Cardiac CT angiography: Patient-centric low-dose imaging
Images
Coronary arterial atherosclerotic disease (CAD) has been and continues to be a significant source of patient morbidity and mortality. Historically, symptomatic patients often presented later in the disease course, leading to high intensity care and allocation of healthcare resources. Concomitant cardiopulmonary disease as well as atherosclerotic disease in other vascular territories further exacerbated outcomes and expenditures.1-4
Over the past 20 to 30 years, a paradigm shift focusing on cardiovascular preventive care, early diagnosis, and minimally invasive diagnostic and therapeutic procedures has led to trends of decreasing morbidity and mortality along with decreasing intensity of healthcare services.1-5 Between 2000 and 2010, the American Heart Association reported a 30% decline in cardiovascular mortality per 100,000 people, decreasing from 0.34% to 0.24%.1 These trends are important and will continue to drive healthcare policy and practice given the potential for reduced healthcare expenditures and improved healthcare efficiency and utilization of healthcare resources.
Cardiac computed tomography (CT) has emerged as an important imaging tool in the diagnosis of CAD. In addition to being noninvasive, advantages include high spatial resolution, fast patient throughput, and relatively low cost compared to other advanced cardiovascular modalities. Its emergence has contributed to the ability to diagnose and manage CAD earlier with less invasive diagnostic risk and greater cost savings.6
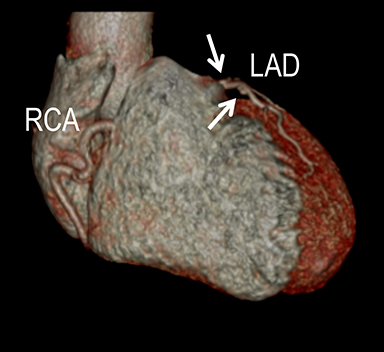
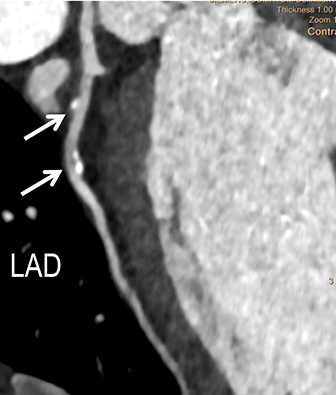
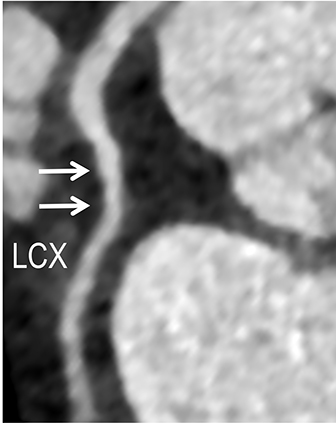
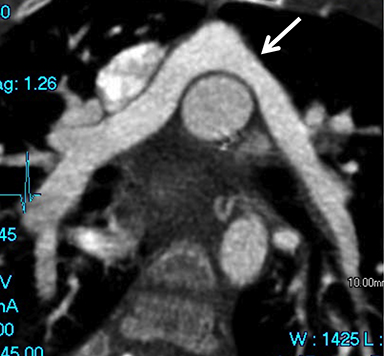
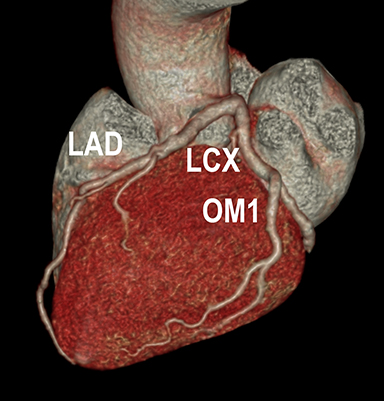
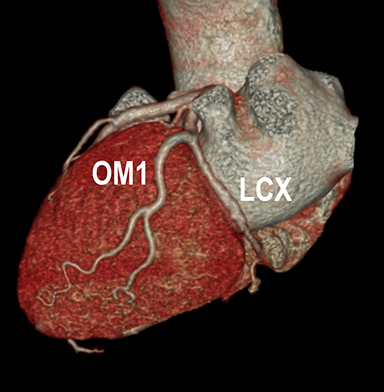
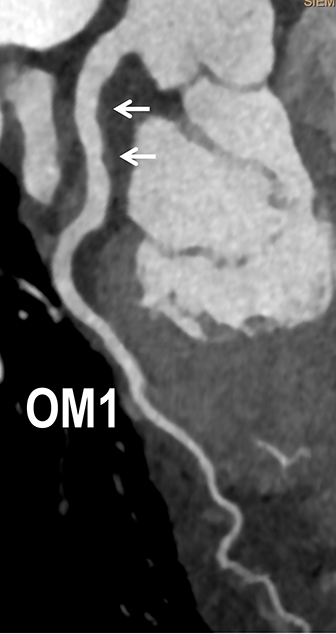
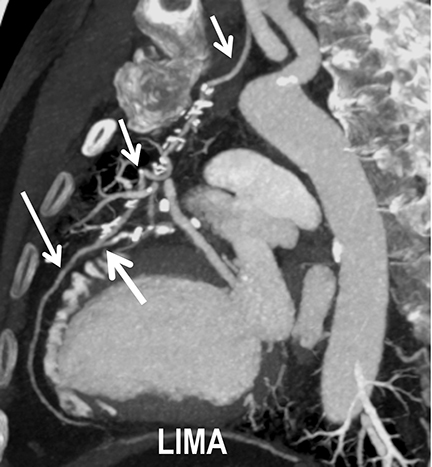
