Abdominal Presentation of COVID-19
Images

CASE SUMMARY
A 64-year-old with hypertension, hypercholesterolemia, and recent exposure to a family member with coronavirus disease 2019 (COVID-19) presented to the emergency department with a two-day history of abdominal pain, nausea, vomiting, diarrhea, and low-grade fever. The patient denied fatigue, myalgia, headache, cough, sore throat, shortness of breath, and chest pain/ tightness. Physical examination was remarkable for a temperature of 101°F, oxygen saturation of 98%, and generalized abdominal tenderness. Laboratory values were remarkable for slightly elevated alanine aminotransferase (ALT) and aspartate aminotransferase (AST). An abdominal–pelvic CT scan with intravenous contrast was subsequently performed to assess for acute pathology. Based upon CT observations at the imaged lung bases, an anterior–posterior chest radiograph was obtained.
IMAGING FINDINGS
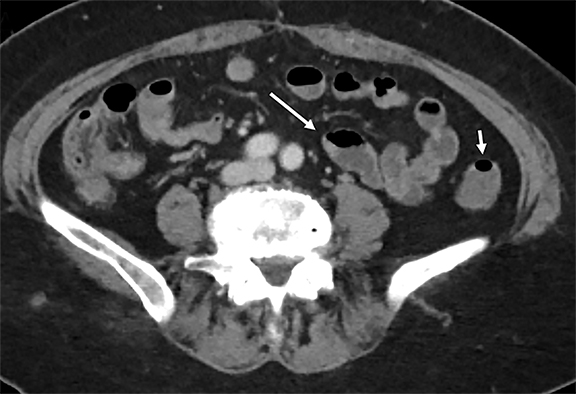
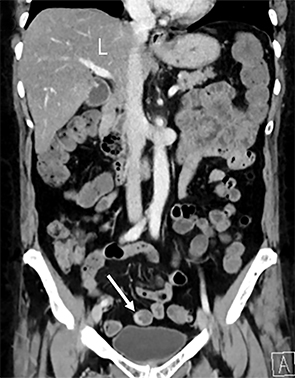
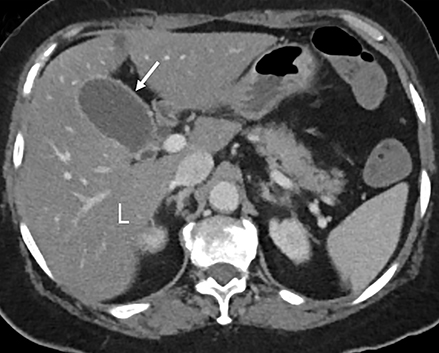
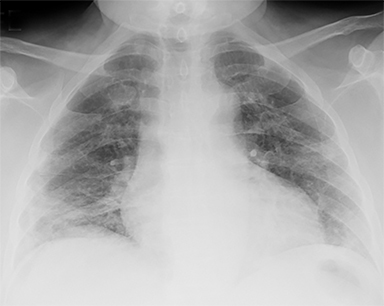
The abdominal-pelvic CT demonstrated non-dilated, fluid-filled small bowel and colon (Figure 1) with air fluid levels and intermittent noncontiguous small-bowel mucosal hyperemia and thickening (Figure 2). Additional pertinent findings included a fatty liver (Figures 2,3) and gallbladder wall hyperemia (Figure 3). There was no bowel obstruction, appendicitis, pancreatitis, diverticulitis, ascites, or adenopathy. Lung bases on the CT revealed bilateral multifocal peripheral and subpleural ground-glass opacities (GGO) (Figure 4). The chest radiograph (Figure 5) demonstrated bilateral mid- and lower-lung zone multifocal, peripherally predominant nodular and coalescent opacities. A presumed diagnosis of COVID-19 pneumonia with enteritis and hepatitis was established based upon the patient’s recent COVID-19 exposure, radiologic findings, and laboratory analysis.
