Adrenal incidentalomas in 2014
Images
The incidental adrenal lesion is the product of the expanding clinical indications of cross-sectional imaging as well as the increasing spatial resolution of modern imaging techniques. Adrenal “incidentalomas” are defined as adrenal masses larger than 1 cm in patients for whom imaging studies are being performed for an unrelated indication. The overall prevalence of incidental adrenal masses at CT is 4%-5%.1,2 The frequency of adrenal incidentalomas has been found to correlate with increasing age, with 0.2% detected in CT studies performed in subjects aged 20-29 years versus 6.9% in those older than 70 years.1, 2 The overwhelming majority of these lesions comprise benign nonfunctioning adenomas in the asymptomatic patient with no malignancy history.3,4 However, this does not hold true for oncologic patients, as the adrenal glands are a common site for metastatic disease. Of additional concern are lesions that are hormonally active and the rare primary adrenocortical carcinomas. For this reason, differentiating the lesions that warrant treatment from those benign, inconsequential masses that should be left alone is essential. This imaging goal should be achieved with minimal workup, given the high prevalence of benign adrenal masses. Consideration of both patient history and imaging characteristics is key to the appropriate management of adrenal incidentalomas.
Risk of malignancy
Several factors determine the pretest probability of malignancy for an adrenal incidentaloma. These include imaging features, lesion size, lesion stability and patient history of malignancy.
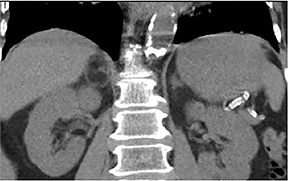
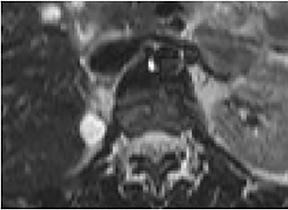
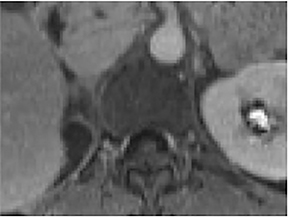
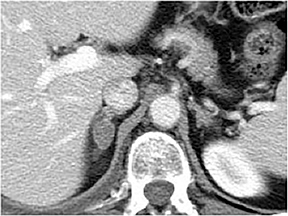
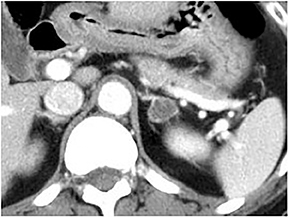
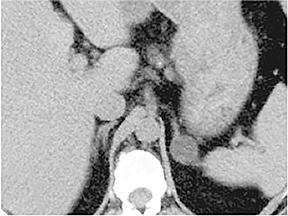
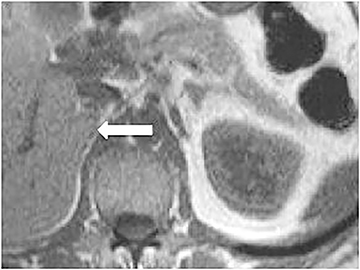
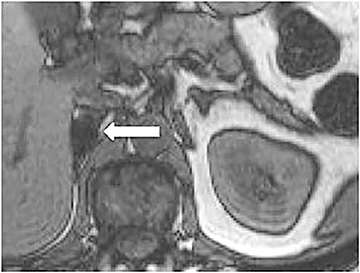
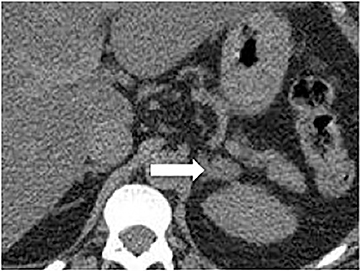
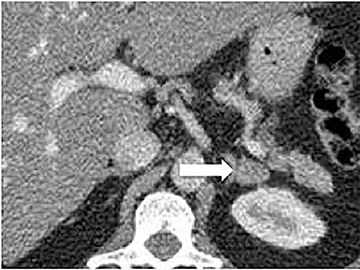
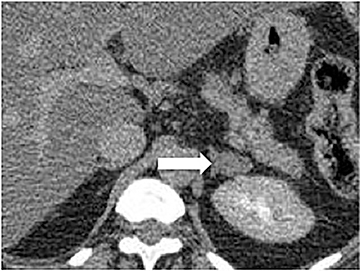
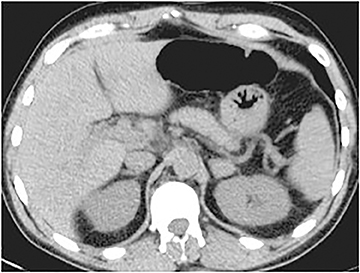
Certain imaging features in adrenal masses are diagnostic of benignity and require no further workup. For example, a myelolipoma is easily recognized by the presence of macroscopic fat (Figure 1). A nonenhancing adrenal mass of fluid density or signal and a thin wall is diagnostic of a simple cyst (Figure 2) requiring no further workup. However, incidental adrenal masses often have a nonspecific appearance at detection, especially at contrast-enhanced CT. Imaging characteristics such as smooth margins and homogeneous density are typically observed in benign lesions (Figure 3) and although reassuring, are considered indeterminate in isolation, as these “benign” morphologic features can also be present in small metastatic masses (Figure 4).5 Imaging features suspicious for malignancy include invasion of adjacent structures, a thick enhancing rim, necrosis, and irregular margins.