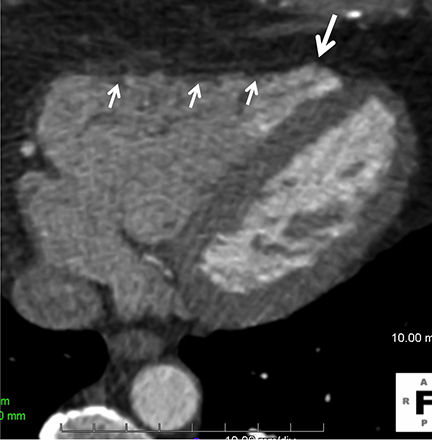
In current best practice medicine, coronary artery calcium scoring CT (CCSCT) is essential to preventive screening and CAD risk stratification for patients with intrinsic and behavioral risk factors.7-8 Intrinsic risks include a family history for atherosclerosis, hyperlipidemia, hypertension and diabetes. Behavioral risks include tobacco use, obesity, sedentary lifestyle, and an unbalanced, “unhealthy” cardiometabolic diet. Current best practice clinical care strategies utilize coronary CT angiography (CCTA) for noninvasive coronary angiographic evaluation in symptomatic patients who have low (<10%) and intermediate pretest (10-90%) probabilities for obstructive CAD.8 Additional common indications for CCTA include patients with 1) typical or atypical chest pain and normal or equivocal stress tests and/or ECG findings; 2) unexplained or atypical chest pain with low risk for CAD and a possible coronary artery anomaly; 3) unexplained acute chest pain with low or intermediate risk, negative cardiac enzymes, and normal or equivocal ECG findings; and 4) preoperative or pre-procedural evaluation of the coronary arteries.6-9 One of the main concerns with CCSCT and CCTA has been radiation exposure. Dose reduction strategies combined with iterative reconstruction both at the CT acquisition and image workstation domain levels have dramatically improved the ability to lower radiation exposure while maintaining diagnostic image quality.10-11 In our cardiac CT practice using a 64- and 128-channel multidetector-row CT (MDCT), with appropriate dose reduction strategies and iterative reconstruction, patients may receive as low as a one-chest-radiograph-equivalent dose exposure (0.2mSv) for CCSCT and a two- to three-radiograph-equivalent dose exposure (0.4-0.6mSv) for coronary CCTA (Figure 1). Across the spectrum of cardiac CT