Angiographic evaluation and treatment for head and neck vascular injury
Images



Dr. Bykowski is a Fellow in the Division of Neuroradiology, and Dr. Wong is a Professor of Clinical Radiology, Interventional Neuroradiology, Professor of Pain and Palliative Medicine, University of California–San Diego Medical Center, San Diego, CA.
A variety of head and neck vascular emergencies, such as nosebleeds or neoplastic hemorrhages, can occur spontaneously or result from blunt or penetrating trauma. As most traumatic venous bleeding can be resolved with direct pressure, the main focus is on arterial injury. The role of catheter angiography in the acute trauma setting has shifted over the past 15 years, with the concomitant advances in computed tomography (CT) angiography for diagnosis, and development of microcatheters and embolic agents for therapy.
Regional trauma associations have proposed algorithms for which patients should be evaluated by CT angiography and/or catheter angiography for traumatic head and neck vascular injuries.1-3 These include high-risk mechanisms such as: high-energy collisions, neck hyperextension injury, intra-oral trauma, and near-hanging with anoxic brain injury. Additionally, CT or catheter angiography should be considered in patients with LeFort/midface fractures, cervical spine or basilar skull fractures, diffuse axonal injury with Glasgow Coma Scale (GCS) < 6, a new focal neurological deficit, neurological examination incongruous with head CT findings, or imaging evidence of a new cerebral infarct in the setting of trauma.
Clinically occult head and neck vascular injury is rare; however, aggressive CT screening in asymptomatic patients has become commonplace given the potentially devastating sequelae of a missed diagnosis,4-6 combined with ease of access to CT angiography. Ongoing discussion in the trauma community continues regarding patient selection criteria, given the concerns of cost effectiveness of broad screening in asymptomatic patients as well as minimizing unnecessary radiation exposure.
There remain cases in which conventional angiogram remains the ‘gold standard.’ These include CT angiograms limited by artifact from dental implants/amalgam, from metal or shrapnel, situations where appropriate bolus timing cannot be achieved, and hemodynamically unstable patients with a high probability of requiring endovascular intervention. Diagnostic catheter angiogram should always be considered in a patient with high suspicion for cervical vascular injury in the setting of a normal CT angiogram, as this is a dynamic disease process and contrast opacification of a vessel on cross-sectional imaging may not fully reflect flow dynamics and collateral pathways.
Large arterial lacerations, pseudoaneurysms, and arteriovenous fistulae
Damage to the arterial wall can result in life-threatening hemorrhage, and patients with large arterial lacerations due to penetrating trauma have significant mortality before reaching hospital care. Alternatively, hemorrhage may be contained by development of a pseudoaneurysm or diverted through a traumatic arterial-venous fistula. In dealing with a patient with a potential arterial laceration, it is crucial to maintain hemodynamic and ventilatory support throughout the search for and treatment of the active bleeding site.
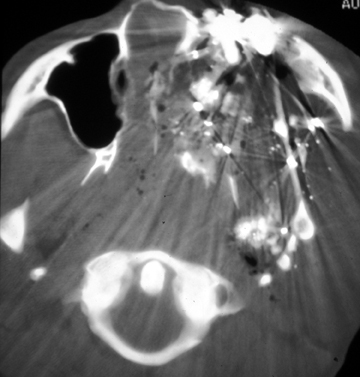
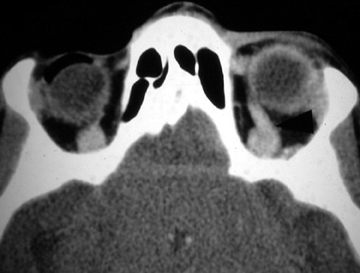
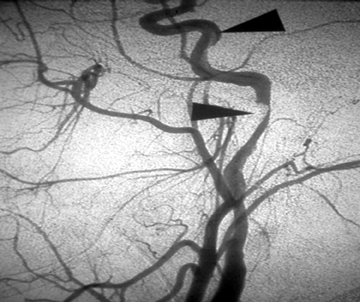
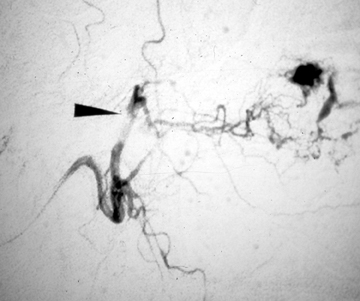
Exploration of anterior neck wounds is usually done surgically, given adequate exposure and direct visualization of the carotid arteries.7 In patients with active hemorrhage from a carotid or vertebral laceration, there is a high risk of stroke or even death despite aggressive treatment, including surgical ligation or endovascular embolization of the vessel. Given the difficult surgical approach to the carotid artery at the skull base and the vertebral arteries,8,9 the interventional neuroradiologist can provide great support to the trauma team with an endovascular approach to treatment at these sites.10,11,12 Additionally, in patients with extensive facial fractures or penetrating injuries, an endovascular approach to control bleeding is preferred (Figure 1).
