Extraosseous multiple myeloma
Images

CASE SUMMARY
A 52-year-old woman presented with a 1-year complaint of unexplained/undesired weight loss of 13 lbs. Additional symptoms derived on review of systems included chills, night sweats, fatigue, and pain at the base of her tongue. Standard laboratory values demonstrated slight anemia and increase in serum creatinine. Subsequent biopsy of the base of tongue spot demonstrated amyloid deposition. Urine protein electrophoresis demonstrated elevated free lambda light chain, and bone-marrow biopsy demonstrated elevated levels of clonal plasma cells, consistent with diagnosis of multiple myeloma with associated amyloidosis.
The patient then underwent high-dose chemotherapy followed by allogenic stem cell transplantation with stabilization of the disease process. On follow-up examinations after initial therapy, the patient complained of new areas of nodularity involving the tongue, eyelids, and lower abdomen. The left lower abdomen was biopsied and pathologic results demonstrated myeloma/plasmacytoma. Additional involvement of the heart and GI tract was noted during the patient’s workup.
IMAGING FINDINGS
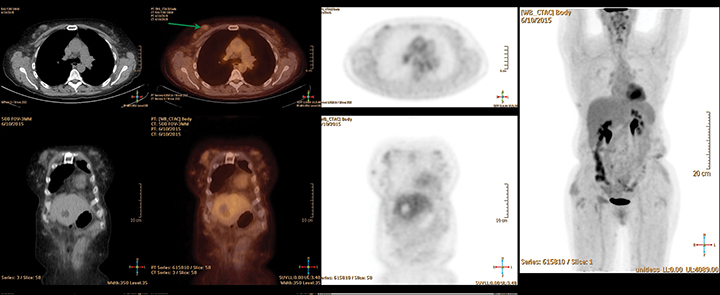
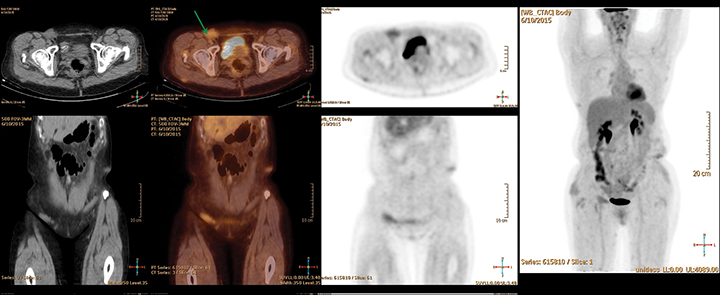
PET/CT images (Figures 1-3) demonstrated numerous hazy and nodular opacities within the subcutaneous tissues of the body wall involving the proximal upper extremities, anterior chest/breasts, anterior abdominal wall, and circumferential around the pelvis. The majority of these areas registered below background metabolism (compared to the average liver SUV), with some mildly hypermetabolic, most notably in the right inguinal region. The area of biopsy-proven myeloma/plasmacytoma was obtained from the left lower abdomen.
DIAGNOSIS
Estraosseous multiple myeloma. Differential considerations included a second primary malignancy, infectious process, extramedullary hematopoiesis, and in a different clinical scenario, the possibility of plasmacytoma should be considered.
DISCUSSION
Multiple myeloma may present with nonspecific clinical findings, including bone pain, fatigue, malaise, anemia, renal dysfunction, and increased susceptibility to infection. Advanced age (average age at diagnosis is 60 years) and abnormalities on standard labs may raise suspicion for this disease process.1 A diagnosis of symptomatic multiple myeloma requires clonal plasma cells >10% on biopsy (usually bone marrow), serum/urine monoclonal protein, and evidence of related end-organ damage.2 Differentiation from plasmacytoma (localized clonal plasma cell proliferation without bone marrow involvement) is important, as there is vast difference in prognosis and treatment.
Skeletal surveys are often performed as part of the standard workup; they can also be used to monitor disease progression and response to therapy. Findings can include lytic lesions, pathologic fractures, and osteopenia.3 Although not standard practice, additional imaging can include PET/CT or whole-body MRI, both of which are excellent in identifying contiguous spread of disease from osseous structures as well as extraosseous disease.4, 5 Contiguous spread of disease from osseous structures is more common than non-contiguous extraosseous spread, but this may be due to under-diagnosis, as extra-osseous involvement of myeloma was present in >63% in a post-mortem series.1,6
Examples of extraosseous spread of disease include involvement of the liver, spleen, lymph nodes, skin, lungs, and central nervous system.1 Determining the presence of extraosseous spread of disease is significant due to its effect on patient prognosis, with shorter progression-free survival and overall survival.7 The presence of extraosseous multiple myeloma has also been noted to be more common in more aggressive variants of the disease, which typically present at a mean age of 50.3, 1
Common imaging findings for extraosseous myeloma include homogenous soft tissue lesion with corresponding T2 hypointensity due to high cellularity. These lesions do not tend to calcify or demonstrate necrotic change, and will demonstrate mild hypermetabolism on FDG PET.3
CONCLUSION
Extraosseous involvement of multiple myeloma has increased in the last several decades, possibly due in part to improved imaging detection and increased patient survival. These increased rates are more pronounced in patients that undergo stem-cell transplant, likely as sanctuary sites.7 Although imaging characteristics for extraosseous myeloma are nonspecific, the presence of extraosseous soft-tissue masses in patients with a known history of multiple myeloma should raise suspicion for extraosseous disease. Tissue biopsy can help to confirm suspicions in equivocal cases. Diagnosing extraosseous metastases is of prognostic importance as their presence correlates with shorter progression-free survival and overall survival.7
REFERENCES
- Moulopoulos LA, Granfield CA, Dimopoulos MA, et al. Extraosseous multiple myeloma: imaging features. AJR Am J Roentgenol.1993;161 (5): 1083-1087.
- Kyle RA, Rajkumar SV. Criteria for diagnosis, staging, risk stratification and response assessment of multiple myeloma. Leukemia. 2009;23 (1): 3–9.
- Hall MN, Jagannathan JP, Ramaiya NH et-al. Imaging of extraosseous myeloma: CT, PET/CT, and MRI features. AJR Am J Roentgenol. 2010;195 (5): 1057-1065.
- Durie BGM. The role of anatomic and functional staging in myeloma: description of Durie/Salmon plus staging system. Eur J Cancer. 2006; 42:1539– 1543.
- Dimopoulos M, Terpos E, Comenzo RL, et al. International Myeloma Working Group. International Myeloma Working Group consensus statement and guidelines regarding the current role of imaging techniques in the diagnosis and monitoring of multiple myeloma. Leukemia. 2009; 23:1545–1556.
- Oshima K, Kanda Y, Nannya Y, et al. Clinical and pathologic findings in 52 consecutively autopsied cases with multiple myeloma. Am J Hematol. 2001; 67:1–5.
- Varettoni M, Corso A, Pica G, et al. Incidence, presenting features and outcome of extraosseous disease in multiple myeloma: a longitudinal study on 1003 consecutive patients. Ann Oncol. 2010; 21:325–330.
Citation
RZ T, IB D. Extraosseous multiple myeloma. Appl Radiol. 2018;(6):32-34.
June 7, 2018