practice, however, there remains wide variability in CT technique and radiation exposure. As an example, in the multicenter PROMISE trial examining 10,003 symptomatic patients undergoing CCTA versus coronary functional testing, the average CCTA radiation exposure was 12.0mSv.12 This performance is not significantly lower compared to early simulated and actual CCTA performance, when authors reported as high as 50-100 radiograph equivalent exposures for CCTA.13
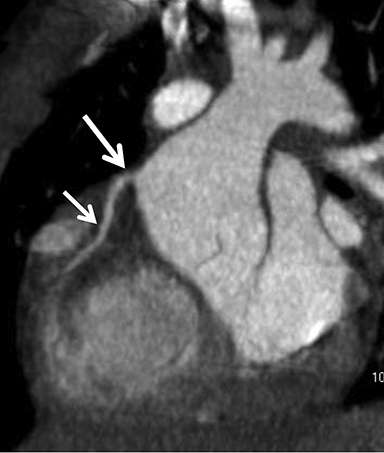
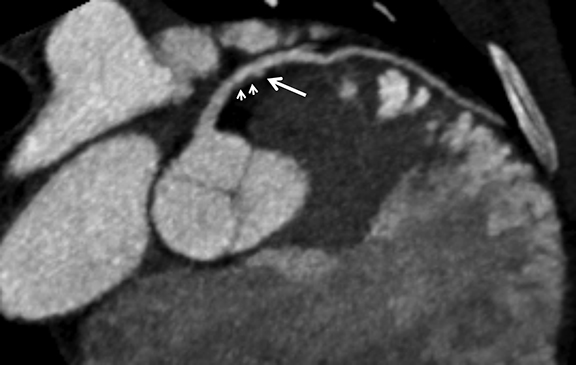
To maximize the full potential use of cardiac CT and its impact on disease detection, patient outcome and healthcare economics in both adult and pediatric (Figure 2) patients, it is essential to apply dose reduction strategies. Cardiac CT imagers should strive to deliver radiation doses that are As Low As Reasonably Achievable (ALARA). As will be discussed and illustrated in the context of CAD, this requires careful review and consideration of all direct and indirect technical parameters which may impact radiation exposure, image noise and diagnostic image quality. Through this patient-centric approach, dose exposure and image quality are optimized while radiation risk is mitigated for each individual patient.
CTA Technique
Acceptable noise, and thus how low radiation can be targeted, will vary among cardiac CT readers and patient care providers. It is important to reach a balance, as excessive noise may diminish the accuracy of exam interpretation and confidence in using the exam for clinical decision making. While recommended protocols are needed for efficient workflow, selection of the acquisition parameters and ancillary dose reduction strategies should be made specific to each patient. This will depend upon a patient’s body habitus, the ability to deliver high intravascular iodine content and the MDCT hardware and software configurations.
