Accelerated hypertension with features of reversible posterior encephalopathy syndrome
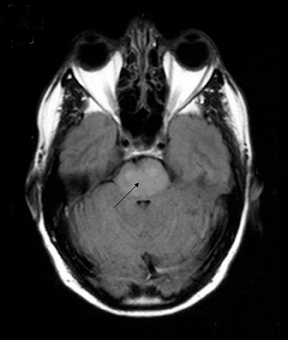
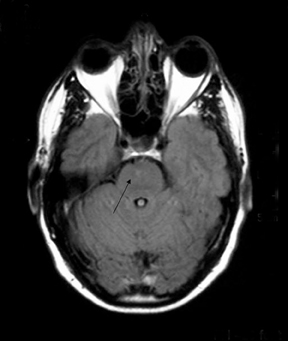

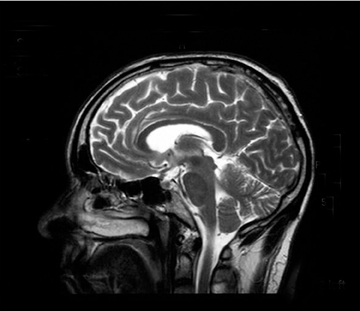
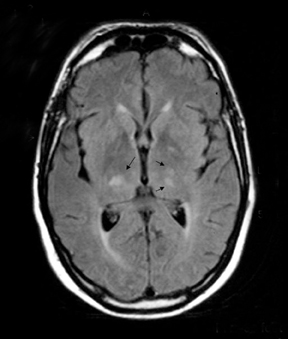



Axial and sagittal T2-weighted and fluid attenuation inversion recovery (FLAIR) sequences were performed. These were followed by an axial T1-weighted gadolinium-enhanced sequence. These showed extensive high-signal changes in the white matter with the pons and periventricular regions most affected. Further high-signal foci were seen within the basal ganglia bilaterally. There was mild swelling within the pons, but the lesions exhibited no significant mass effect, and no enhancement post contrast.
The patient’s blood pressure was slowly lowered with intravenous labetolol and diuretics and his vision gradually improved. He was discharged after 5 days with a blood pressure of 160/100 mmHg.
A repeat MRI examination was performed 5 weeks later. This showed a dramatic improvement, with complete resolution of all the brain stem and basal ganglia abnormalities, thus confirming the diagnosis of RPES. There was minor residual high-signal change seen in the left parietal and occipital lobes. His home blood pressure records since discharge were noted to be 150-160/90-100 mmHg, and he remained asymptomatic.
Discussion
Hinchey, et al.,1 first described reversible posterior leukoencephalopathy syndrome just 10 years ago. It has a diverse etiology, the most common cause being abrupt elevation of blood pressure from accelerated hypertension, acute renal failure or eclampsia. It has also been described inassociation with immunosuppressive therapy (particularly cyclosporine and tacrolimus) and has been linked with a variety of other conditions,including hemolytic uremic syndrome, systemic vasculitides, and intravenous immunoglobulin administration.2
The clinical manifestations result from increased cerebral perfusion with loss of blood-brain barrier integrity, resulting in exudation of fluid into the surrounding brain (vasogenic edema).3 Changes predominate in the white matter, but gray matter can be affected as well (hence some authors suggest that the syndrome should be renamed as reversible posterior encephalopathy syndrome). The posterior cerebrum tends to be more affected than other areas.
CT scanning typically shows symmetrical areas of low attenuation in the white matter of the posterior cerebral cortex. With MRI, lesions are high signal on FLAIR and T2-weighted sequences. Involvement of the overlying cortical gray matter may also be demonstrated. Diffusion weighted imaging shows the lesions as a low signal with an increased diffusion coefficient indicative of vasogenic edema.1,3,4 MR spectroscopy has also been evaluated in the diagnosis and follow-up of this condition.3 Raised choline and creatine levels with low N-acetylaspartate have been observed in the acute phase with levels of these neurochemicals returning to normal post treatment.
The closest differential diagnosis for this condition is bilateral infarction of the posterior cerebral arteries. However, in these cases, one would expect to find more focal neurological signs.1
The overall prognosis of RPES is good. Treatment involves lowering of blood pressure, control of seizures, and removal of any offending agent. However, if not recognized and treated promptly, conversion to irreversible cytotoxic edema can occur.5 With appropriate treatment,the symptoms and signs generally resolve quickly, but the imaging findings can take several months to fully disappear (anywhere from 8 days to 17 months in one study). A few patients have been reported to have residual hemosiderin deposits in the affected brain areas despite complete clinical recovery.
Conclusion
Reversible posterior encephalopathy is a rapidly evolving neurological syndrome with characteristic radiological features. Clinicalpresentations include headache, seizures, speech and visual disturbances, and alteration of mental status. Clinicians must recognize cortical visual loss as a presenting feature of RPES and that blood pressure control results in good clinical recovery.
- Hinchey J, Chaves C, Appignani B, et al. A reversible posterior leucoencephalopathy syndrome. N Engl J Med.1996;334:494-500.
- Casey SO, Sampsio RC, Michel E et al. Reversible posterior leucoencephalopathy syndrome. Utility of fluid attenuated inversion recovery magnetic resonance in the detection of cortical and subcortical lesions. AJNR Am J Neuroradiol. 2000;21:1199-1208.
- Eichler FS, Wang P, Wityk RJ, Beauchamp NJ Jr, Barker PB. Diffuse metabolic abnormalities in reversible posterior leukoencephalopathy syndrome. AJNR Am J Neuroradiol. 2002;23:833-837.
- Lamy C, Oppenheim C, Meder JF, Mas JL. Neuro-imaging in posterior reversible encephalopathy syndrome. J Neuroimaging. 2004;14:89-96.
- Schwartz RB. A reversible posterior leukoencephalopathy syndrome [letter]. N Engl J Med. 1996;334:1743.
