Dyke-Davidoff-Masson Syndrome
Images
Case Summary
A young adult presented with a history of recurrent, generalized tonic-clonic seizures, developmental delay, and right-sided weakness. There were no complications at birth. At the age of 9 months, the patient had developed weakness of the right upper and lower extremities and subsequently demonstrated delayed milestones. They developed focal motor seizures with secondary generalization at a frequency of three per month despite being antiepileptic medication. On examination, there was left facial atrophy and right-body hemiatrophy. Scalp electroencephalogram was normal.
Imaging Findings
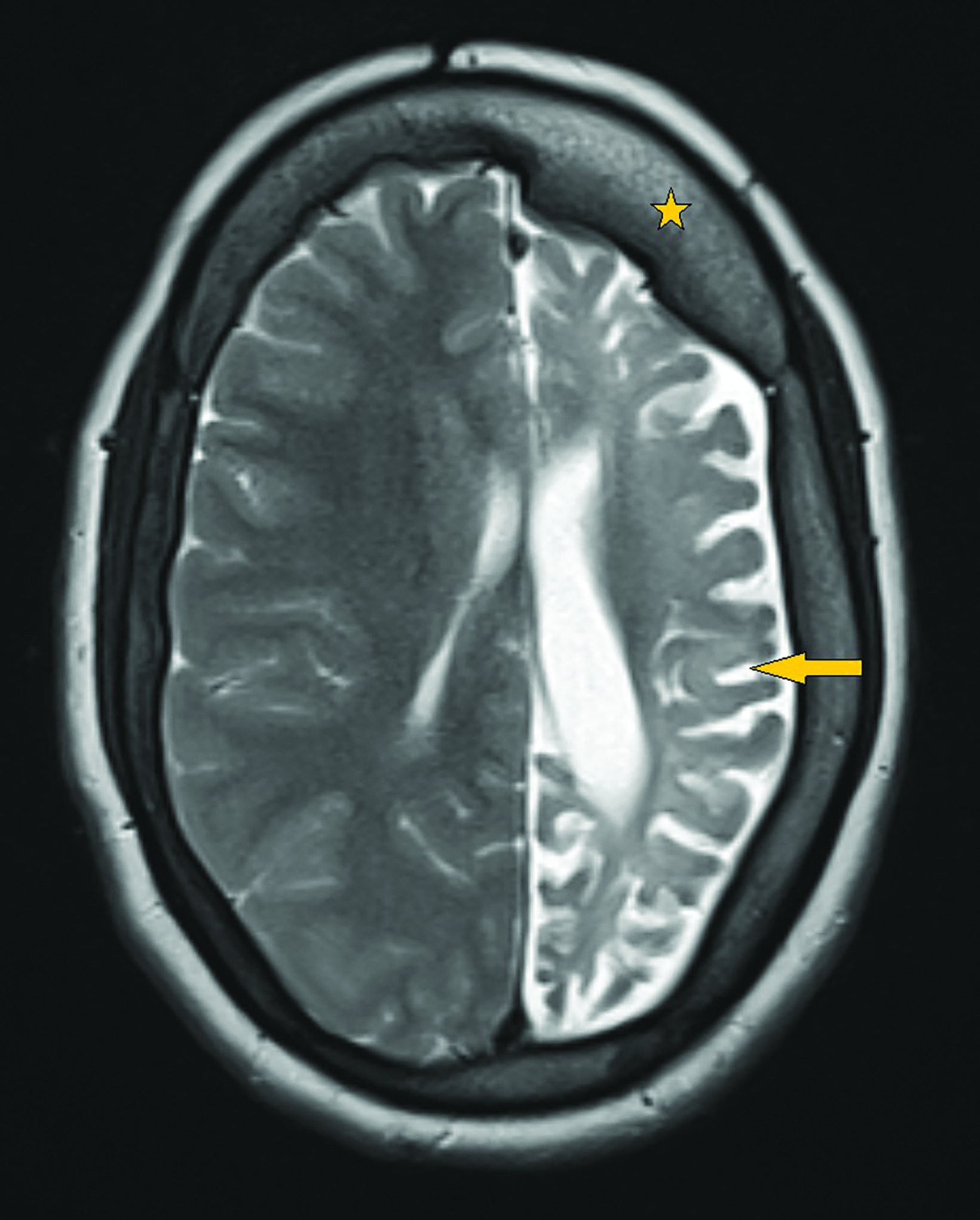
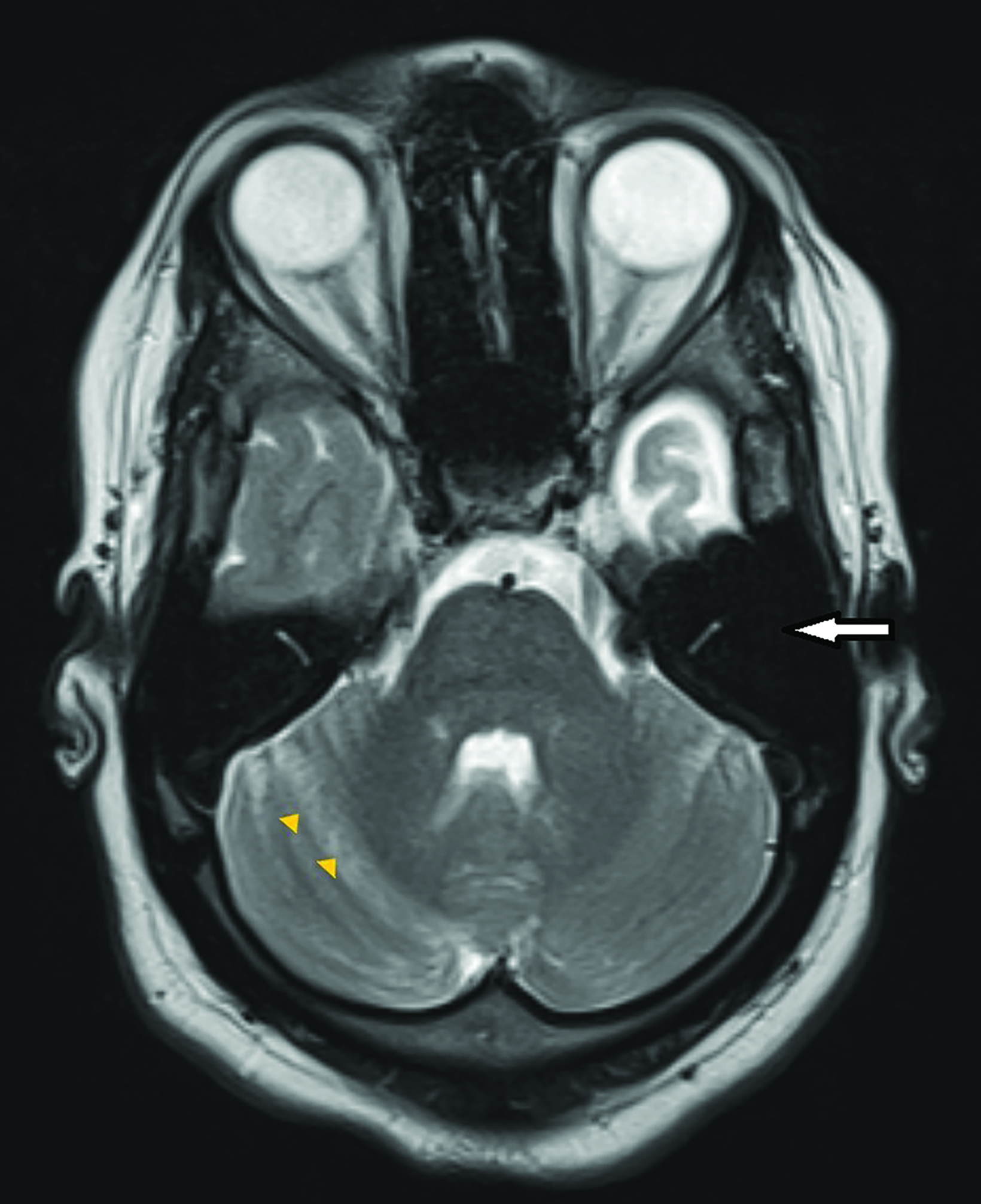
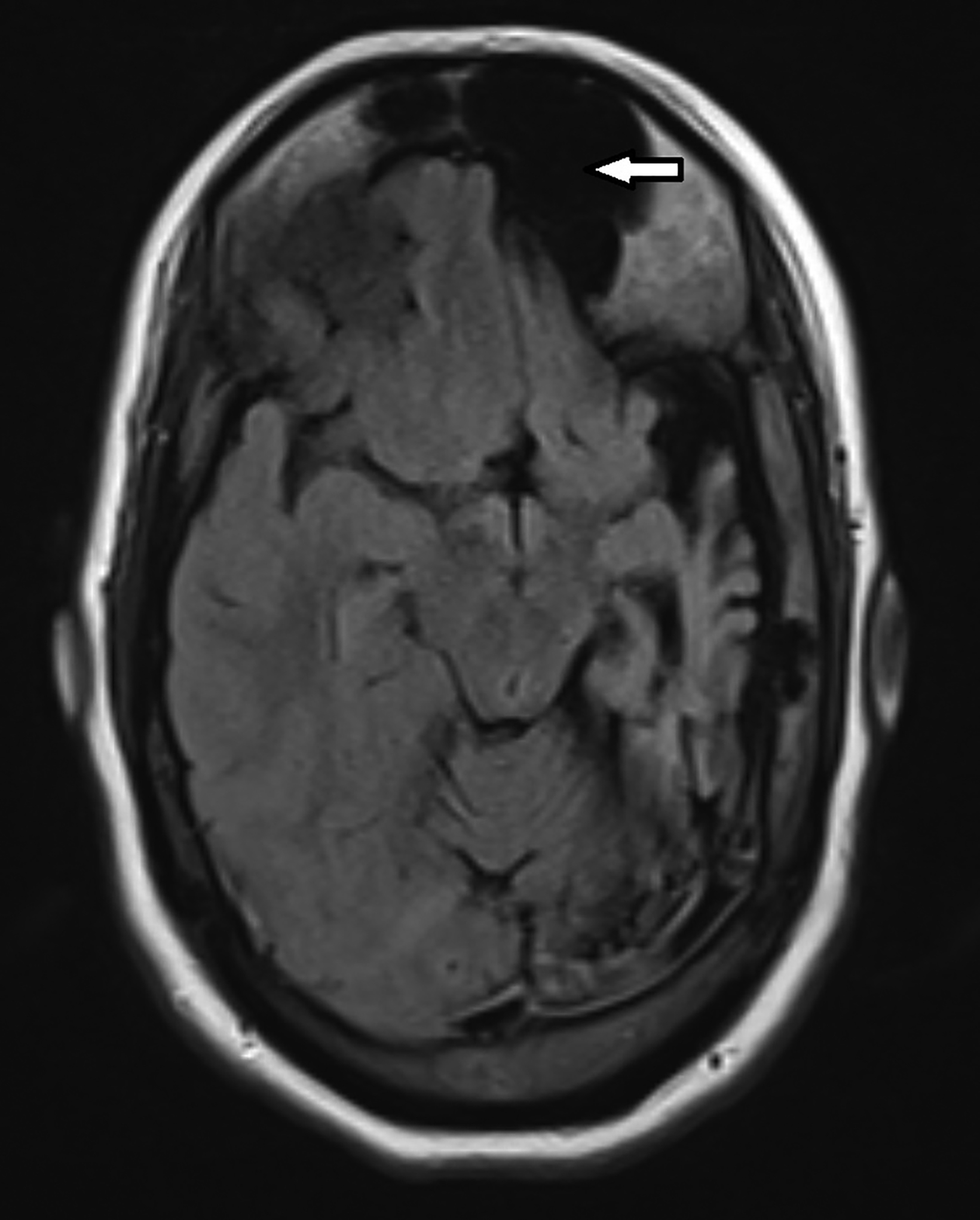
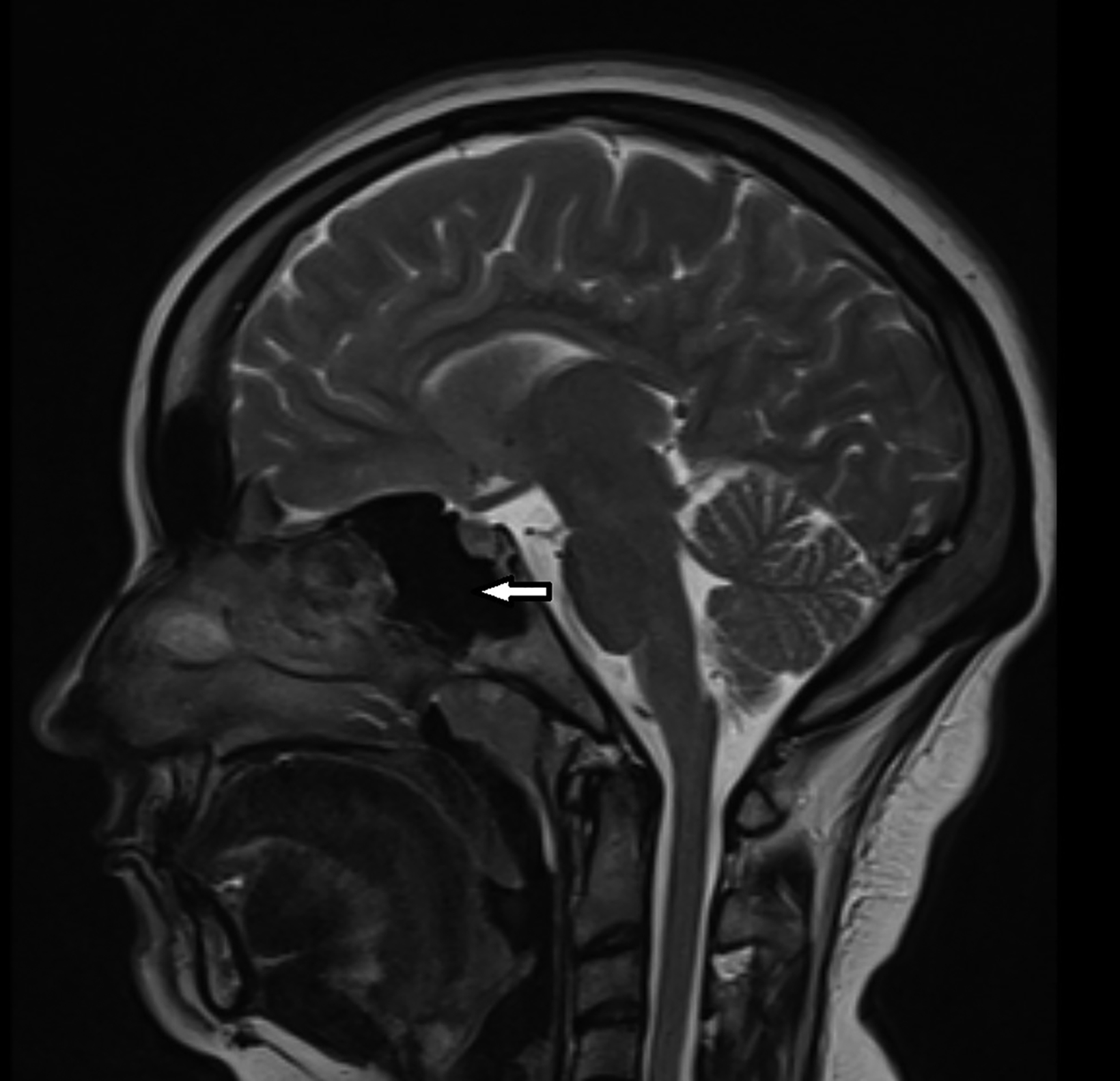
Contrast-enhanced magnetic resonance imaging (MRI) showed diffuse atrophy of the left cerebral hemisphere with prominent sulci and ventricles (Figures 1, 2). Minimal atrophy of the right cerebellar hemisphere was seen (Figures 1, 2). There was diffuse calvarial thickening (Figure 3) with hyper-pneumatization of the left frontal, ethmoid, and sphenoid sinuses. There was also hyperpneumatization of the ipsilateral mastoid air cells (Figures 1,2). Pertinent negatives included normal gyral thickness, no diffusion restriction, no blood products or mineralization, and no abnormal contrast enhancement.
Diagnosis
Dyke-Davidoff-Masson syndrome. Differential diagnoses include Rasmussen encephalitis and Sturge-Weber syndrome.
Discussion
Dyke, Davidoff, and Masson in 1933 described the uncommon condition hemicerebral atrophy in a series of nine patients with infantile hemiplegia. The radiographic findings were described on skull radiographs of these patients with seizures, facial asymmetry, hemiparesis, and developmental delay. They observed thickening of the skull and hyperpneumatization of the ipsilateral frontal sinuses, ethmoid air cells, and air cells in the petrous temporal bone. They postulated that these were compensatory changes owing to the loss of cerebral cortical volume from congenital or acquired brain insults.1
This syndrome refers to atrophy or hemiatrophy of a cerebral hemisphere secondary to a brain insult in the fetal or early childhood period. Clinically, patients can present with seizures, developmental delay, hemiparesis, and facial asymmetry. Involvement of the left side and male gender are more common in the literature.2 The incidence of the condition is unknown, as only a few cases are reported in the literature.
