Computed Tomography: Spinning a Revolution in Pediatric Imaging
Images



Over the past 50 years, computed tomography (CT) has had a profound effect on our understanding of pediatric diseases. Although considered a mature technology, CT has undergone a remarkable evolution that has made the modality a mainstay of pediatric radiology.
The Birth and Infancy of CT
The earliest CT scanners of the 1980s made the modality impractical for imaging children. Long scan times, in particular, caused respiratory misregistration and severe motion artifacts. That began to change in the 1990s, however, with the introduction of the four-row multidetector spiral CT, ushering in the era of pediatric CT. 1 Spiral scanning permitted faster gantry rotation times and better temporal resolution, substantially reducing—but not eliminating—motion artifacts.
In those days, alleviating patient motion in pediatric CT meant intravenous sedation and occasional general anesthesia in patients younger than five years. Sedation remained widely utilized through the 1990s and early 2000s, but even with that limitation, CT imaging became routine in the evaluation of pediatric trauma, tumors, complicated infections, and some lung and vascular diseases.
Four-detector ultimately gave way to 16-, and then 64-detector technology, but gantry rotation speeds still fell short of eliminating motion artifacts in most children.
Adolescence: Dual-Source CT
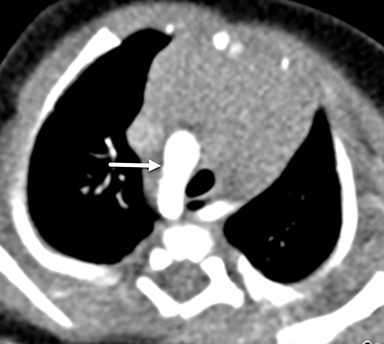
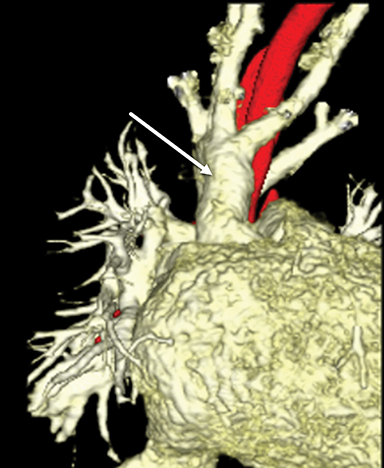
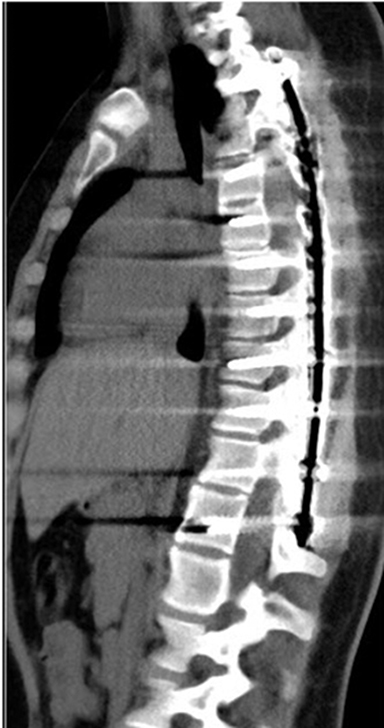
A game changer arrived with the 2006 debut of the dual-source CT scanner, which featured two tubes and two corresponding detectors. On the single-source system, pitch was limited to less than 1.5; scans acquired at higher pitch values resulted in sampling gaps and led to image reconstruction artifacts. Dual-source CT, conversely, allowed for pitch values as high as 3.4, permitting faster table speeds and shorter scan times as the second detector filled in the sampling gaps. The clinical benefit was a high-quality, motion-free CT scan that covered large anatomic areas and eliminated the need for sedation in infants and children (Figures 1,2).2
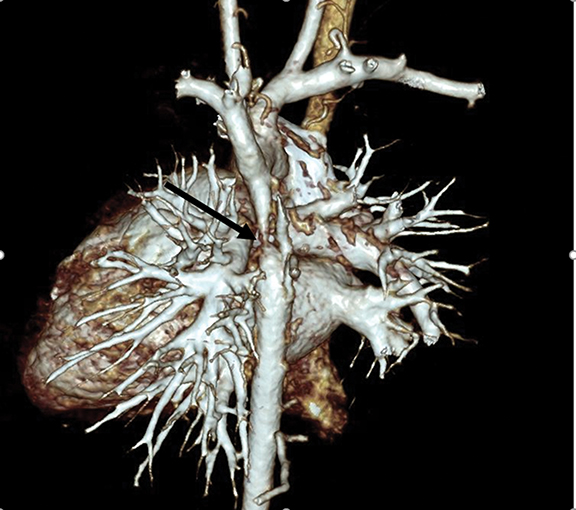
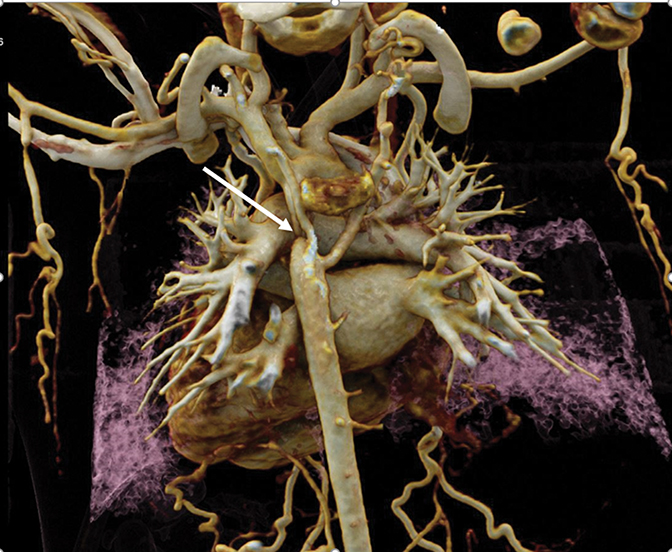