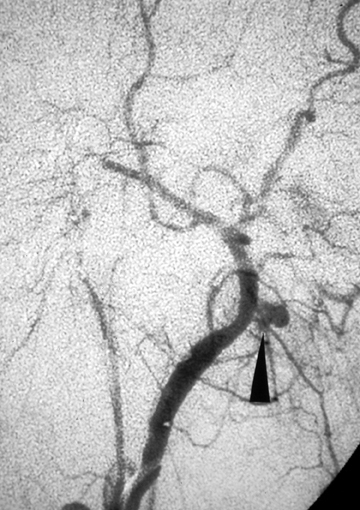
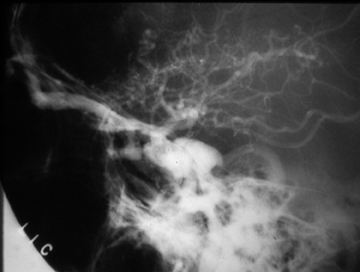
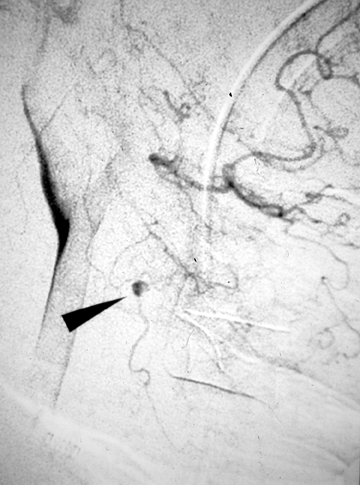
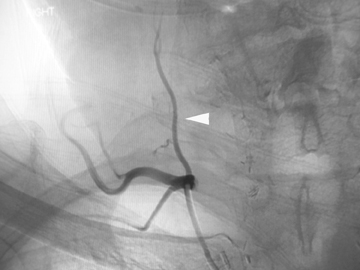
Pseudoaneurysms result from arterial bleeding into the wall of an injured vessel. This can manifest as a focally expanded dissection with containment by the adventitia, or containment of leakage outside of the vessel wall by a layer of clot. While some extremity pseudoaneurysms have been reported to resolve spontaneously,13 asymptomatic pseudoaneurysms of the carotid arteries are generally treated to preclude thromboembolic stroke and reduce the risk of re-bleeding. Endovascular embolization with coils or balloon occlusion is often favored over direct surgical exploration;14,15 however, it should be done with care as re-bleeding is common given the fragility of structures containing the site of injury (Figure 2). In some situations, a stent may be sufficient to divert flow, allowing the pseudoaneurysm to thrombose without coil deployment.16-18 Some controversy persists regarding the risks and benefits of stent placement, with long-term stent occlusion rates reported in up to 45% of patients in early series.19 Discussion continues about the ideal timing of treatment, perceived benefits of different stent features, and optimal concomitant antiplatelet therapy in these patients.20
Traumatic arteriovenous fistulae can occur in the setting of arterial transection, with resulting communication between the injured artery and adjacent vein. Most commonly, these occur in the facial arterial or at the cavernous carotid artery, due to the prevalence of facial and skull base injury, respectively.