Comparison to prior imaging is essential in the assessment of an adrenal incidentaloma. An adrenal mass that demonstrates stability for at least one year is highly unlikely to represent metastatic or primary malignancy, making further imaging workup unnecessary.6 Rarely, benign adenomas or myelolipomas can enlarge,7 and a recent study has shown that adrenal cysts can also increase in size.8 However, as a general rule in clinical practice, a mass that increases in size warrants further workup to exclude malignancy.
The correlation between size and relative risk of malignancy is well established and, although there is no specific size below which a mass can definitely be diagnosed as benign, the risk of malignancy has been shown to increase with mass size. In patients with an adrenal mass greater than 4 cm and no history of extra-adrenal malignancy, resection is typically pursued unless the mass can definitively be classified as benign.6,9
A history of malignancy is arguably one of the most heavily weighted factors in determining the malignant potential of an adrenal lesion. It is exceedingly rare for an extra-adrenal malignancy to present as an isolated adrenal metastasis.4,10 However, in oncologic patients, the rate of metastasis depends on the type of primary cancer. In these patients, differentiating a benign adrenal mass from metastasis is critical, especially if the adrenal gland is the only potential site of metastasis.
Imaging studies
Several imaging tools are invaluable in evaluating adrenal incidentalomas, and imaging diagnosis relies on the presence or absence of lipid content, contrast washout pattern, and degree of metabolic activity.
Imaging of intracellular lipid: CT and MRI
The rationale for determining the lipid content of an adrenal lesion is based on the finding that adrenal adenomas contain variable amounts of intracellular lipid, whereas metastatic lesions generally do not.11 An unenhanced CT (NC-CT) attenuation value of up to 10 HU has been shown to diagnose a lipid-rich adenoma with 71% sensitivity and 98% specificity, and is therefore used as the threshold below which adrenal lesions are considered to be definitively benign (Figure 5).12 It is important to note that up to 30% of adenomas are lipid poor and have attenuation values greater than 10 HU, deemed indeterminate at NC-CT.
At MR, intracellular lipid can be detected using chemical-shift imaging (CSI), which takes advantage of the differing resonant frequencies of protons in lipid and water molecules. As the frequency of lipid protons is slower, voxels containing both fat and water will demonstrate net signal cancellation when the protons are out of phase compared to when they are in phase. Therefore, adrenal adenomas containing intracellular lipid generally will demonstrate signal loss on out-of-phase imaging (Figure 6), unlike metastatic lesions which will not show signal loss.13, 14 Quantitative methods have been developed to measure the degree of signal intensity loss. For example, a lesion-to-spleen chemical shift ratio can be calculated with upper diagnostic threshold set at 0.71, or an index of adrenal signal intensity loss can be calculated with lower diagnostic threshold of 16.5%.14,15 However, in clinical practice, visual analysis using the spleen as a reference is more commonly used as it is as effective as quantitative methods and is simpler to apply.16 The main advantage of CSI-MR over NC-CT is in young patients in whom radiation exposure is a concern. In a few small series comparing NC-CT to CSI-MR, CSI-MR allowed additional diagnosis of some, but not all, adenomas that were lipid-poor on CT, due to the increased sensitivity of MR to low levels of lipid compared to CT.17,18
Perfusion imaging: CT washout
After intravenous contrast administration, adrenal adenomas lose contrast more rapidly compared to non-adenomas. Based on this principle, Korobkin et al19 developed a technique in which washout percentage is calculated using delayed phase imaging obtained 15 minutes after injection. When both unenhanced and contrast enhanced images are available, the absolute percentage of enhancement washout (APW) is calculated as:
(E–D)/(E–U) x 100%
where E is the attenuation value of the lesion on enhanced CT, D is the attenuation value of the lesion on 15-minuted delayed CT, and U is the attenuation value of the lesion on unenhanced CT. A value of 60% or greater is considered diagnostic of an adenoma (Figure 7).