DIAGNOSIS
COVID-19, confirmed with viral antigen reverse transcription polymerase chain reaction (RT-PCR)
DISCUSSION
COVID-19, caused by severe acute respiratory syndrome coronavirus 2 (SARS-Cov-2), was first identified in patients in late 2019 in Wuhan, Hubei province, China. It rapidly spread to other nations and was declared a global pandemic on March 11, 2020.1 As of April 27, there were 3 million cases with just over 200,000 deaths, affecting 185 countries.2
The virus is spread in humans primarily through respiratory droplets entering the nose and mouth. Direct and indirect contact and aerosol particles are also means for viral transmission. Once inside the respiratory tract, SARS-Cov-2 may gain access to nasopharyngeal, tracheobronchial, and/or alveolar epithelial cells by binding to angiotensin converting enzyme 2 (ACE2) receptors, leading to cytokine-mediated immune response and inflammation.3
The COVID-19 incubation period is up to two weeks. Common presenting symptoms include fever, cough, shortness of breath, myalgia, and fatigue. Hypertension, cardiovascular disease, diabetes, chronic pulmonary disorders, and chronic liver disease are comorbidities that may exacerbate the extent of disease. Clinical presentation, radiologic findings, and disease course are further affected by severity of the cytokine-mediated inflammation (eg, cytokine storm syndrome). Circulating cytokines can impact multiple other organ systems, causing secondary diseases and complications.4
ACE2 receptors are also found in the brain, heart, arterial and venous endothelial cells, kidneys, liver (hepatocytes and cholangiocytes), gastrointestinal tract, and gallbladder.3,5-7 This also offers the potential for SARS-Cov-2 to impact multiple organ systems, leading to atypical COVID-19 clinical presentations and the need for a wider range of radiologic imaging, with variable radiologic findings. In our experience and as reported in the literature, less common symptoms and presentations include headaches (7-23%), syncope, seizures, altered mental status, chest pain, acute cardiovascular events, abdominal pain, vomiting, and diarrhea (2-14%).4
With regard to liver involvement, up to approximately 50% of COVID-19 patients may present with liver dysfunction, evidenced by elevated ALT and AST.5 Most dysfunction is mild, with prevalence varying with severity of COVID-19. Liver dysfunction is likely secondary to the virus binding hepatic ACE2 receptors causing an immune cytokine storm, leading to hepatocellular inflammation and damage (eg, hepatitis).
Ji et al demonstrated that COVID-19 patients with high body mass index and nonalcoholic fatty liver disease (NAFLD) have a greater risk for both progression in liver damage and COVID-19.5 Progression in liver damage related to NAFLD (and potentially other chronic liver disorders) will complicate COVID-19 presentation, clinical course, and the role of imaging, with the potential for hepatic failure, hepatic encephalopathy, and gastrointestinal bleeding.
Definitive COVID-19 diagnosis is made with viral antigen (RT-PRC) and antibody testing. Chest radiography and CT are also key diagnostic tools, with higher sensitivity and specificity demonstrated by CT. Common chest radiologic findings include bilateral, multilobar, multifocal nodular and coalescent GGO in a peripheral distribution.8 Diffuse GOOs, consolidation, and reticular densities may also be found, simulating other infectious and inflammatory diseases.8
The spectrum of radiologic findings for COVID-19 pneumonia will vary with the duration and severity of disease and presence of comorbid cardiopulmonary disease. Chest imaging may be performed in patients with suspected COVID-19 and respiratory symptoms. Recognition of typical pulmonary findings would lead to a presumed diagnosis of active COVID-19. For other patients, neck, chest, and/or abdominal CT may be performed to evaluate symptoms not thought to be related to COVID-19. In these instances, evaluating the complete or partially imaged lungs may reveal unsuspected COVID-19 pneumonia.