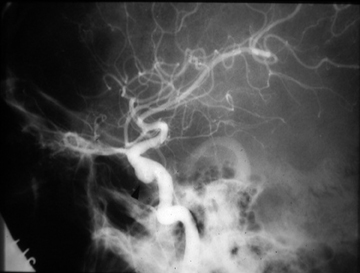
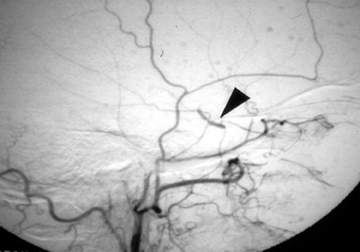
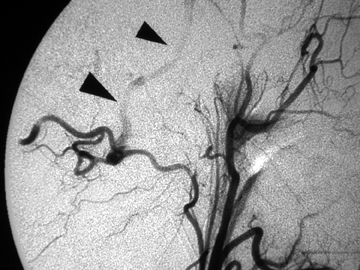
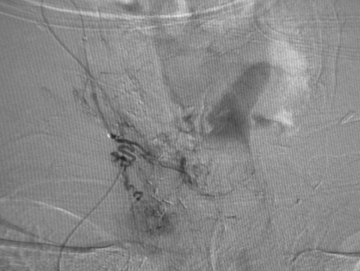
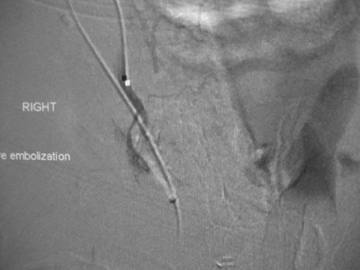
Different methods of embolization have been described, depending on the flow rate, site, and available technology.21-23 Small fistulous communications may be embolized with polyvinyl alcohol particles (Figure 3). In larger fistulae, deployment of micro coils, detachable balloons or liquid embolic agents through the fistula may be necessary to obtain cessation of shunting (Figure 4).
In situations where vessel sacrifice is considered, occlusion by balloons or coils should only be done after a thorough test balloon occlusion to ensure there will not be undesired, irreversible neurological sequelae. Unilateral vertebral artery occlusion is considered more forgiving as long as the contralateral, uninjured vertebral artery has adequate caliber and the embolization material can be deployed proximal to the posterior inferior cerebellar artery (PICA), preserving collateral supply on the side of injury.24,25 A typical balloon test occlusion is performed by anticoagulating the patient with heparin and then advancing an occlusive balloon across or distal to the site of injury, to cause cessation of blood flow. Neurological testing for the carotid artery would include evaluation of pronator drift, motor, sensory, and memory function. Vertebral artery neurological testing during balloon occlusion is less reliable, however, and emphasis should be placed on coordination, motor, and sensory function. The balloon test occlusion is typically maintained for 30 minutes or until the patient fails the procedure.
Extracranial arterial dissections and occlusions
Arterial dissections in the head and neck usually are associated with deceleration and shear injuries. These include injuries to the proximal cervical vertebral artery, and the distal internal carotid and vertebral arteries below the skull base. Vertebral artery dissections can also occur at the sites of transverse foramen fractures, and these areas should be carefully evaluated in the setting of cervical spine trauma.26 Occlusions can result from sluggish flow in the dissected vessel, compounded by underlying atherosclerotic disease.
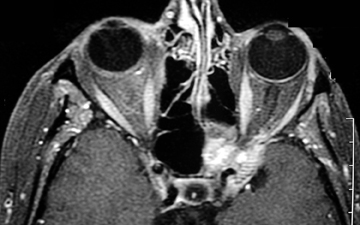
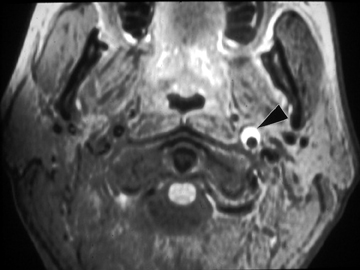
In the acute setting, CT angiography is commonly used to evaluate for vessel irregularity and filling defects. MR imaging, particularly T1 fat-saturated sequences, is sensitive for the detection of methemoglobin in a false lumen of a dissection27 (Figure 5). However, within the first 3 days after the traumatic event, the blood products often have only intermediate signal changes. Diagnostic catheter angiography may be needed in patients with artifact from bullet fragments or dental amalgam or difficult evaluation at the skull base.