If initial unenhanced CT images are not available, the relative percentage washout can be calculated as:
(E–D)/E x 100%.
A value of 40% or greater is considered diagnostic of an adenoma. Contrast washout is independent of the amount of lipid content and allows accurate diagnosis of both lipid-rich and lipid poor-adenoma. In one study, the combined adrenal protocol was 96% accurate in distinguishing adenoma from non-adenoma.20
Functional imaging: PET and PET/CT
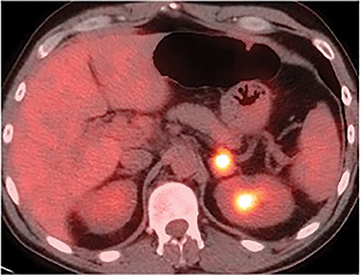
PET and PET/CT are usually reserved for oncologic patients. Adrenal adenomas can be differentiated from malignant lesions on PET by their differences in FDG avidity. Unlike benign lesions, malignant adrenal masses are usually hypermetabolic and will demonstrate increased FDG uptake (Figure 8). In practice, qualitative assessment can be performed using liver activity as a standard of reference. PET and PET/CT has been demonstrated to detect malignant lesions with sensitivity ranging from 93% to 100% and specificity ranging from 80% to 100%.21-23 Pitfalls include lesions less than 1 cm in size, as small malignant lesions tend to demonstrate FGD activity levels lower than that of liver.21 Additionally, false positive findings can be produced by inflammatory or infectious processes and, less commonly, by some metabolically active adrenal adenomas.7
Algorithm for incidental adrenal mass
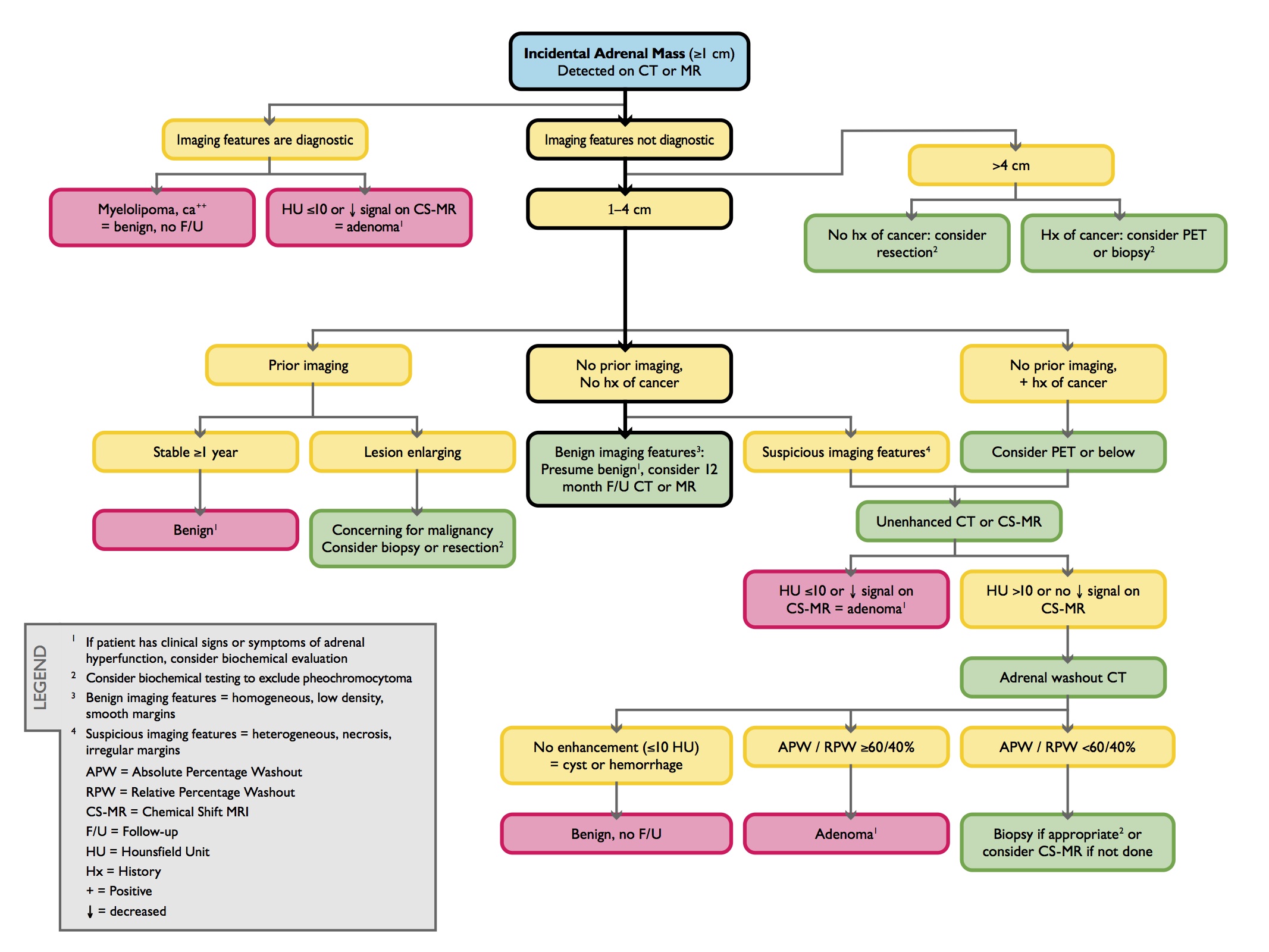
An algorithm for imaging of the incidental adrenal mass was published in the 2010 white paper of the American College of Radiology Committee on Incidental Findings and is summarized as follows (Figure 9):6
An adrenal lesion with imaging features diagnostic of a myelolipoma, lipid-rich adenoma or cyst is benign and requires no further work up, regardless of size.
For lesions less than 4 cm in size without diagnostic features, comparison should be made to prior studies. If the lesion has been stable for at least one year, it is likely benign and no follow up is necessary. However, lesions that demonstrate growth are suspicious for malignancy and warrant biopsy or resection.
