mg of metoprolol) before each scan to achieve a heart rate of 60 bpm. Nitroglycerin tablets (0.6 mg) are administered sublingually just before scanning. The scan range, based on the scout images, is planned from the carina to the base of the heart. Scans are acquired in full inspiration. A timing bolus scan is performed to determine peak opac-ification in the ascending aorta using an injection of 20 mL of iodinated contrast (iopamidol 300 mgI/mL) at 5 mL/sec is followed by a 20-mL saline bolus chaser. For the cardiac scan, 60 to 90 mL of iopamidol 370 mgI/mL is injected into the antecubital vein at 5 mL/sec followed by 40 mL of saline at 5 mL/sec, and the scan is initiated with a delay according to the timing bolus delay. Cardiac acquisition parameters include a collimation of 0.6 mm, slice reconstruction thickness of 0.75 mm, slice overlap of 0.5 mm, gantry rotation time of 330 msec, table feed of 3.8 mm/rotation, tube voltage of 120 kVp, and tube current of 850 mAs. Axial images are reconstructed with a slice thickness of 0.75 mm with 0.4-mm slice overlaps using a half-scan reconstruction algorithm to achieve a temporal resolution of 165 msec in the center of gantry rotation. In subjects with a heart rate >65 bpm or insufficient image quality due to motion artifacts, multisegment reconstruction is performed. We find the initial reconstruction phase that produces optimum image quality is 65% of the cardiac cycle, usually representing the most motion-free phase of the cardiac cycle. Multifunctional data sets are reconstructed at 10% intervals of the R-R interval for functional depiction of the valves throughout the cardiac cycle. 3 Data sets are transferred to a workstation (Leonardo, Siemens Medical Solutions, Forchheim, Germany) and multiplanar reformatted images of each valve are rendered. Valve motion is reviewed in anatomic appropriate planes in both static multi-phase reconstruction images and in cine-mode images.
Mitral stenosis
Global pathophysiology
Mitral stenosis (MS) is a narrowing of the inlet valve of the left ventricle. It commonly leads to inadequate diastolic filling of the left ventricle. The majority of cases are acquired and are secondary to rheumatic heart disease. Less common causes include systemic lupus erythematosus, rheumatoid arthritis, and pulmonary cardiac carcinoid. Less commonly, left atrial myxoma, thrombus, and malignant tumor may prolapse into the mitral orifice, causing "hemodynamic MS." Massive mitral annular calcification may cause functional MS. 4,5 Infective endocarditis with large vegetations may cause thickening and stenosis of the leaflets. Rarely, congenital MS may be caused by a "parachute mitral valve" (subvalvular MS) or hypoplastic left heart syndrome. 6
Global radiological assessment
Criteria for grading MS using valve area, transvalvular flow rate, and transvalvular pressure gradient are well established in echocardiography. 7 The normal mitral valve orifice usually measures between 4 and 6 cm 2 . Narrowing of the valve area to <2.5 cm 2 impedes the free flow of blood into the left ventricle. When the orifice is reduced to <1 cm 2 , severe MS results. Willmann et al 3 found that MDCT allows good to excellent morphological detail of the mitral valve and apparatus (namely, annulus, leaflets, and papillary muscles), with the exception of the tendinous chords.
Morphologic changes that are seen on cardiac MDCT in patients with chronic MS result from left atrial outflow obstruction. An enlarged left atrium (>4 cm in anteroposterior diameter at the level of the aortic root) and left atrial appendage are commonly seen, combined with a normal-sized left ventricle. This leads to chronic venous pulmonary hypertension and, eventually, right heart failure develops. 8 Additional cardiac MDCT findings include an enlarged main pulmonary artery (PA) of >3 cm at the bifurcation, increased lung parenchymal groundglass opacity, increased septal lines (Kerley B lines) and pleural and pericardial effusions.