Treatment of carotid and vertebral arterial dissections remains somewhat controversial.2 The most conservative approach includes medical management, with ongoing debate as to whether anticoagulation with heparin and/or antiplatelet therapy is more effective.19,26,28,30 There remains concern about the use of these agents in the setting of acute multitrauma,30 although successful treatment with antiplatelet agents has also been described in the setting of pre-existing intracranial hemorrhage.31 Medical management has resulted in 50% to 70% successful arterial recanalization rates.32,33 However, these patients remain at risk for thromboembolic events in the days to weeks following the traumatic event34 or delayed formation of dissecting aneurysms. Much of the healing of dissections occurs 3-6 months after the inciting event.35,36
Stents have been used to treat patients who have contraindications to anticoagulation or antiplatelet therapy,37 although adjunctive antiplatelet therapy is often used to ensure long-term stent patency.20 Endovascular treatment with stents has also been described in patients who fail medical management either with ongoing or new neurological symptoms, or enlargement of a dissecting aneurysm on follow-up evaluation.38
If an ischemic stroke has occurred, coordination with the stroke neurology team is essential. Brain imaging, including diffusion- and perfusion-weighted imaging, should be a consideration, understanding that there may be time constraints if revascularization is indicated. If imaging suggests an embolic mechanism for the stroke, techniques similar to stroke thrombolysis or thromboembolectomy may be used. If a large vessel occlusion is present, angioplasty or stenting may not be wise as this may cause a reperfusion hemorrhage in the brain.
Branch vessel arterial lacerations
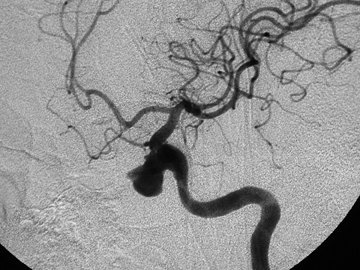
Trauma to the face, neck, and scalp can result in damage to branches of the external carotid arteries that cannot be controlled by direct pressure alone. Understanding the trauma mechanism and having cross-sectional imaging of the head and neck are helpful in the acute setting to tailor the angiogram most expeditiously to areas of interest. One should always consider the rich collateral supply to the face and neck, and the thyrocervical trunk, vertebral artery, and internal carotid artery branches should also be scrutinized (Figure 6).
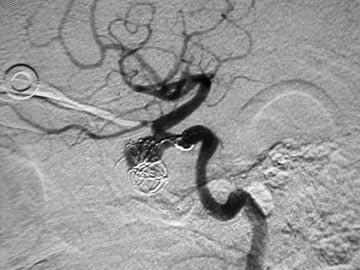
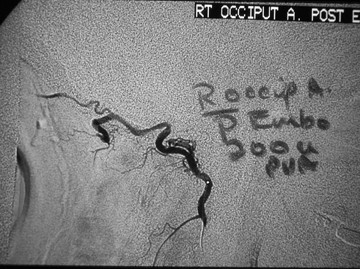
The goal is to decrease the pressure head within the injured vessel with resulting cessation of bleeding. Generally, it is important to place the tip of the catheter as close to the bleeding site as practical to avoid occlusion of normal branches. Additionally, prior to any particle embolization, one should be well aware of potential dangerous anastamotic collaterals.39 These include: distal external carotid artery ethmoidal perforators to the ophthalmic artery, superficial temporal artery to the middle cerebral artery, middle meningeal artery to the ophthalmic artery, and occipital artery to the vertebral artery (Figure 7). Additional embolization hazards, such as scalp necrosis, should be kept in mind when targeting sites in the superficial temporal and occipital arteries.
In areas where potential neurological deficit or collateral flow would be detrimental, provocative testing with 2 ml 1% lidocaine (20 mg) with concomitant neurological testing can be helpful. For example, provocative testing can reveal neurological deficits of cranial nerves V, VII, and X associated with the ascending pharyngeal artery before embolization, allowing for appropriate change in the therapy plan.
Temporary occlusive agents, such as gel foam and particles, are the preferred embolization material in most situations, as coils, glue, and balloons may preclude access in the setting of re-bleeding. Gel foam can be made into a slurry with contrast, allowing safe, targeted delivery through a 3 or 4 French catheter. Particles, such as 200-700 micron polyvinyl alcohol, are usually mixed with Iohexol 240 contrast to create an evenly distributed isobaric solution. Polyvinyl alcohol particles are injected via a microcatheter fast enough to be visualized but not so fast as to create reflux into normal vessels. As the embolization progresses, the injection rate typically slows until stagnation and flow are angiographically evident. The use of smaller particles increases the risk of nontarget embolization by particle migration via small collateral vessels.
Epistaxis
Nosebleeds are common and can be spontaneous, traumatic, or secondary to underlying telangiectasia, arteriovenous malformations or neoplasms, such as juvenile nasal angiofibromas. The first step is to identify the site of bleeding.