Patient preparation
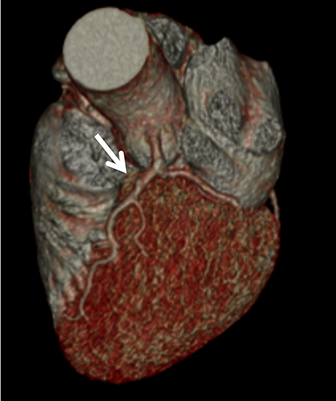
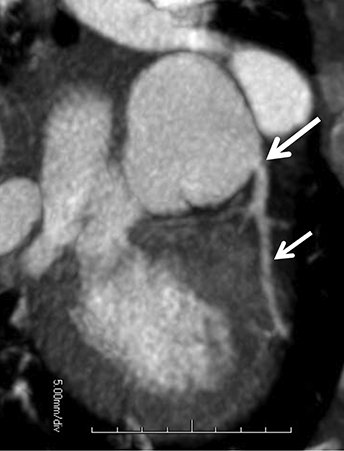
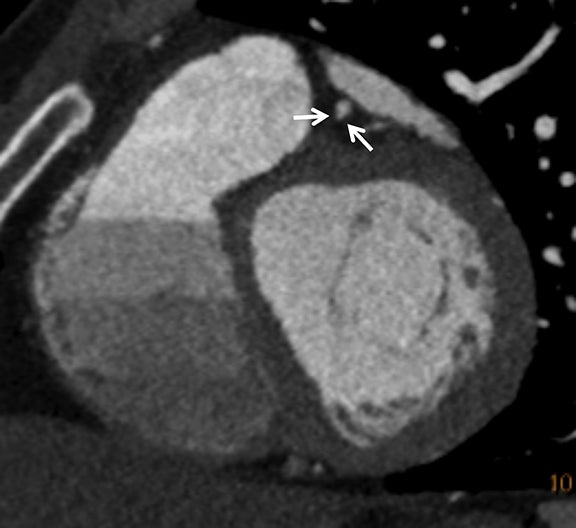
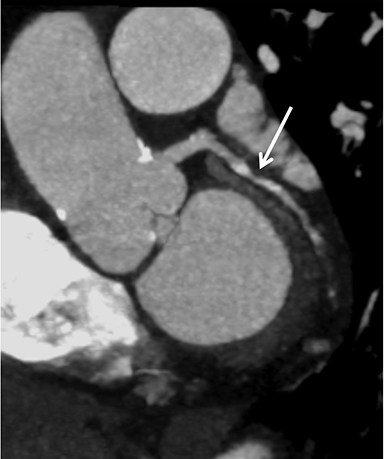
Among recommended steps to prepare for a low-dose cardiac CCTA, initial review centers on a patient’s body mass index (BMI), renal function, vein site and size for intravenous (IV) contrast delivery, and heart rate (Figure 3). The BMI is a primary determinant in the selection of radiation parameters while renal function and venous access will factor into contrast medium delivery and prevention of adverse events. In general, large gauge IV catheters (eg, 18g) placed in larger caliber peripheral veins (eg, antecubital fossa) will safely allow for high contrast flow rates. The higher flow rates are necessary to optimize iodine delivery and the intravascular contrast to noise ratio (CNR). Heart rate control to an ideal range of 50-60 bpm is advised. This affords selection of prospective ECG gating with confidence that image quality will not be compromised by cardiac motion. Cardiac motion may exacerbate image noise while rendering segments non-diagnostic
When the patient is placed on the CT gantry table, several measures should be taken to optimize photon delivery and minimize the impact of noise. First, the patent should be positioned with the targeted region isocenter in the gantry. This will yield more uniform photon delivery. Next, all external metallic objects should be removed from the expected scan region. The high-density material will limit photon exposure, cause streak artifact, and may also exacerbate potential increased noise when prescribing low-dose parameters. Raising the upper extremities out of the field of view will reduce noise resulting from the increased soft tissue attenuation. Once the exact position is finalized, breast shields can be applied for female patients; general shielding of non-targeted anatomy is also a consideration.
