Imaging in acute trauma-induced dissection and ischemic stroke: A Review
Images
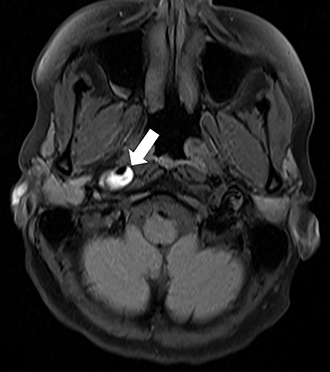
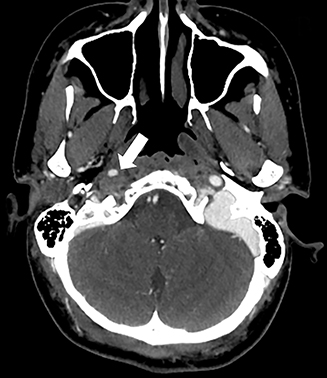
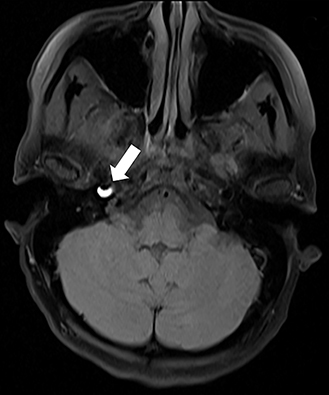
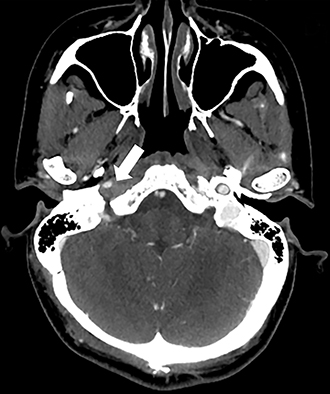
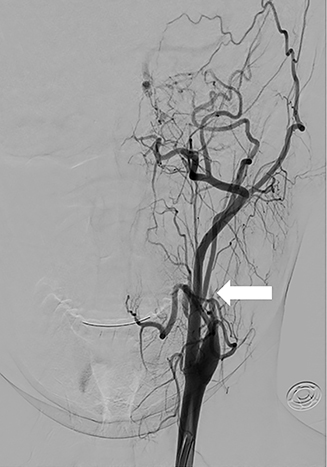

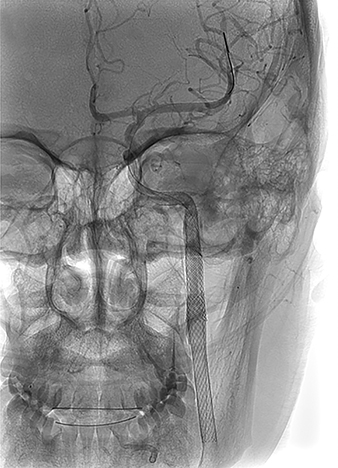
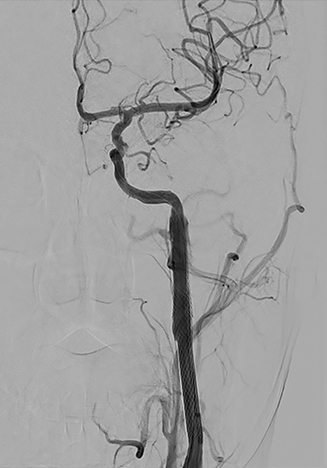
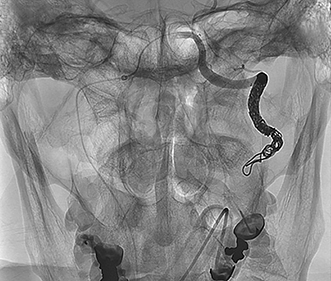


Cervicocerebral vascular trauma can lead to a spectrum of conditions, including dissection, laceration, pseudoaneurysm, occlusion, and arte riovenous fistula. These are relatively common events that can result in potentially devastating strokes. Typical areas of vascular injury are the distal cervical internal carotid artery (ICA), just below the skull base entry, and the V1 and V3 segments of the cervical vertebral artery. Early identification and characterization of these neurovascular injuries is important, as this will significantly improve patient outcome.1-4 In this review article, we provide an overview of the different imaging modalities used to diagnose cervicocerebral vascular injury/dissection. Further, we discuss the classification of dissection and related vascular injury, and review the current guidelines concerning treatment.