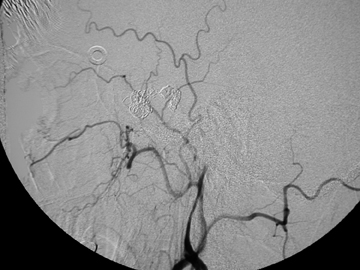
Most commonly, the bleeding site is anterior, supplied from Kiesselbach plexus (sphenopalatine, descending palatine, superior labial branches from ECA and anterior and posterior ethmoidal arteries from the ophthalmic artery).40,41 Anterior nasal bleeding can often be stopped with direct pressure, packing, or cautery, given the ease of access. If the bleeding site is posteriorly located, endovascular embolization is preferred over arterial ligation, as it allows repeated access in the event of re-bleeding via collateral branches.42, 43 This is usually best accomplished via the internal maxillary arteries (Figure 8), with microcatheter placement distal to the origins of the middle meningeal and accessory meningeal arteries.
In all cases of nasal and facial embolization, it is essential to evaluate collateral supply via the ophthalmic and facial arteries to avoid undesirable non-target embolization.44 Collateral supply can occur via the artery of the foramen rotundum, the vidian and ascending pharyngeal arteries, as well as communications between the facial, sphenopalatine and ophthalmic arteries. Preferred treatment is with temporary occlusive agents, such as 200-500 micron polyvinyl alcohol particles. It is important to closely monitor the injection rate, to avoid reflux into other branch vessels. If subselective arterial positioning cannot be achieved or the vascular anatomy is altered by prior surgical intervention, gelfoam injection into the larger, feeding artery may sufficiently diminish the pressure and stop the bleeding. We typically avoid using coils to treat epistaxis, as these permanent devices preclude future access, if re-bleeding occurs. Having to access the bleed via collateral sources such as the ophthalmic artery makes the embolization procedure much more hazardous.
Neoplastic bleeds
Vascular head and neck neoplasms, such as thyroid cancer and paraganglioma, may bleed spontaneously and be difficult to control externally. Often, the only finding is hypervascular oozing. In such cases, partial embolization of the tumor may sufficiently shut down the vascular bed.
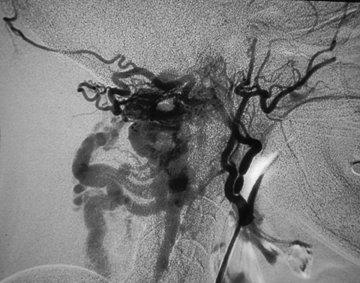
More commonly, head and neck cancers can erode into a blood vessel wall and cause spontaneous hemorrhage. The search for neoplastic bleeding source can be challenging (Figure 9), and surgical exploration can be difficult in patients with prior neck dissection or radiation therapy. In the setting of neoplastic bleeding, one may see hypervascular tumor blush or there may be actual active extravasation.45 In some cases, such as carotid blow-out, bleeding can be profuse and life-threatening. In this setting, emergent endovascular therapy with stents, balloon occlusion and liquid glue have been reported,46,47,48 with the understanding that these are often palliative measures.
Conclusion
Catheter angiography continues to serve a role in the diagnosis of head and neck vascular trauma, particularly in cases with high suspicion for vascular injury or where CT angiography is limited due to artifact from dental amalgam or gunshot debris. The neurointerventionalist continues to play an increasing role in the acute setting to identify and stop bleeding, with an increasing number of temporary and permanent agents within their armamentarium. Before embolization, it is crucial to assess collateral vascular supply, both to avoid nontarget embolization and undesired permanent sequelae when vessel sacrifice is required. Endovascular procedures can also be a useful adjunct in patients who have failed conservative management. The population of head and neck vascular trauma and bleeding is heterogeneous and techniques continue to advance to serve these unique cases.
References
- Biffl WL, Cothren CC, Moore EE, et al. Western Trauma Association critical decisions in trauma: screening for and treatment of blunt cerebrovascular injuries. J Trauma. 2009;67:1150-1153.
- Miller PR, Fabian TC, Croce MA, et al. Prospective screening for blunt cerebrovascular injuries: Analysis of diagnostic modalities and outcomes. Ann Surg. 2002;236:386–393.
- Bromberg WJ, Collier BC, Diebel LN, et al. Blunt cerebrovascular injury practice management guidelines: The Eastern Association for the Surgery of Trauma. J Trauma. 2010;68:471-477.
