Cirsoid renal arteriovenous malformation
Images
CASE SUMMARY
A 57-year-old Caucasian female presents with recurrent urinary tract infections and hematuria. She reports intermittent left flank and pelvic pain, which she has had for several months. Past medical history of hyperlipidemia and osteoarthritis. Past surgical history includes cystoscopy and hysterectomy. No family history of recurrent urinary tract infections or hematuria. Physical exam of the patient was non-contributory, and the only lab value of significance was a serum creatinine 0.6 mg/dL.
IMAGING FINDINGS
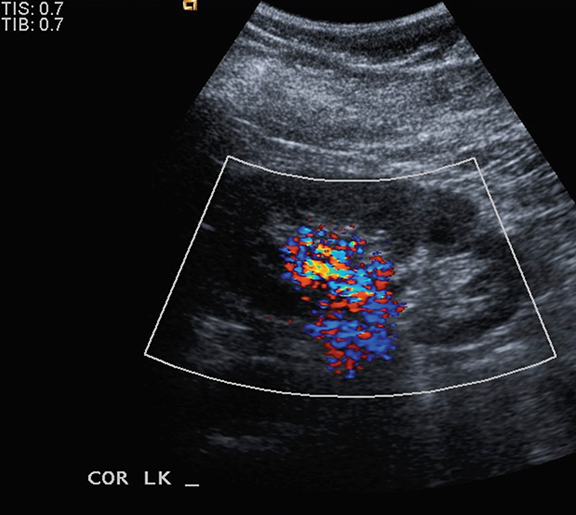
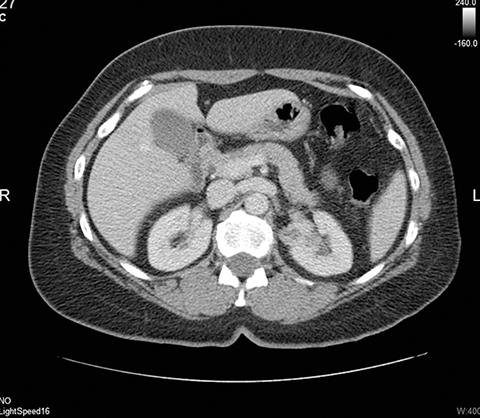
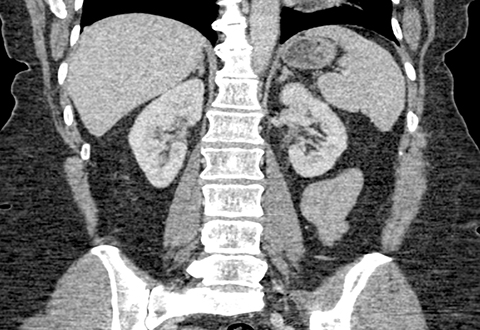
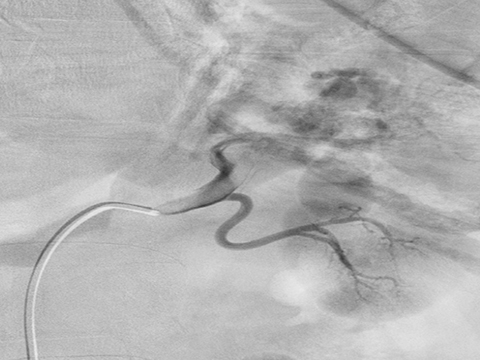
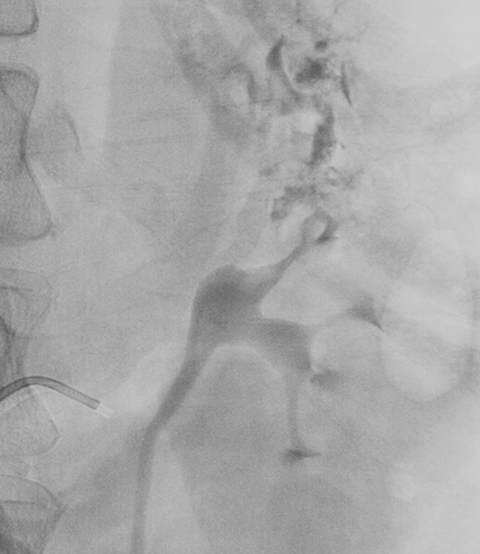
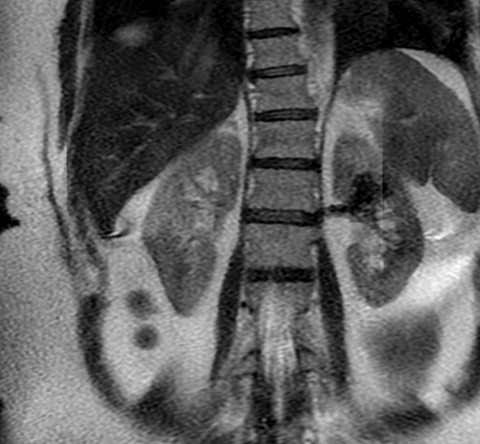
Ultrasound shows a left upper pole renal arteriovenous malformation, which measures approximately 2.6cm in diameter (Figure 1). CT urogram shows a lobular mass within the left upper pole kidney, which was indeterminate but suspicious for vascular malformation (Figure 2). Left renal arteriogram shows a large cirsoid type renal arteriovenous malformation. This occupies the majority of the upper and central interpolar kidney (Figure 3). Coronal MRI shows a focal mass like area exhibiting characteristics of a flow void in the upper and central interpolar left kidney (Figure 4).