Multidetector CT boasted another significant advantage: expanding image interpretation from transverse reconstructions to volumetric visualization and analysis. This led to a greater role for CT in evaluating congenital heart, vascular, and coronary artery diseases. The recent addition of cinematic rendering has led to more photorealistic 3D images than was possible with other 3D reconstruction methods (Figure 3).
Early Adulthood: Making CT Safer for Kids
After the race for detector power slowed down, the race for dose reduction sped up. This ushered in technologies to automate the selection of key acquisition parameters that had a strong impact on dose, most notably tube current (milliamperage, mAs) and peak tube voltage (kilovoltage, kVp).3,4 The ability to individualize mAs and kVp to patient size profoundly impacted dose reduction.
Automated tube current modulation emerged in the late 1990s, replacing longstanding fixed tube current. Automated kilovoltage imaging followed in the early 2000s, lowering the voltage from 120 to 70 and 80 kVp in younger children Iterative reconstruction then appeared on the scene, cleaning up image noise associated with lower tube current and kilovoltage. The Image Gently® campaign launched in the 2000s brought national attention to the importance of dose reduction in pediatric CT imaging.
Not all dose reduction methods, however, were successful. Bismuth breast shields were widely recommended in the early 2000s, but it later became known that incorrect use of the shields actually raised the radiation dose and shielding was abandoned in 2012. 5
The story of dose reduction is not over. Recently, a tin (Sn) filter was added to dual-source systems for noncontrast chest CT examinations performed at 100 kVp. This filter attenuates low-energy photons that contribute to dose but generally contribute little to image quality and leads to significantly greater dose reduction compared to conventional CT protocols at 70, 80, and 100 kVp.
CT at Maturity: Improving Contrast, Optimizing Images
There was one more challenge in children that is not routinely present in adults; ie, inherently low levels of image contrast owing to smaller body size and less perivisceral fat. Early on, faster gantry rotations, dual-source geometries, and low kilovoltage imaging, in particular at 70 and 80 kVp, created images with high levels of iodine conspicuity, improving vascular imaging and lesion detection and characterization.