Congenital: Parachute mitral valve
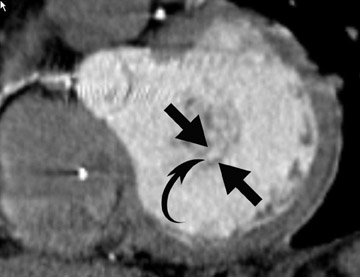
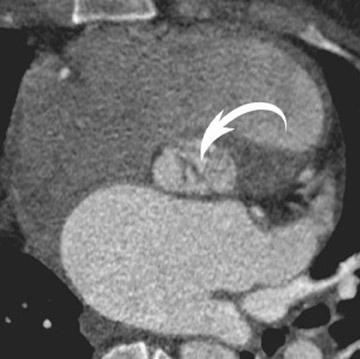
Pathophysiology- Parachute mitral valve is caused when the chordae converge on a major papillary muscle instead of diverging to insert into 2 papillary muscles. The usual 2 mitral valvular leaflets and commissures are present. The effect creates a stenotic mitral valve, since the leaflets are held in close apposition. The only effective communication between the left atrium and the left ventricle is through the interchordal spaces. Parachute mitral valve associated with subaortic stenosis occurs 40% of the time and is termed Shone's syndrome. 6
Radiological assessment- On MDCT, the leaflets resemble the canopy of a parachute; the chordae, its shrouds or strings; and the papillary muscle, the harness. The chordae are often short and thick. This, coupled with their convergent papillary insertion, allows little mobility of the leaflets (Figure 1).
Acquired mitral stenosis: Rheumatic heart disease
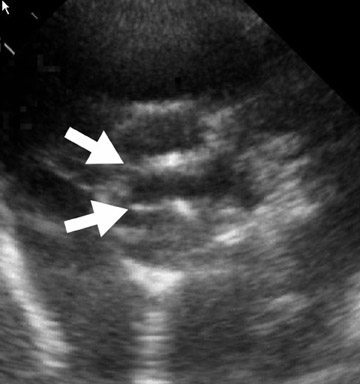
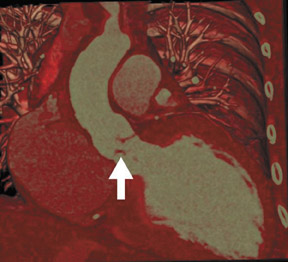
Pathophysiology- Rheumatic heart disease (RHD) is the most serious complication of rheumatic fever (RF) and occurs following pharyngitis with group A beta-hemolytic Streptococcus pyogenes . 9,10 Although the most common cause of MS is RF, the prevalence of MS has decreased due to the decline in RF because of antibiotic treatment of "strep throat." Approximately 40% of patients with RHD have isolated MS. Conversely, rheumatic involvement is present in 99% of stenotic mitral valves excised at the time of mitral valve replacement.
Radiological assessment- Calcification of the mitral valve leaflets are classically seen in MS of rheumatic origin. There is a narrowing of the orifice during diastole. The left atrium can be enlarged, although usually not to the extent seen in mitral regurgitation (Figure 2). 3
Acquired mitral stenosis: Cardiac myxoma prolapse into mitral orifice
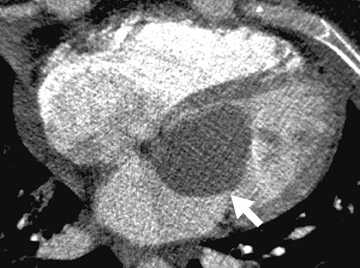
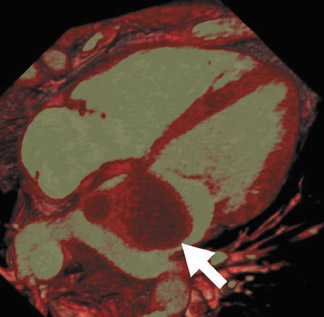
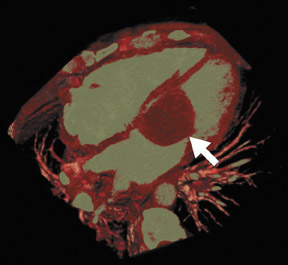
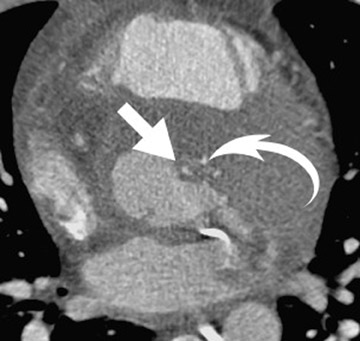
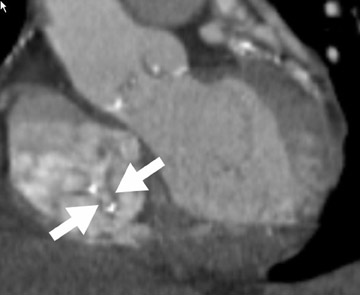
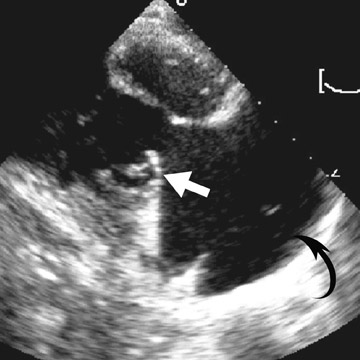
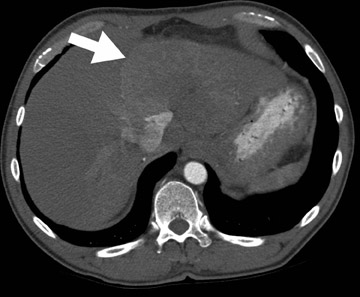
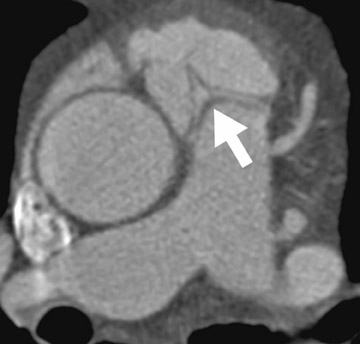
Pathophysiology- Cardiac myxoma is a benign neoplasm and the most common primary tumor of the heart. Clinical presentation depends on the location and morphology of the tumor and its tendency to embolize. It classically arises as an endocardial intracavitary mass in the left atrium attached to the fossa ovalis of the interatrial septum. 11 Fibrous lesions are more likely to produce valvular obstruction (mitral stenosis), while polypoid, predominantly myxoid, lesions are more likely to embolize. 12,13
