Duplication of the inferior vena cava


Duplication of the inferior vena cava
Findings
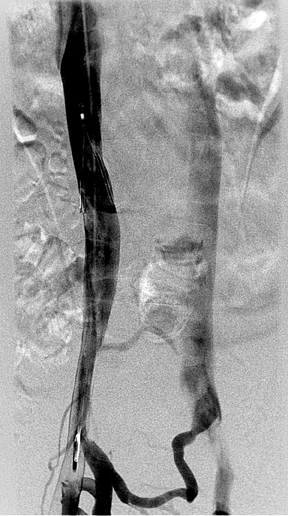
The original venogram was obtained by right common femoral approach. No thrombus was seen within the inferior vena cava (IVC), and the caliber of the cava was normal. However, the entire left border of the vessel was smooth and continuous, and inflow from the left common iliac was not appreciated (Figure 1).
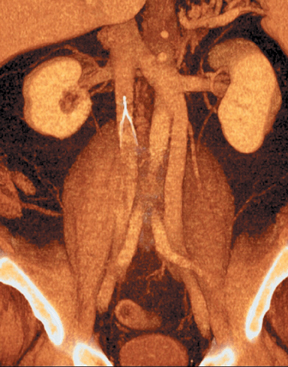
The CT scan demonstrated a double IVC (Figure 2) with thrombus in the right external iliac vein (not shown). The filter legs were expanded and no significant filter tilt was seen.
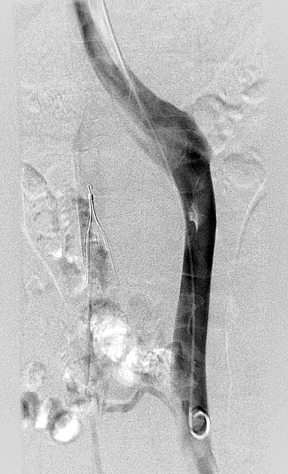
Using a right internal jugular approach, we advanced a pigtail catheter into the right common iliac vein and obtained a venogram. The venogram confirmed both right and left IVC moieties and communication via pelvic collaterals (Figure 3). We then positioned the pigtail catheter into the left IVC. Venography demonstrated a similar-caliber left IVC terminating in the left renal vein and then communicating with the common suprarenal IVC (Figure 4). We placed a second filter in the left IVC (Figure 5).
Discussion
Given the complex embryogenesis of the IVC, it is not surprising to encounter anatomic variations. Duplication of the IVC occurs in up to 2.8% of the population.1 Failure of the left caudal supracardinal vein to regress results in persistent communication between the left common iliac vein and the left renal vein. The left renal vein then typically crosses anterior to the aorta to join the right IVC. Though asymptomatic, double IVC has important clinical implications when attempting caval filtration or when retroperitoneal surgery is performed. The left IVC can also be misdiagnosed as lymphadenopathy on cross-sectional imaging.
Inferior vena cava filtration is commonly performed to prevent pulmonary embolism in patients with hypercoaguable states or documented venous thromboembolic disease and contraindications to anticoagulant therapy. The Günther Tulip vena cava filter may be deployed by the femoral or jugular approach and can be used as a retrievable or permanent filter. Ideally, caval filters are placed below the renal veins. However, in cases of congenital anomalies of the IVC and its tributaries, alternative placement locations or additional filters must be considered to combat embolization through accompanying pathways. Venography is routinely performed prior to filterplacement. In addition to determining the level of the renal veins, cavography allows measurement of caval diameter and detection ofIVC and renal vein anomalies. Cavograms have revealed that in 15% of the cases there are abnormalities that significantly affect the placement of the filter.2 Clues to duplication of the IVC on cavography include lack of visualization of the left iliac inflow and larger-than-expected left renal vein inflow.
Caval interruption options in patients with duplicated IVC include single filter deployment in each IVC limb, single filter deployment in the unpaired suprarenal IVC, and filter placement in the right IVC with concurrent embolization of the left moiety or point of communication with the duplicated segment.3 In our case, we elected to deploy an additional filter in the left IVC.
Conclusion
Anatomic variations of the inferior vena cava and its tributaries are generally asymptomatic, but they must be recognized during vena cava filter placement because collateral pathways for emboli to bypass the filter may exist. Double IVC occurs in up to 3% of the population and necessitates a single suprarenal or paired caval filters.
- Anne N, Pallapothu R, Holmes R, Johnson MD. Inferior vena cava duplication and deep venous thrombosis: Case report and review of the literature. Ann Vasc Surg. 2005;19:740–743.
- Martin KD, Kempczinski RF, Fowl RJ. Are routine inferior vena cavograms necessary before Greenfield filter placement? Surgery. 1989;106:647–651.
- Vo NJ, Wieseler KW, Burdick TR, et al. The use of paired optionally retrievable Gunther Tulip filters in trauma patients with anatomical variants. Semin Intervent Radiol. 2007;24:20-28.