Cerebral hemiatrophy can be categorized into congenital and acquired etiologies.3 The former results from neonatal vascular insults or infection, with patients becoming symptomatic in the perinatal period or infancy. The latter can be a result of trauma, tumor, infection, ischemia, or hemorrhage.4
The classic imaging findings occur only in the event of a brain insult before 3 years of age. This is based on the postulation that normal development of the growing brain exerts outward pressure on the calvarium. If there is substantial brain atrophy, this outward pressure is decreased, resulting in calvarial thickening (increased width of diploic space) and enlargement of ipsilateral paranasal sinuses.5,6
The classic radiological features can be depicted both on computed tomography (CT) and MRI. These include cerebral hemiatrophy associated with dilatation of the ipsilateral lateral ventricle and prominent sulcal spaces. Rarely, thalamus and brainstem atrophy can be seen.7 Crossed cerebellar diaschisis may also be encountered. This is a condition in which the functional disconnection of the cerebellar hemisphere from the cerebral cortex results in cerebellar hypometabolism; interruption of the cortico-ponto-cerebellar pathway results in contralateral cerebellar atrophy.8
Associated thickening of the overlying skull with enlargement of the diploic space may be better appreciated on CT. Dilatation of the ipsilateral frontal, ethmoid, and sphenoid sinuses with elevation of the greater wing of the sphenoid bone and petrous ridge are also seen.9 Wallerian degeneration may be present.
The differential diagnoses include chronic Rasmussen encephalitis and Sturge-Weber syndrome. However, the former is a chronic, progressive disorder that does not demonstrate calvarial changes. The latter demonstrates enhancing pial angiomas and eventual calcifications in the atrophic cortex (“tram track”), with ipsilateral facial cutaneous capillary malformation (“port wine stain”).
Treatment primarily takes the form of seizure and behavioral controls. Functional hemispherectomy or hemispherotomy has demonstrated limited results with refractory seizures. Prognosis may be better in patients in whom hemiparesis appears after 2 years of age or in whom seizures are not recurrent.10
Conclusion
Dyke-Davidoff-Masson syndrome is a rare cause of refractory epilepsy with unknown etiology. The condition is diagnosed mainly through radiological evaluation, as clinical features are nonspecific. Treatment is largely supportive and aims at controlling seizures.
References
- Dyke CG, Davidoff LM, Masson CB. Cerebral hemiatrophy and homolateral hypertrophy of the skull and sinuses. Surg Gynecol Obstet. 1933;57:588-600.
- Kumar TS, Vipul A, Yadav R. Dyke-Davidoff-Masson Syndrome: a delayed diagnosis of an acquired variant. J Med Res and Innov. 2018;2(2):e000121.
- Sener RN, Jinkins JR. MR of craniocerebral hemiatrophy. Clin Imaging. 1992;16(2):93-97.
- Roy U, Panwar A, Mukherjee A, Biswas D. Adult presentation of Dyke-Davidoff-Masson syndrome: a case report. Case Rep Neurol. 2016;8(1):20-26.
- Sharma S, Goyal D, Negi A, Sood RG, Jhobta A, Surya M. Dyke-Davidoff Masson Syndrome. Indian J Radiol Imaging. 2006;16(2):165-166.
- Parker CE, Harris N, Mavalwala J. Dyke-Davidoff-Masson Syndrome: five case studies and deductions from dermatoglyphics. Clin Pediatr. 1972;11(5):288-292.
- Ayas ZÖ, Asil K, Öcal R. The clinico-radiological spectrum of Dyke-Davidoff-Masson syndrome in adults. Neurol Sci. 2017;38(10):1823-1828.
- Narra R, Kamaraju S, Pasupaleti B, Jukuri N. Dyke-Davidoff-Masson syndrome: A case report of cerebral hemiatrophy with crossed cerebellar diaschisis. Afr J Med Health. 2015;14(2):150.
- Arora R, Rani JY. Dyke-Davidoff-Masson syndrome: imaging features with illustration of two cases. Quant imaging Med Surg. 2015;5(3):469-471.
- Aneja J, Jangli S, Singh M, Mittal A. Acquired Dyke-Davidoff-Masson syndrome (DDMS). Int J Adv Med Health Res. 2015;2(1):55-58.
References
Citation
R A, D S, R D. Dyke-Davidoff-Masson Syndrome. Appl Radiol. 2022;(3):41-43.
April 26, 2022