Radiological assessment- For large lesions causing significant hemodynamic compromise, a pattern mimicking that of MS and pulmonary venous hypertension may be seen on radiographs. On MDCT, cardiac myxomas appear with a well-defined spherical or ovoid intracavitary shape, with smooth contours (Figure 3A). Tumor enhancement is usually homogenous, although heterogeneous attenuation of myxomas may reflect hemorrhage, necrosis, cyst formation, fibrosis, or calcification. 14 Multifunctional data reconstruction provides a cine loop that is useful for demonstrating tumor motion and possible prolapse of the mass into the mitral orifice (Figures 3B through 3D).
Mitral regurgitation
Global pathophysiology
Mitral regurgitation occurs because of incomplete coaptation of the mitral valve leaflets, thereby allowing backward blood flow into the left atrium. Causes include: 1) leaflet abnormalities, such as leaflet thickening in rheumatic heart disease, perforation in infective endocarditis, or floppy redundant leaflets; 2) chordae tendinae abnormalities seen in mitral valve prolapse; 3) chordae tendinae rupture from trauma, infection, congenital malformation, or cystic medial necrosis; 4) papillary muscle rupture from infarct; and 5) mitral annular dilation from left ventricular dilation of any cause.
Global radiological assessment
In the acute setting, such as acute myocardial infarction, rapid left atrial and ventricular volume overload result in acute severe left heart failure and acute pulmonary edema. In individuals with chronic mitral regurgitation, adaptation of the left atrium and the left ventricle to the volume load results in left atrial and left ventricular dilation, with less severe pulmonary vascular congestion, and, rarely, if ever, pulmonary hypertension. The left ventricular mass increases with increased chamber volume, resulting in thickening of the left ventricular myocardium. Pulmonary arterial dilation and right heart dysfunction are, therefore, less commonly seen in compensated patients. 15 If left atrial enlargement is severe, chest radiography may show a double density sign over the right heart border, which represents the edge of the left atrium, and there may be splaying of the carina. Some complications that are seen in MR include left ventricular failure, atrial fibrillation, and left atrial thrombus.