DIAGNOSIS
Cirsoid renal arteriovenous malformation
DISCUSSION
Renal arteriovenous malformations (AVM) are rare lesions that can be congenital or acquired. Acquired AVMs are more common, accounting for approximately 70% of cases, and typically are a result of previous biopsy, trauma, infection, malignancy, or renal surgery. Congenital AVMs accounts for the other 20%. Although the exact cause of congenital AVMs is unknown, it is thought to be associated with a defect in the elastic component of the vessel. There are two variants of congenital AVMs. “Cirsoid” variant are described as having a tortuous knotted appearance of multiple arterial vessels with interconnecting fistula. The other variant, called “cavernous” or “angiomatous,” has a single arterial vessel supply that feeds many small connecting fistulas.2 Acquired AVMs are characterized by a single direct communication between an artery and a vein without an intervening vascular nidus.3 Congenital AVMs usually present with hematuria in up to 72% of cases, while acquired AVMs are more likely to present with hemodynamic changes, such as hypertension, cardiomegaly and congestive heart failure.1 The peak incidence occurs in patients between 30 and 40 years of age and women are affected twice as much as men.4
The gold standard for diagnosing renal AVM is angiography. Color Doppler US is the first-line imaging due to its low cost and ability to easily identify turbulent flow. However, lighter-colored flow of a small AVM can be obscured by the flow of normal vessels grouped in the hilum. Additionally, Color Doppler US has difficulty differentiating between the turbulent flow of an AVM and the flow from an aneurysm.6 Mulitdecetor Row CT has improved the depiction of the renal veins in the early stage of the arterial phase; allowing for minimally invasive, three-dimensional visualization of the arteries and veins.7 Classic magnetic resonance imaging is capable of distinguishing high-flow malformations suggestive of AVMs as demonstrated by areas of multiple flow voids on gradient-echo images.8 Although MRI without contrast can be a useful imaging modality in patients with concerning renal function or with contraindications for CT, it is often more costly and time consuming than other available modalities.9
REFERENCES
- 1. Crotty KL, Orihuela E, Warren MM. Recent advances in the diagnosis and treatment of renal arteriovenous malformations and fistulas. J Urol. 1993;150:1355–1359.
- Tarif N, Mitwali A, Samayer S, Abu-Aisha H, Memon N, Sulaimani F, Alam A, Al Wakeel J. Congenital renal arteriovenous malformation presenting as severe hypertension. Nephrol. Dial. Transplant. 2002;17(2):291-294.
- Cura M, Elmerhi F, Suri R, Bugnone A, Dalsaso T. Vascular malformations and arteriovenous fistulas of the kidney. Acta Radiol. 2010;51:144-149.
- Messing E, Kessler R, Kavaney PB. Renal arteriovenous fistulas. Urology. 1976;8:101–107.
- Muraoka N, Sakai T, Kimura H, et al. Rare causes of hematuria associated with various vascular diseases involving the upper urinary tract. Radiographics. 2008;28(3)855–867.
- Takebayashi S, Aida N, Matsui K. Arteriovenous malformations of the kidneys: diagnosis and follow-up with color Doppler sonography in six patients. AJR Am J Roentgenol. 1991;157:991–995.
- Takeuchi, N., Nomura Y. Ruptured renal arteriovenous malformation successfully treated by catheter embolization: a case report. BMC Res Notes. 2014;7(19).
- Chimpiri, A. Rao, and Balasubramani Natarajan. Renal vascular lesions: Diagnosis and endovascular management. Seminars in Interventional Radiology. 2009;26.3:253–261. PMC. Web. 13 Apr. 2015.
- Donnelly L., Adams D., Bisset G. Vascular malformations and hemangiomas: a practical approach in a multidisciplinary clinic. AJR Am J Roentgenol. 2000;174:597-608.
Citation
D K, N P, T E, R C. Cirsoid renal arteriovenous malformation. Appl Radiol. 2016;(6):35-37.
June 6, 2016