If no prior imaging is available for comparison of an indeterminate lesion less than 4 cm in a patient without a history of cancer, follow up NC-CT or CSI-MR in 12 months can be recommended if benign characteristics are identified (eg, homogeneous density, low density, smooth margins). In the presence of suspicious characteristics (eg, heterogeneous density, necrosis, irregular margins), or if the patient has a prior history of cancer, NC-CT or CSI-MR can be obtained, and if needed, adrenal CT with washout analysis. If these studies do not confirm a benign diagnosis, biopsy should be considered to exclude malignancy. In patients with a history of an extra-adrenal malignancy, PET may also be considered prior to pursuing biopsy.
For lesions greater than 4 cm in patients without a history of cancer, resection is typically pursued because of the risk of adrenocortical carcinoma. In patients with a history of extra-adrenal malignancy, PET or biopsy should be performed to exclude metastatic disease.
Endocrine function
It is important to note that imaging characteristics do not correlate with the functional status of an adrenal mass. It is currently the consensus among most endocrinologists that screening should be performed in all patients with an incidental adrenal mass to exclude an occult, asymptomatic hyperfunctioning adrenal mass.24-26 However, because this approach is costly, others opt to screen only those patients with clinical signs and symptoms of adrenal hyperfunction.
Conclusion
Adrenal masses are common at contemporary cross-sectional imaging and most are benign. Adrenal imaging can accurately distinguish benign from malignant lesions. Judicious use of imaging is important for the appropriate management of these common lesions.
References
- Kloos RT, Gross MD, Francis IR, et al. Incidentally discovered adrenal masses. Endocr Rev. 1995; 16: 460-484.
- Young WF, Jr. Management approaches to adrenal incidentalomas. A view from Rochester, Minnesota. Endocrinol Metab Clin North Am. 2000; 29:159-185.
- Mansmann G, Lau J, Balk E, et al. The clinically inapparent adrenal mass: Update in diagnosis and management. Endocr Rev. 2004;25:309-340.
- Song JH, Chaudhry FS and Mayo-Smith WW. The incidental adrenal mass on CT: Prevalence of adrenal disease in 1,049 consecutive adrenal masses in patients with no known malignancy. AJR Am J Roentgenol. 2008;190:1163-1168.
- Song JH, Grand DJ, Beland MD, et al. Morphologic features of 211 adrenal masses at initial contrast-enhanced CT: Can we differentiate benign from malignant lesions using imaging features alone? AJR Am J Roentgenol. 2013;201:1248-1253.