Recent studies show a strong correlation between the regurgitant orifice area at MDCT and the degree of regurgitation. 16,17 Moreover, MDCT accurately depicted the underlying morphological cause of mitral regurgitation, with the exception of rupture of the chordae tendinae. 16,17 Difficulty in the visualization of the tendinae should be overcome by increased spatial and temporal resolution of more recently developed scanners. The prognosis of patients with mitral regurgitation depends on the underlying etiologies and the state of the left ventricular function with an 80% 5-year survival rate. 18,19
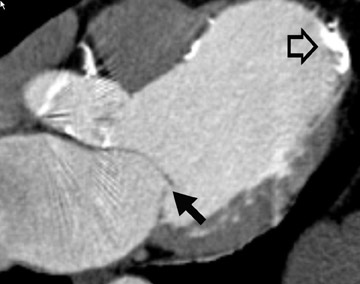
Secondary mitral regurgitation: Mitral annular dilatation secondary to cardiomyopathy
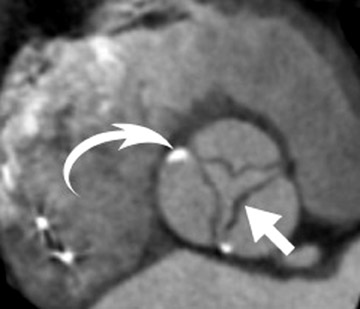
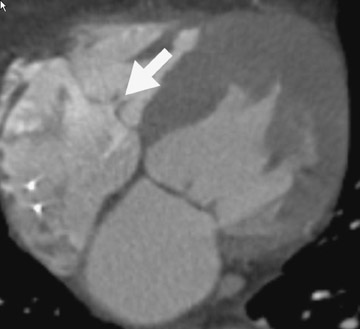
Pathophysiology- The most common cause of secondary mitral regurgitation is ventricular dilatation causing stretching of the mitral valve annulus and displacement of the papillary muscles.
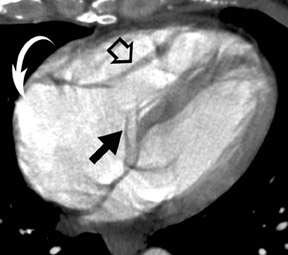
Radiological assessment- Cardiac MDCT has recently been shown to illustrate noncoaptation of the mitral leaflets and to estimate the severity of MR accurately (Figure 4). 16,17 Ancillary findings as to the mechanism of mitral regurgitation, such as left ventricular enlargement and the resultant atrial dilatation, are depicted well on CT.
Primary mitral regurgitation: Myxomatous degeneration with mitral valve prolapse
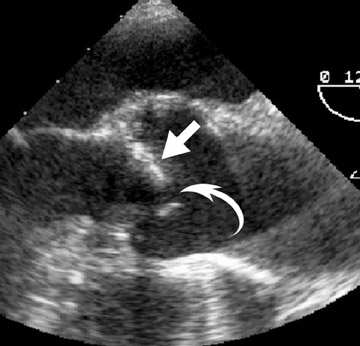
Pathophysiology- Mitral valve prolapse (MVP) is the overall most common cause of MR and occurs when the valve leaflets "billow" backward into the atrium during systole. The concept that there is an underlying defect of the connective tissue, which is possibly systemic, is favored currently. This is further suggested by the finding that MVP is a prevalent feature in connective tissue disease, such as Marfan syndrome and Ehlers-Danlos syndrome. Deposition of mucoid material causes myxomatous degeneration of the mitral valve leaflets, and associated structures and the collagenous structures within tendinae are decreased. 20
Radiological assessment- Lengthening of the chordae tendineae is a cardinal feature of the MVP syndrome. The chordae may be congenitally abnormally long or may become abnormal secondary to trauma, infection, or cystic medial necrosis. The posterior leaflet is most commonly affected. Annular dilatation is characteristic, a finding that is rare in other causes of primary mitral insufficiency. Cardiac MDCT may show doming and hooding of the leaflets, which may be thickened. Lack of complete coaptation of the valve leaflets in systole results in MR. The entity may be complicated by acute rupture of the chordae tendinae (Figure 5), which is usually a cata- strophic event that leads to profound cardiogenic and respiratory shock.