Pathology of acute dissection
A carotid or vertebral artery dissection starts of as a tear in the tunica intima or directly within the tunica media.5 Blood dissects along the artery to create an intramural hematoma, which will lead to thrombus formation. This thrombus can narrow the vessel lumen and become a nidus for distal embolization.6 The dissection plane can also lie between the tunica media and adventitia, resulting in expansion of the arterial wall and development of a false channel or lumen. This can become a source of distal emboli and can also result in mass effect on nearby structures such as lower cranial nerves and sympathetic fibers in the case of carotid dissection.5,6
Symptoms
Patients with carotid or vertebral artery dissection can have local and distal ischemic manifestations.5 In carotid dissection, local manifestations are pain in one side of the neck (25%), unilateral facial or orbital pain (50%), unilateral headache (65%), oculosympathetic palsy or Horner’s syndrome with miosis and ptosis (40%), cranial nerve palsy (10%) and pulsatile tinnitus (25%). Between 50% and 95% of patients have cerebral or retinal ischemic symptoms, with transient ischemic attacks (TIA) usually preceding ischemic stroke. Some 20% of patients have an ischemic stroke without any warning signs. In vertebral artery dissection, local manifestations are pain in the back of the neck (50%) and suboccipital headache (65%) that can be bilateral. Ischemic symptoms occur in more than 90% of patients, with TIA’s being much less frequent. The most common ischemic regions involved are the lateral medulla (Wallenberg’s syndrome), thalamus, and cerebral or cerebellar hemispheres. In rare cases, there can also be ischemia of the cervical spinal cord.
Imaging modalities
Ultrasound
Ultrasound is readily available but has limited value in the diagnosis of carotid artery dissection, as it is only possible to visualize the mid-cervical carotid segments. There is, however, potential value in the follow-up of limited dissection in selected settings.
Digital Subtraction Angiography (DSA)
Digital subtraction angiography has traditionally been considered the diagnostic gold standard, as subtle dissections can still sometimes be missed on high-resolution, non-invasive vascular imaging studies. Unfortunately, DSA is an invasive, costly, and time-consuming screening modality. There is also a small risk of serious complications. In our practice, we reserve DSA for selected cases that require better characterization of the dissection or dissection-related complications, and to help guide potential management. Signs to look for on DSA are vessel-wall irregularities, a thin channel of intravenous contrast material distal to a stenotic focus, described as the string sign,7 focal narrowing with a distal site of dilatation known as the string and pearl sign, and pseudoaneurysm formation.
MRI/MR Angiography (MRA)
A variety of MRI findings are associated with dissection. The best-known is a high-signal crescent sign within the vessel wall, which is best seen with T1 fat saturation (Figure 1) or T2-weighted sequences and represents acute thrombus. As the signal intensity of blood changes over time, this sign is only present in the acute phase of dissection. Another sign to look for is an absent flow-void, best seen on T2-weighted sequence. In the case of vessel-wall imaging with MRA, the same findings as previously described on DSA can often be seen. Finally, with diffusion weighted imaging (DWI), MRI can also detect evidence of cerebral ischemia as a consequence of a dissection.
Multidetector CT (MDCT)
Computed tomography is widely available and can be performed quickly and easily. Thus, it is preferred in the acute care setting.8 With multi-detector CT technology, it has become possible to perform rapid CTA evaluation of patients with suspected traumatic neurovascular injury.9 CTA can provide sufficient information to guide management decisions in the acute trauma patient. A normal artery has a consistently rounded cross-section, except on turns where it can appear oval, irregular, or slit-like. Deviations from a round cross-section are more easily detected on the axial (or short axis) images, while changes in caliber are often more obvious on the reconstructed images. The true extent and nature of the lesion is usually more easily perceived on the reconstructed MIP and MPR images. The signs to look for on CTA are expansion of the dissected artery, an abnormal vessel contour, narrowed eccentric lumen surrounded by a crescent-shaped mural thrombus, and thin annular enhancement which represents vasa vasorum enhancement in the adventitia, an intimal flap, and a dissecting aneurysm. Bullet fragments, other metallic penetrating objects, and dental amalgams can result in streak artifact that obscures portions of the vessels. Blood vessels surrounded by or adjacent to bone, such as the ICA at the skull base or the vertebral artery as it courses through the foramen transversarium, can be difficult to visualize. Bolus timing is sometimes imperfect and good opacification does not always equal good flow. Shahan et al found that 45% of screening CTA results were false positive, making the positive predictive value of CTA 55%.10 The use of CTA alone for blunt cerebrovascular injury diagnosis may result in overestimation of the frequency of dissection, especially if the findings are subtle.10 Confirmation with MRA or DSA may be appropriate prior to initiation of therapy.
Grading System
In 1999, Biffl et al.11 introduced a five-category grading scale describing the spectrum of blunt carotid injury, based on the angiographic appearance of lesions, which can also be extended to describe vertebral artery injury (Table 1). This grading system was not intended to apply to atherosclerotic narrowing, or to intracranial arterial dissection.