In our case, the patient presented only with abdominal symptoms. Liver transaminases were slightly elevated; all other laboratory parameters were normal. Despite the patient’s recent exposure, active COVID-19 was not clinically suspected given the absence of respiratory symptoms. Obtaining an abdominal-pelvic CT was an appropriate diagnostic approach.
The fluid-filled bowel loops identified on CT were consistent with the clinical diarrhea; the intermittent small-bowel mucosa hyperemia and thickening were consistent with active inflammation. Once COVID-19 was suspected based upon the typical lung abnormalities, bowel observations could be attributed to the presence of COVID-19 in the gastrointestinal tract. ACE2 receptors are found throughout the gastrointestinal tract, with greatest functional role in the small bowel and colon.6 SARS-Cov-2 binding to bowel epithelial cells leads to mucosal inflammation (eg, enteritis). Viral alterations in intestinal microbial flora and cellular regulation of fluid and electrolytes can lead to diarrhea.
The fatty liver could have been interpreted as an incidental, isolated finding. However, once COVID-19 pneumonia was identified at the lung bases, hepatic steatosis became a significant finding. The elevated ALT and AST indicated mild liver dysfunction. The presence of hepatic steatosis indicated our patient was at risk for progression in liver dysfunction and COVID-19.
The gallbladder-wall hyperemia without gallstones, sludge, or pericholecystic fluid also became a significant finding when interpreted in conjunction with lung observations. To date, gallbladder-wall hyperemia in the setting of COVID-19 has not been reported. We propose that SARS-Cov-2 binding to gallbladder epithelial cells, leading to mucosal inflammation, can explain the radiologic observations.
CONCLUSION
In symptomatic patients, COVID-19 primarily infects and impacts the respiratory system. This case highlights that COVID-19 should be approached as a multisystemic disease secondary to its affinity for ACE2 receptors across multiple organ systems, cytokine-mediated inflammation, and patient co-morbid disease(s). By adopting this approach, radiologists can play a significant role in diagnosing COVID-19 secondary diseases and complications.
REFERENCES
- World Health Organization: Director-General’s opening remarks at the media briefing on COVID-19. March11, 2020. https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020.
- Johns Hopkins University: COVID-19 Dashboard by the Center for Systems Science and Engineering. https://coronavirus.jhu.edu/map.html. Accessed April 27, 2020.
- Lia G, Hea X, Zhang L, et al. Assessing ACE2 expression patterns in lung tissues in the pathogenesis of COVID-19. J Autoimun. 2020 Apr 13; 10.1016/j.jaut.2020.102463 [Epub ahead of print].
- Rodriguez-Morales AJ, Cardona-Ospinaa JA, Gutierrez-Ocampoa E, et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med Infect Dis. 2020 Mar 13; 10.1016/j.tmaid.2020.101623 [Epub ahead of print].
- Ji D, Qin E, Xu J, et al. Non-alcoholic fatty liver diseases in patients with COVID-19: A retrospective study. J Hepatol. 2020 Apr 8; 10.1016/j.jhep.2020.03.044 [Epub ahead of print].
- Garg M, Angus PW, Burrell LM, et al. Review article: the pathophysiological roles of the renin–angiotensin system in the gastrointestinal tract. Aliment Pharmacol Ther. 2012;35(4):414-428J.
- Zong, H., Yin, B., Zhou, H. et al. Loss of angiotensin-converting enzyme 2 promotes growth of gallbladder cancer. Tumor Biol. 2015;36:5171–5177.
- Simpson S, Kay FU, Abbara S, et al. Radiological Society of North America Expert Consensus Statement on Reporting Chest CT Findings Related to COVID-19. Endorsed by the Society of Thoracic Radiology, the American College of Radiology, and RSNA. 2020; Mar 25; https://doi.org/10.1148/ryct.2020200152.
Citation
JC H, R S, RL H, A K. Abdominal Presentation of COVID-19 . Appl Radiol. 2020; (3):24-26.
May 4, 2020