Aortic stenosis
Global pathophysiology
Aortic stenosis is a narrowing of the aortic valve orifice which causes resistance to blood flow across the aortic valve. In young patients, the major causes are congenital malformation of the cusps and rheumatic fever. 20-23 In older individuals ( >60 years of age), the major causes are calcification of a bicuspid aortic valve or "degenerative" senile calcification of a morphologically normal valve. 22 Average survival depends on the clinical symptoms as follows: congestive heart failure, 1.5 to 2 years; dyspnea, 2 years; and an-gina, 3 years.
Global radiological assessment
Cardiac multiplanar reformats allow imaging in any plane. We usually image the valves in a true-2-chamber view, 4-chamber view, and short axis view. We also evaluate the aortic valve in a true cross-sectional plane, similar to echocardiography. This has 2 advantages: 1) they are usually the optimum imaging planes with which to view the valves; and 2) they are the views cardiologists are most familiar with. On cardiac MDCT, the 3 cusps of the normal aortic valve form a "Y," the sinuses forming a clover-leaf appearance. The normal aortic valve area measures 3 to 4 cm 2 . Planimetric measurement of an aortic valve area <2 cm 2 is clinically significant, and <0.8 cm 2 defines critical aortic stenosis. Studies have shown a strong correlation between the degree of aortic valve calcification assessed on MDCT and transvalvular gradients and aortic valve area measured on Doppler echocardiography. 24 One group of researchers has suggested that patients with severe aortic stenosis and marked calcification on MDCT belong to a high-risk subgroup that may benefit from early aortic valve replacement, even in the absence of symptoms. 25 Concentric left ventricular muscle hypertrophy is seen in all cases, regardless of the pathogenesis.
Congenital aortic stenosis: Bicuspid aortic valve
Pathophysiology- Bicuspid aortic valve (BAV) is the most common congenital cardiac malformation. It results from complex abnormal cusp formation during valvulogenesis, not just the fusion of 2 cusps. Since BAV causes premature fibrosis and calcification of the aortic valves, aortic stenosis is the most common complication of BAV. In contrast to the degenerative form of AS, AS from bicuspid valve presents earlier, at an age range of 30 to 50 years.
Radiological assessment- In the absence of the normal 3 cusp clover-leaf appearance, a bicuspid valve can be easily diagnosed. The classical bicuspid valve shows 2 symmetric aortic cusps. During diastole, the open bicuspid aortic valve assumes an ellipsoid shape (Figure 6).
Secondary aortic stenosis: Degenerative aortic valve stenosis
Pathophysiology- Degenerative (se-nile) calcific stenosis is thought to develop secondary to normal "wear and tear" from hemodynamic injury and manifests itself usually in the eighth decade. In de-generative aortic stenosis, the degree of calcification is the strongest independent risk factor for disease progression and an adverse clinical outcome. 21,25-27
Radiological assessment- MDCT shows 3 distinct separate cusps, thickened by calcifying fibrosis and distorted by calcified nodular excrescence. The "updoming" of valve leaflets and the stenosis of the aortic valve orifice is also seen (Figure 7).
Aortic regurgitation
Global pathophysiology
Aortic regurgitation is the diastolic flow of blood from the aorta into the left ventricle. Incompetence of the aortic valve or any disturbance of the valvular apparatus (eg, leaflets, annulus of the aorta) results in aortic regurgitation and may be caused by abnormalities of the valve leaflets (commonly due to rheumatic heart disease, bacterial endocarditis) or by dilatation of the aortic root (commonly due to Marfan syndrome, syphilitic aortitis). 28
Global radiological assessment
On cardiac MDCT, the severity of valvular dysfunction may be reflected by the size of the aortic caliber, as well as how far toward the aortic arch the aortic dilatation extends. The impact of aortic regurgitation on cardiac hemodynamics is dependent on the speed of onset of regurgitation. In acute regurgitation, such as in infective endocarditis (IE) or aortic dissection, the left ventricle has no time for adaptation, whereas in chronic regurgitation hypertrophy of the left ventricle allows some adaptation and clinical presentations may be less severe. 19
Acute aortic regurgitation: Infective endocarditis
Pathophysiology- Infective endocarditis (IE) is an infection of the endocardial surface of the heart, most com- monly the cardiac valves. Most people have a pre-existing underlying cardiac valve condition predisposing them to the development of left-sided IE. Streptococcus viridians is the most common organism.