- Berland LL, Silverman SG, Gore RM, et al. Managing incidental findings on abdominal CT: White paper of the ACR incidental findings committee. J Am Coll Radiol. 20107: 754-773.
- Boland GW, Blake MA, Hahn PF, et al. Incidental adrenal lesions: Principles, techniques, and algorithms for imaging characterization. Radiology. 2008; 249:756-775.
- Ricci Z, Chernyak V, Hsu K, et al. Adrenal cysts: Natural history by long-term imaging follow-up. AJR Am J Roentgenol. 2013;201:1009-1016.
- American College of Radiology. ACR Appropriateness criteria: Incidentally discovered adrenal mass. http://www.acr.org/Quality-Safety/Appropriateness-Criteria/Diagnostic/Urologic-Imaging. Published 1996. Updated 2012. Accessed March 15, 2014.
- Lee JE, Evans DB, Hickey RC, et al. Unknown primary cancer presenting as an adrenal mass: Frequency and implications for diagnostic evaluation of adrenal incidentalomas. Surgery. 1998;124:1115-1122.
- Korobkin M, Giordano TJ, Brodeur FJ, et al. Adrenal adenomas: Relationship between histologic lipid and CT and MR findings. Radiology. 1996;200: 743-747.
- Boland GW, Lee MJ, Gazelle GS, et al. Characterization of adrenal masses using unenhanced CT: An analysis of the CT literature. AJR Am J Roentgenol. 1998; 171:201-204.
- Mitchell DG, Crovello M, Matteucci T, et al. Benign adrenocortical masses: Diagnosis with chemical shift MR imaging. Radiology. 1992; 185:345-351.
- Outwater EK, Siegelman ES, Huang AB, et al. Adrenal masses: Correlation between CT attenuation value and chemical shift ratio at MR imaging with in-phase and opposed-phase sequences. Radiology.1996;200:749-752.
- Fujiyoshi F, Nakajo M, Fukukura Y, et al. Characterization of adrenal tumors by chemical shift fast low-angle shot MR imaging: Comparison of four methods of quantitative evaluation. AJR Am J Roentgenol. 2003;180:1649-1657.
- Mayo-Smith WW, Lee MJ, McNicholas MM, et al. Characterization of adrenal masses (< 5 cm) by use of chemical shift MR imaging: observer performance versus quantitative measures. AJR Am J Roentgenol. 1995;165:91-95.
- Israel GM, Korobkin M, Wang C, et al. Comparison of unenhanced CT and chemical shift MRI in evaluating lipid-rich adrenal adenomas. AJR Am J Roentgenol. 2004;183:215-219.
- Haider MA, Ghai S, Jhaveri K, et al. Chemical shift MR imaging of hyperattenuating (>10 HU) adrenal masses: Does it still have a role? Radiology. 2004;231:711-716.
- Korobkin M, Brodeur FJ, Francis IR, et al. CT time-attenuation washout curves of adrenal adenomas and nonadenomas. AJR Am J Roentgenol. 1998;170:747-752.
- Caoili EM, Korobkin M, Francis IR, et al. Adrenal masses: Characterization with combined unenhanced and delayed enhanced CT. Radiology. 2002;222:629-633.
- Chong S, Lee KS, Kim HY, et al. Integrated PET-CT for the characterization of adrenal gland lesions in cancer patients: diagnostic efficacy and interpretation pitfalls. Radiographics.2006;26:1811-1824; discussion 1824-1816.
- Yun M, Kim W, Alnafisi N, et al. 18F-FDG PET in characterizing adrenal lesions detected on CT or MRI. J Nucl Med. 2001;42:1795-1799.
- Blake MA, Slattery JM, Kalra MK, et al. Adrenal lesions: Characterization with fused PET/CT image in patients with proved or suspected malignancy—initial experience. Radiology. 2006;238: 970-977.
- NIH state-of-the-science statement on management of the clinically inapparent adrenal mass (“incidentaloma”). NIH Consens State Sci Statements. 2002;19:1-25.
- Grumbach MM, Biller BM, Braunstein GD, et al. Management of the clinically inapparent adrenal mass (“incidentaloma”). Ann Intern Med. 2003;138:424-429.
- Zeiger MA, Thompson GB, Duh QY, et al. The American Association of Clinical Endocrinologists and American Association of Endocrine Surgeons medical guidelines for the management of adrenal incidentalomas. Endocr Pract. 2009;15 Suppl 1: 1-20.