- Cogbill TH, Moore EE, Meissner M, et al. The spectrum of blunt injury to the carotid artery: A multicenter perspective. J Trauma. 1994;37: 473-479.
- Biffl WL, Moore EE, Ryu RK, et al. The unrecognized epidemic of blunt carotid arterial injuries: Early diagnosis improves neurologic outcome. Ann Surg. 1998;228:462-470.
- Berne JD, Norwood SH, McAuley CE, et al. The high morbidity of blunt cerebrovascular injury in an unscreened population: More evidence of the need for mandatory screening protocols. J Am Coll Surg. 2001;192:314-321.
- Feliciano DV. Management of penetrating injuries to the carotid artery. World J. Surg. 2001; 25:1028-1035.
- Sclafani SJ, Panetta T, Goldstein AS, et al. The management of arterial injuries caused by penetration of Zone III of the neck. J Trauma. 1985;25:871-881.
- Sclafani AP, Sclafani SJ. Angiography and transcatheter arterial embolization of vascular injuries of the face and neck. Laryngoscope. 1996;106:168-173.
- Reuben BC, Whitten MG, Sarfati M, Kraiss LW. Increasing use of endovascular therapy in acute arterial injuries: Analysis of the National Trauma Data Bank. J Vasc Surg. 2007;46:1222-1226.
- Rao PM, Ivatury RR, Sharma P, et al. Cervical vascular injuries: A trauma center experience. Surgery. 1993;114:527-531.
- Herrera DA, Vargas SA, Dublin AB. Endovascular treatment of penetrating traumatic injuries of the extracranial carotid artery. J Vasc Interv Radiol. 2011;22:28-33.
- Toursarkissian B, Allen BT, Petrinec D, et al. Spontaneous closure of selected iatrogenic pseudoaneurysms and arteriovenous fistulae. J Vasc Surg. 1997;25:803-809.
- Cox MW, Whittaker DR, Martinez C, et al. Traumatic pseudoaneurysms of the head and neck: Early endovascular intervention. J Vasc Surg. 2007;46:1227-1233.
- Herrera DA, Vargas SA, Dublin AB. Endovascular treatment of traumatic injuries of the vertebral artery. AJNR Am J Neuroradiol. 2008;29:1585-1589.
- Berne JD, Reuland KR, Villarreal DH, et al. Internal carotid artery stenting for blunt carotid artery injuries with an associated pseudoaneurysm. J Trauma. 2008;64:398-405.
- Yi AC, Palmer E, Luh GY, et al. Endovascular treatment of carotid and vertebral pseudoaneurysms with covered stents. AJNR Am J Neuroradiol. 2008;29:983-987.
- Coldwell DM, Novak Z, Ryu RK, et al. Treatment of posttraumatic internal carotid arterial pseudoaneurysms with endovascular stents. J Trauma. 2000;48:470-472.
- Cothren CC, Moore EE, Biffl WL, et al. Anticoagulation is the gold standard therapy for blunt carotid injuries to reduce stroke rate. Arch Surg. 2004;139:540-545.
- DuBose J, Recinos G, Teixeira PG, et al. Endovascular stenting for the treatment of traumatic internal carotid injuries: Expanding experience. J Trauma. 2008;65:1561-1566.
- Gemmete JJ, Ansari SA, Gandhi DM. Endovascular techniques for treatment of carotid-cavernous fistula. J Neuro-Ophthalmol. 2009;29:62–71.
- Higashida RT, Halbach VV, Tsai FY, et al. Interventional neurovascular treatment of traumatic carotid and vertebral artery lesions: Results in 234 cases. AJR Am J Roentgenol. 1989;153:577-582.
- Kirsch M, Henkes H, Liebig T, et al. Endovascular management of dural carotid-cavernous sinus fistulas in 141 patients. Neuroradiology. 2006;48:486-490.
- Vinchon M, Laurian C, George B, et al. Vertebral arteriovenous fistulas: A study of 49 cases and review of the literature. Cardiovasc Surg. 1994;2:359-369.
- Hoshino Y, Kurokawa T, Nakamura K, et al. A report on the safety of unilateral vertebral artery ligation during cervical spine surgery. Spine. 1996;21:1454-1457.