Radiological assessment- In the acute setting, vegetations can be clearly depicted (Figure 8A) and are classically on the ventricular side of the aortic valve in the direction of intracardiac blood flow. Multifunctional reconstructions can depict incomplete coaptation of the cusps (Figures 8B and 8C). Multidetector CT may also show complications, such as aortic wall erosion by infected vegetations and subsequent endovascular leak into the epicardial fat (Figure 9).
Tricuspid regurgitation:
Global pathophysiology
Tricuspid regurgitation occurs because of incomplete coaptation of the tricuspid valve leaflets, allowing blood to flow backward into the right atrium. Similar to mitral regurgitation, tricuspid regurgitation occurs when there is an abnormality of any of the 5 components of the atrio-ventricular valve apparatus, which includes the leaflets, chordae, annulus, papillary muscles and adjacent right ventricular (RV) muscle abnormalities. Ebstein's anomaly accounts for the most common congenital form of this abnormality. Other causes include endocarditis, carcinoid, floppy valve syndrome, and connective tissue disorders, such as Marfan syndrome.
Global radiographic assessment
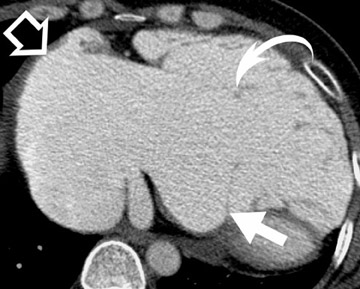
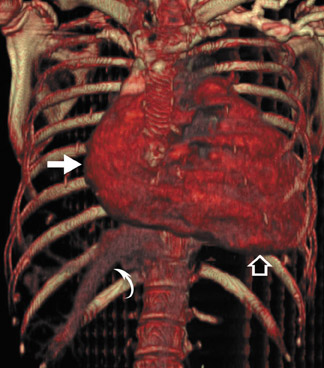
The normal tricuspid valve area measures 3 to 9 cm 2 . On MDCT, tricuspid regurgitation is seen by incomplete leaflet coaptation and the presence of reflux of contrast into the inferior vena cava and hepatic veins during the first pass of a contrast-enhanced CT. The presence of reflux can be associated with tricuspid regurgitation and has a very high specificity and sensitivity. Tricuspid regurgitation results in right atrial and ventricular dilation (Figure 10) and clockwise cardiac rotation, in severe cases. Furthermore, the etiology of tricuspid regurgitation can usually be inferred from associated morphologic abnormalities depicted with cardiac MDCT. For example, enlargement of the pulmonary arteries indicates that pulmonary hypertension may have mediated the tricuspid regurgitation. 8 Left heart dilation with left ventricular dysfunction indicates left heart disease as the likely etiology of the right heart dysfunction. Rheumatic heart disease may cause calcification of the tricuspid as well as the mitral valve leaflets.
Congenital tricuspid regurgitation: Ebstein's anomaly
Pathophysiology- The pathophysiology and morphology of Ebstein's anomaly reflect the embryological develop- ment of the tricuspid valve. The anterior leaflet develops first, arising from the mesenchyme. The posterior and septal leaflets arise through the creation of a diverticulum and the undermining of the right ventricular myocardium. Failure or partially occurring undermining of the myocardium causes Ebstein's anomaly. This explains why the proximal portion of the anterior valve leaflet is not usually displaced and the anterior leaflet is the most dysplastic leaflet. 29
Radiological assessment- Ebstein's anomaly is defined by apical displacement of the septal and posterior tricuspid valve leaflets, leading to atrialization of the inlet of the right ventricle with a variable degree of dysplasia of an enlarged anterior leaflet. The posterior and septal tricuspid valves may be deficient or absent (Figure 11). 29,30 Most patients exhibit septal bowing due to the increased volume of the right side of the heart. Roughly 90% of patients have a concomitant patent foramen ovale or secundum atrial septal defect.