Acquisition
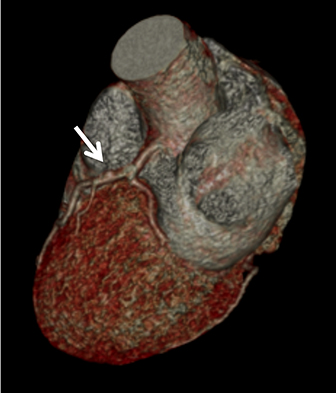
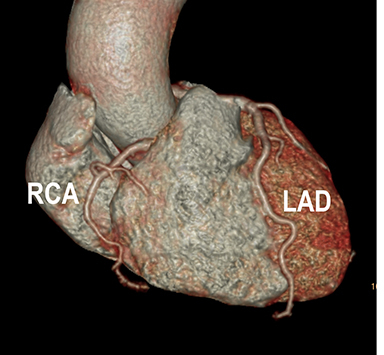
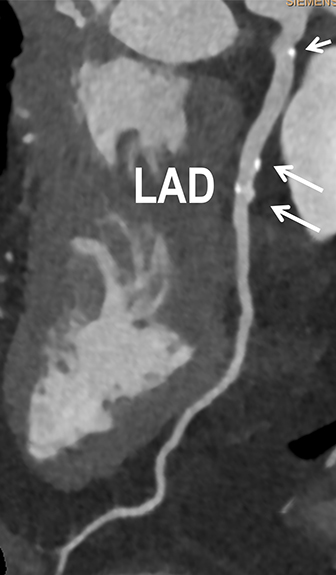
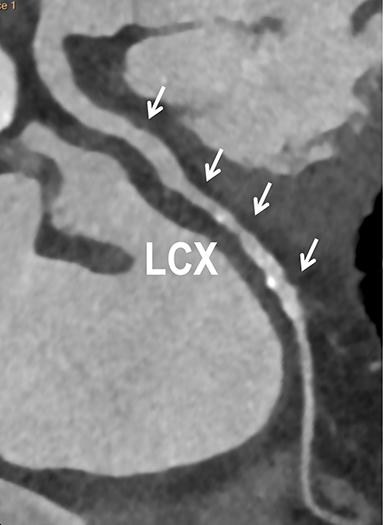
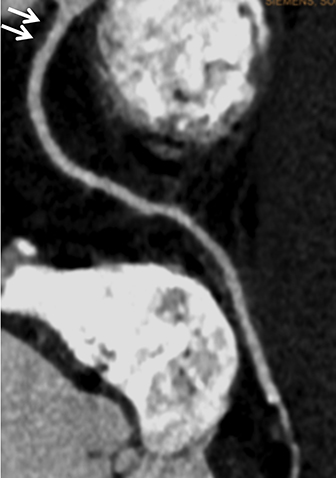
The objective of cardiac CT angiography is to synchronize image acquisition with the delivery of iodine at the peak of arterial enhancement within the left-sided cardiac chambers, coronary arteries and aorta. The quality of the CCTA images is characterized by the degree and detail of enhanced structures in relationship to background organs and soft tissues (Figure 4). Acquisition parameters are selected to maximize spatial and contrast resolution while minimizing noise and artifacts. Artifacts may include cardiac and arterial pulsation, respiratory motion, patient motion, high density streaks, beam hardening and edge pixilation.
Spatial resolution, as defined by the matrix size, is controlled by the detector width and collimation configuration. For cardiac CT angiography, isotropic submillimeter acquisitions are necessary for the highest spatial resolution. Noise and edge pixilation are inversely proportional to the square root of matrix size, such that noise will increase when reducing the detector width.14 Reconstruction into thicker datasets can reduce noise at a cost of reduced spatial resolution and diagnostic accuracy. To avoid compromising spatial resolution, reducing radiation parameters in moderate and high BMI patients should proceed with caution.
Contrast resolution is directly dependent upon the degree of iodine attenuation and the tube voltage (kVp). Single-source and single-energy MDCT scanners may have up to five voltage options: 70, 80, 100, 120, and 140. Reducing the voltage will increase iodine attenuation and the relative intra-cardiac and vascular density as the photons will be closer to the k-edge of iodine (33.2keV).15 Reducing the voltage has the additional benefit of reducing radiation dose. This occurs, however, at the cost of increased noise.15 Automated voltage determination software on select scanners can assist in accurate kVp selection to optimize reduced dose and image quality.