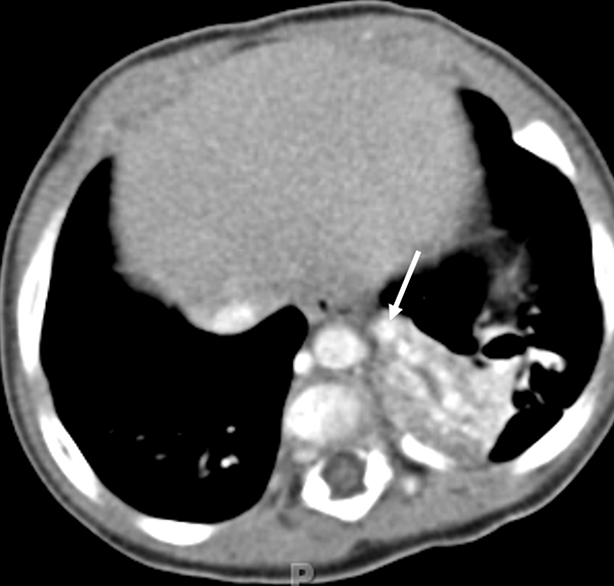
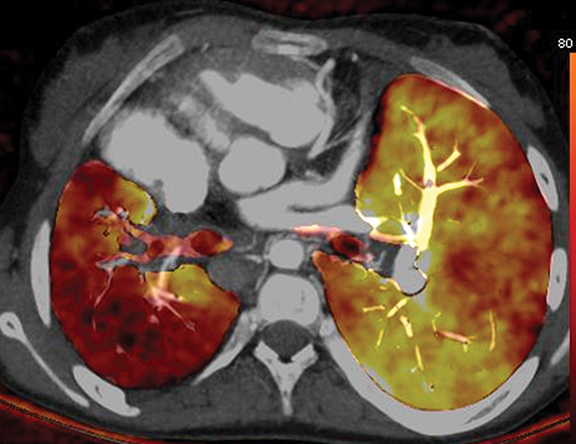
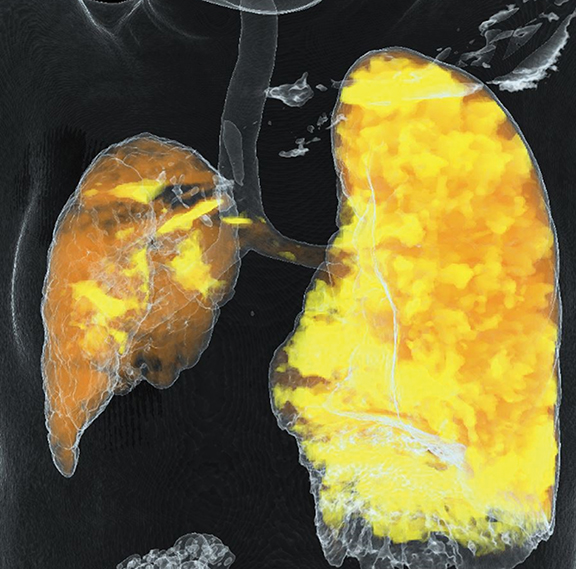
Image optimization improved even more when dual-energy CT arrived on the scene, allowing generation of monoenergetic and material decomposition images.6 The former play a role in improving image quality by reducing metal-implant artifacts and increasing contrast conspicuity in children with poor vascular enhancement related to slow injection rates and small intravenous catheters (Figure 4). The latter images expand CT into new clinical applications, such as assessing lung perfusion (Figure 5) and predicting tumor response to treatment.
Looking Ahead
What new developments can we expect in pediatric CT going forward? Near-term innovations include photon-counting CT to further improve image quality and reduce radiation output. Photon scanning will also offer more options for multi-energy imaging. In this era of precision or personalized medicine, look for greater use of radionomics, linking imaging phenotypes to genotypes.
The past few decades have undeniably been a golden age for pediatric imaging. If the past is any indication, this specialty can expect an even brighter future.
References
- Booij R, Budde R, Dijkshoorn ML, et al. Technological developments of X-ray computed tomography over half a century: User’s influence on protocol optimization. Eur J Radiol. 2020; 131, 109261. doi: 10.1016.
- Lell MM, May M, Deak P, et al. High-pitch spiral computed tomography. Effect on image quality and radiation doe in pediatric chest computed tomography. Invest Radiol. 2011; 46(2):116-123.
- Greess H, Lutze J, Noemary A, et al. Dose reduction in multislice spiral CT of children by online tube current modulation. Eur Radiol. 2004; 14:995–999
- Siegel MJ, et al. Effects of automated KV selection technology on contrast-enhanced pediatric CT. Radiology. 2013; 268(2); 538-547
- Marsh RM, Silosky M. Patient shielding in diagnostic imaging: discontinuing a legacy practice. AJR AM J Roentgenol. 2019; 212:755-757.
- Siegel MJ, Ramirez-Giraldo JC. Dual energy CT in children: Imaging algorithms and clinical applications. Radiology. 2019; 291:286-297
References
Citation
MJ S. Computed Tomography: Spinning a Revolution in Pediatric Imaging. Appl Radiol. 2021; (5):16-19.
September 10, 2021