Since the surgical method is tailored for each patient, accurate assessment of morphologic features and type of Ebstein's anomaly is important and cardiac MDCT is ideal for such evaluation. 18
Acquired tricuspid regurgitation: Carcinoid heart disease
Pathophysiology- Cardiac involvement has been recognized in more than half of the patients with carcinoid syndrome and heralds a worse prognosis. 31 Carcinoid tumors are rare neuroendocrine malignancies that arise from neural crest amine precursor uptake decarboxylation cells. Approximately 90% of all carcinoid tumors are located in the gastrointestinal system. Carcinoid syndrome is characterized by facial flushing, intractable secretory diarrhea, and bronchoconstriction. Carcinoid valve lesions characteristically manifest as fibrous white plaques located on the valvular and mural endocardium. The valve leaflets are thickened, rigid, and reduced in area with fibrous tissue proliferation that is present on both the atrial and ventricular surfaces of the valve.
Radiological assessment- Carcinoid heart disease is characterized by pathognomonic plaquelike deposits, resulting in fibrous endocardial thickening, retraction, and fixation of valvular cusps of the tricuspid valve and, less frequently, the pulmonary valve. Tricuspid regurgitation is found in nearly all patients with carcinoid heart disease and leads to right-heart enlargement. 32,33 Cardiac MDCT in cardiac carcinoid depicts thickened tricuspid valves, which show restricted movement. There may also be thickened chordinae tendinae (Figure 12). There is almost always concomitant right atrial enlargement, and, in severe cases, an enlarged superior vena cava and inferior vena cava. The liver also contains hypervascular carcinoid metastases.
Pulmonary stenosis
Global pathophysiology
Pulmonic stenosis is most commonly congenital and can be valvular (90%), subvalvular, or peripheral (supravalvular). Valvular pulmonic stenosis is typically an isolated anomaly and comprises 10% of all congenital heart disease. Acquired causes of pulmonic stenosis are rare and include carcinoid, rheumatic fever, and infective endocarditis. Although uncommon in isolated valvular pulmonic stenosis, a bicuspid valve is found in as many as 90% of patients with tetralogy of Fallot. 34
Global radiographic assessment
The normal trileaflet pulmonic valve area measures 2 cm 2 /m 2 of body surace area (2.5 to 4.0 cm 2 ). Mild pulmonic stenosis is defined as <1 cm 2 and severe as <0.5 cm 2 . Classically, the 3 leaflets are thin and pliant, with partially fused commissures, resulting in a conical or dome-shaped structure with a narrowed central orifice. Poststenotic pulmonary artery dilatation may occur owing to "jet-effect" hemodynamics. 35
Valvular pulmonary stenosis: Dysplastic pulmonic valves
Pathophysiology- Approximately 10% to 15% of patients with valvular pulmonic stenosis have dysplastic pulmonic valves, which are composed of myxomatous tissue. Failure of normal development of the pulmonic valves at 6 to 9 weeks gestation can result in the following malformations: fusion of 2 cusps, 3 leaflets that are thickened and partially fused at the commisures, or a single cone-shaped valve.
Radiological assessment- MDCT depicts a valvular annulus that is small, and the supravalvular area of the pulmonary trunk is usually hypoplastic. Poststenotic dilatation is less common with dysplastic valve stenosis than it is with classic pulmonic stenosis (Figure 13). 35 Approximately two thirds of patients with Noonan syndrome have pulmonic stenosis due to dysplastic valves. 36
Conclusion
Cardiac MDCT continues to broaden its role in the evaluation of cardiac valve diseases. It may provide an alternative imaging method for valve evaluation in patients in whom alternative imaging modalities are unable to provide optimal valve depiction.