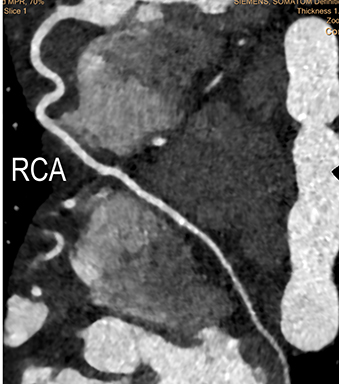
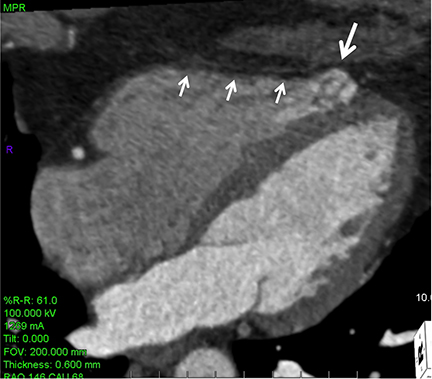
Based upon the anticipated noise and what the operator feels will be acceptable noise for a given patient’s BMI, the operator can manipulate the tube current (mA) and exposure time (seconds) to control the flow of photons and radiation exposure (tube current x exposure time, mAs). Increasing exposure inversely decreases noise by the square root of the exposure while directly increasing the amount of radiation dose.16 Automated tube current modulation affords greater objective control and balance between reduced exposure and noise (Figure 5). ECG-pulsing of the tube current should be utilized in retrospective ECG-gated CT angiograms (Figures 6 and 7).
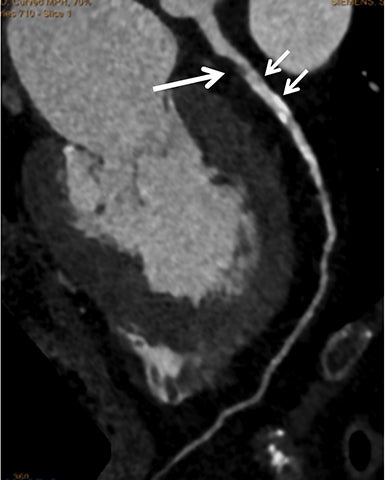
Weight-based (eg, BMI) voltage and tube current parameters are recommended. Parameters may require modification, depending upon the MDCT scanner; contrast delivery protocol; patient positioning; and presence of metallic devices, stents, surgical clips, wires, and other high density material; and the preference of the cardiac imager, primary care provider, or both (Figures 5 and 6). When imaging younger pediatric patients (eg, coronary anomaly to complex congenital heart disease, Figure 2), voltage and tube current adjustments may be more aggressive depending upon the patient’s age and relative organ radiosensitivities. For both adult and pediatric patients undergoing contrast-enhanced cardiac CT, the utilization and degree of iterative reconstruction techniques will also factor into how aggressively an operator will reduce dose, specific to the patient and needs to maintain diagnostic image quality.10-11
Protocol series
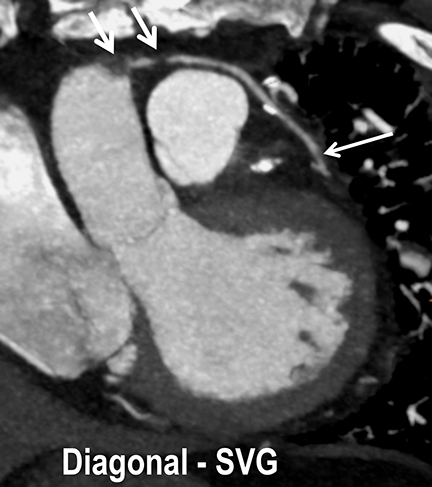
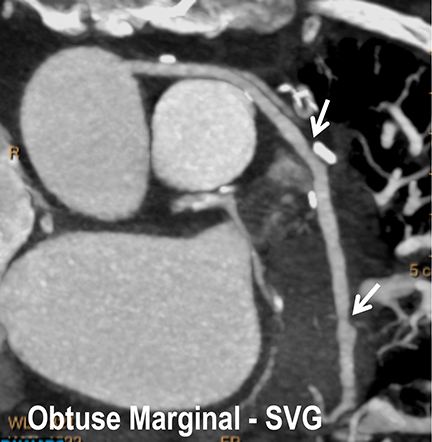
A CCTA protocol typically has 4 series. The first is the required topogram, necessary to prescribe the axial images. To reduce radiation exposure, a single anterior-posterior topogram is recommended. The topogram voltage should be set to equal the anticipated voltage for the angiographic acquisition, in order to utilize automated tube current modulation. The second series is the CCS acquisition with prospective ECG-gating. Scoring requires the acquisition of 3-mm-thick images at 120kVp. The tube current, however, can be reduced to an appropriate level in accordance with ALARA principles; iterative reconstruction can be utilized with CCS datasets to optimize quality. The third series is a timing acquisition (automated bolus triggering vs. test bolus), which is essential for precise synchronization of cardiovascular enhancement and image acquisition. Reasonable low-dose parameters should be selected. By nature of the cardiac CT angiogram, all protocols require the fourth series, in which the contrast enhanced angiographic images are obtained. Precise attention to technical detail is essential to avoid repeating the series and delivering additional radiation dose. The Z-axis field of view