- Fusco MR, Harrigan MR. Cerebrovascular dissections—A review part I: Spontaneous dissections. Neurosurgery. 2011;68:242-257.
- Ozdoba C, Sturzenegger M, Schroth G. Internal carotid artery dissection: MR imaging features and clinical-radiologic correlation. Radiology. 1996;199:191-198.
- Engelter ST, Brandt T, Debette S, et al. Antiplatelets versus anticoagulation in cervical artery dissection. Stroke. 2007;38:2605-2611.
- Lyrer P, Engelter S. Antithrombotic drugs for carotid artery dissection. Cochrane Database Syst Rev. 2010;10:CD000255.
- Cothren CC, Moore EE, Biffl WL, et al. Anticoagulation is the gold standard therapy for blunt carotid injuries to reduce stroke rate. Arch Surg. 2004;139:540-546.
- Hinson HE, Stallmeyer MJ, Furuno JP, et al. Antithrombotic therapy and outcomes of cervical arterial dissection in the trauma patient: A case series. J Trauma Manag Outcomes. 2010;4:13.
- Fabian TC, Patton JH, Jr., Croce MA, et al. Blunt carotid injury: Importance of early diagnosis and anticoagulant therapy. Ann Surg. 1996;223:513-525.
- Sturzenegger M. Spontaneous internal carotid artery dissection: Early diagnosis and management in 44 patients. J Neurol. 1995;242: 231-238.
- Arauz A, Hoyos L, Espinoza C, et al. Dissection of cervical arteries: Long-term follow-up study of 130 consecutive cases. Cerebrovasc Dis. 2006;22:150-154.
- Gonzales-Portillo F, Bruno A, Biller J. Outcome of extracranial cervicocephalic arterial dissections: A follow-up study. Neurol Res. 2002;24:395-398.
- Schwartz NE, Vertinsky AT, Hirsch KG, Albers GW. Clinical and radiographic natural history of cervical artery dissections. J Stroke Cerebrovasc Dis. 2009;18:416-423.
- Donas KP, Mayer D, Guber I, et al. Endovascular repair of extracranial carotid artery dissection: current status and level of evidence. J Vasc Interv Radiol. 2008;19:1693-1698.
- Edwards NM, Fabian TC, Claridge JA, et al. Antithrombotic therapy and endovascular stents are effective treatment for blunt carotid injuries: Results from long term followup. J Am Coll Surg. 2007;204:1007-1013.
- Geibprasert S, Pongpech S, Armstrong D, Krings T. Dangerous extracranial-intracranial anastomoses and supply to the cranial nerves: Vessels the neurointerventionalist needs to know. AJNR Am J Neuroradiol. 2009;30:1459-1468.
- Koh E, Frazzini VI, Kagetsu NJ. Epistaxis: Vascular anatomy, origins, and endovascular treatment. AJR Am J Roentgenol. 2000;174:845-851.
- Razack N, Goldstein J, Jensen ME. Evaluation of the patient with epistaxis. Appl Radiol. 2001;30:18-21.
- Breda SD, Choi IS, Persky MS, Weiss M. Embolization in the treatment of epistaxis after failure of internal maxillary artery ligation. Laryngoscope. 1989;99:809-813.
- Layton KF, Kallmes DF, Gray LA, Cloft HJ. Endovascular treatment of epistaxis in patients with hereditary hemorrhagic telangiectasia. AJNR Am J Neuroradiol. 2007;28:885-888.
- Willems PWA, Farb RI, Agid R. Endovascular treatment of epistaxis. AJNR Am J Neuroradiol. 2009;30:1637-1645.
- Chen YF, Lo YC, Lin WC, et al. Transarterial embolization for control of bleeding in patients with head and neck cancer. Otolaryngol Head Neck Surg. 2010;142:90-94.
- Luo CB, Teng MM, Chang FC, Chang CY. Transarterial embolization of acute external carotid blowout syndrome with profuse oronasal bleeding by N-butyl-cyanoacrylate. Am J Emerg Med. 2006;24:702-708.
- Chang FC, Lirng JF, Luo CB, et al. Patients with head and neck cancers and associated postirradiated carotid blowout syndrome: Endovascular therapeutic methods and outcomes. J Vasc Surg. 2008;47:936-945.
- Sorial E, Valentino J, Given CA, et al. The emergency use of endografts in the carotid circulation to control hemorrhage in potentially contaminated fields. J Vasc Surg. 2007;46:792-798.