coverage should not extend beyond the cardiac region of interest. For patients who do not require functional evaluation and have regular heart rates of 50-60 beats per minute, prospective ECG-gating is recommended (Figures 1, 3-5). Images are acquired with lower anatomical sampling as compared to retrospective ECG-gating and as such, results in lower exposure. At the discretion of the imager, prospective versus retrospective ECG-gating may be chosen when the heart rate is between 66 and 70 bpm (Figure 2). If proceeding forward with a heart rate of 71-80 bpm, retrospective ECG-gating is recommended. This will afford cardiac multiphase arterial segment interrogations along with the added benefit of chamber and valvular functional evaluations (Figure 7). The cost, however, is higher radiation exposure. Exposure and image quality can be controlled with core radiation reduction strategies and iterative reconstruction (Figures 6 and 7).
Contrast medium delivery
As noise increases, to achieve a constant CNR for a given cardiovascular territory (eg. contrast resolution), the intra-cardiovascular iodine attenuation (density) must increase. In addition to decreasing the voltage, the other means to increase iodine attenuation is to directly increase the iodine content in the targeted territory. The strategy is to deliver an appropriate weight-based amount of iodine (iodine dose) at an appropriate, relatively fast rate (iodine flux) and acquire images when the iodine reaches peak accumulation in the targeted territory. The amount of iodine and volume of contrast that can be safely delivered is limited by a patient’s body weight and renal function, while the rate of injection (eg, 5-5.5ml/sec) is limited by the venous access. High- concentration (350-370mgI/ml) contrast medium is recommended to optimize the iodine dose and reduce the volume of contrast (Figure 4).
A dual-lumen power injector is recommended for contrast delivery. Infusion of saline (in the second lumen) following the contrast injection assists in the low dose CCTA initiative by improving contrast volume utilization, prolonging the peak of injection, and reducing perivenous streak artifacts. The saline volume varies depending upon the injector tubing length and body weight. The rate of injection should equal the contrast injection rate.
Practice quality improvement
Routine quality assurance reviews of patient-centric low-dose cardiac CT examinations are recommended. The objectives are to confirm safe radiation practice, safe contrast delivery, and acceptable and reliable image quality.
Reviews should address the prescribed parameters, exam coverage, contrast medium administration, radiation output (eg. CTDIvol, DLP), absorbed patient radiation dose, CNR, image noise, motion and other artifacts. Subjective image quality feedback should be obtained from both readers and patient care providers. Where needed, protocols should be revised to improve the success of low dose and ultra-low dose cardiac CT imaging.
Conclusion
Screening for CAD and primary and secondary preventive clinical management have made a significant impact on decreasing cardiac morbidity and mortality. Cardiac CT has emerged over the past 10 years as an important imaging tool for the diagnosis of coronary arterial disorders. Cardiac CT has contributed to the ability to diagnose and manage CAD earlier with less invasive diagnostic risk and greater cost savings. To realize the full potential of cardiac CT to impact disease detection, patient outcomes and healthcare economics, it is essential to apply dose reduction strategies through a patient-centric approach. In this manner, radiation exposure and image quality are optimized, while a patient’s radiation risk is mitigated.
References
- Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics – 2014 update. Circulation. 2014;129:e28-e292.
- Smoldern KG, Bell A, Lei Y, et al. One-year costs associated with cardiovascular disease in Canada: Insights from the Reduction of Atherothrombosis for Continued Health (REACH) registry. Can J Cardiol. 2010; 26(8):297-305.
- Smoldern KG, Wang K, de Pouvourville G, et al. Two-year vascular hospitalisation rates and associated costs in patients at risk of atherothrombosis in France and Germany: highest burden for peripheral arterial disease. Eur J Vasc Endovasc Surg. 2012; 43(2):198-207.
- Ademi Z, Liew D, Gorelik A, et al. Predictors of healthcare use among patients with or at high risk of atherothrombotic disease: two-year follow-up data. Int J Cardiol. 2014; 175(1):72-77.
- Ritchey MD, Wall HK, Gillespie C, et al. Million hearts: Prevalence of leading cardiovascular disease risk factors – United States, 2005-2012. MMWR Morb Mortal Wkly Rep. 2014; 63(21):462-467.
- Poon M, Cortegiano M, Abramowicz A, et al. Routine coronary computed tomography angiography reduces unnecessary hospital admissions, length of stay, recidivism rates, and invasive coronary angiography in the emergency department triage of chest pain. J Am Coll Cardiol. 2013;62(6): 543-552.
- Wolk MJ, Bailey SR, Doherty JU, et al. ACCF / AHA / ASE / ASNC / HFSA / HRS / SCAI / SCCT / SCMR / STS 2013 multimodality appropriate use criteria for the detection and risk assessment of stable ischemic heart disease. J Am Coll Cardiol. 2014; 63(4):380-406.
- Taylor AJ, Cerqueira M, Hodgson JM, et al. ACCF / SCCT / ACR / AHA / ASE / ASNC / NASCI / SCAI / SCMR 2010 Appropriate use criteria for cardiac computed tomography. J Am Coll Cardiol. 2010; 56(22)1864-1894.
- Cury RC, Budoff M, Taylor AJ. Coronary CT angiography versus standard of care for assessment of chest pain in the emergency department. J Cardiovascular Comput Tomogr. 2013; 7(2):79-82.
- Hellinger JC, Jacobs S, Salazar P, et al. Image quality improvement using a 3D edge-sensitive noise reduction filter in low dose pediatric retrospective ECG-gated cardiac CT angiography. J Cardiovasc Comput Tomogr. 2011; 5(4S):S6-7.
- Kassam Z, Arifi B, Nura G, et al. Ultra-low dose coronary calcium CT with iterative reconstruction: enhanced image quality with accurate risk stratification. J Cardiovasc Comput Tomogr. 2013;7(3):S52
- Douglas PS, Hoffman U, Patel MR, et al. Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med. 2015; 372(14):1291-1300.
- Einstein AJ, Henzlova MJ, Rajagopalan S. Estimating risk of cancer associated with radiation exposure from 64-slice computed tomography coronary angiography. JAMA. 2007; 298(3)317-323.
- Goodsitt MM, Johnson RH. Precision in quantitative CT: impact of x-ray dose and matrix size. Med Phys. 1992;19(4):1025-1036.
- Huda W, Ogden KM, Khorasani MR. Effect of dose metrics and radiation risk models when optimizing CT x-ray tube voltage. Phys Med Biol. 2008; 53(17): 4719-4732.
- Kalender WA, Wolf H, Suess C, et al. Dose reduction in CT by on-line tube current control: principles and validation on phantoms and cadavers. Eur Radiol. 1999;9(2): 323-328.