Complications
Grades 1-3
Thrombus may form on the injured intimal lining, or within a pseudoaneurysm, false lumen, or markedly narrowed true lumen. Distal thromboembolism occurs in a significant proportion of patients with traumatic cervical carotid or vertebral arterial dissections and pseudoaneurysms with consequent risk of stroke and death.
Occlusion
Occlusions are more common in patients with penetrating rather than blunt injury.12 Stroke occurring from hemodynamic compromise as a result of cervical arterial occlusion alone is less common than embolic complications with a lesser degree of injury.13
Transection
Patients with carotid or vertebral transection often have associated catastrophic injuries that prove lethal before reaching medical attention. Those who survive to reach a hospital have high risk of stroke or death despite aggressive surgical or endovascular management.14
Stroke
From 2001 to 2012, the incidence of traumatic carotid dissection increased 72% among Medicare beneficiaries.15 In a study performed in our center in 2006, we found that CTA screening increased the detected incidence of blunt vascular neck injury 8-fold, with rates similar to angiographically based screening protocols with associated significant decreases in morbidity and mortality.16 Medical treatment of blunt cerebrovascular injury is intended to prevent distal thromboembolism and stroke using systemic anticoagulation or antiplatelet therapy. The CADISS trial investigators found no significant difference in the efficacy of antiplatelet and anticoagulant drugs in the prevention of stroke and death in patients with symptomatic carotid and vertebral artery dissection.17 There is little evidence to support the routine stenting of cervical arterial dissections, although worrisome imaging characteristics such as severe flow-limiting stenosis, large pseudoaneurysm or increasing size of pseudoaneurysm, false lumen and intraluminal thrombus may lead to consideration of intervention especially if symptoms persist or recur on medical therapy.18 The CADISS trail investigators also found that 33% of grade 4 injuries recanalize and do so without sequelae. Seth et al published their experience in which they demonstrated that endovascular repair of traumatic cervical internal carotid artery injuries is a safe and effective treatment option.19 Zoarski found that unilateral endovascular occlusion of the cervical segment of the vertebral artery without antecedent balloon test occlusion can be safely performed without risk of a postprocedural infarct.20 Scott et al identified that 148 (97.4%) of 152 injuries as Grade 1 or 2 blunt VA injury were stable, improved, or resolved on final follow-up imaging evaluation. The use of ASA or other antiplatelet or anticoagulant medication in these low-grade blunt VA injuries did not correlate with radiological injury stability, nor with a decreased rate of cerebral infarction.21 The same group also found that the majority of high-grade blunt VA injuries remain stable or improved at final follow-up. Despite a 4% rate of radiographic worsening in the Grade 3 injuries and a 35% recanalization rate in Grade 4 injuries, there were no adverse clinical outcomes associated with these radiographic changes.22
Endovascular treatment
The goal of endovascular therapy, both in the acute and ongoing setting, is to prevent stroke resulting from thromboembolic and bleeding complications. Endovascular therapy consists of coil embolization of the vessel stump in arterial occlusions, reconstruction of a hemodynamically significantly narrowed artery with self-expanding or balloon-expandable stents (Figure 2), stenting of non-hemodynamically significant dissections with highly irregular intimal flaps, primary coiling of pseudoaneurysms, and stent and coil embolization of pseudoaneurysms. Covered stent-grafts for treatment of lacerations and pseudoaneurysms of the extracranial internal carotid artery caused by penetrating injury are strongly preferred over non-covered stents.23 The vertebral artery is difficult to approach surgically and is usually treated by endovascular occlusion of the artery.24 Unilateral vertebral artery occlusion seldom results in neurologic deficits if the contralateral vertebral artery is not injured, is of adequate size, and the posterior inferior cerebellar artery (PICA) blood supply on the side of occlusion is preserved (Figure 3). However, it is always important to assess the location of important radiculo-medullary arteries, such as the artery of the cervical enlargement (artery of Lazorthes). Vertebral artery sacrifice should never be undertaken until the location of the anterior spinal artery (ASA) has been considered. For example, vertebral artery occlusion immediately distal to the radiculomedullary ASA contribution risks the possibility of thrombosis of the vertebral artery and compromise of this ASA segment.
Conclusion
Dissection of the extracranial carotid and vertebral arteries is a common cause of stroke in young patients. Diagnosis is most commonly made with noninvasive vascular imaging, including MRA or CTA. Most ischemic cerebral symptoms arise from thromboembolic mechanisms and are adequately treated with anticoagulation or antiplatelet medications. It is important to document healing of the dissection on follow up imaging studies before cessation of antithrombotic therapy. The uncommon patient presenting with acute hemodynamic insufficiency should be managed with measures to increase cerebral blood flow such as keeping the head of the bed flat, volume expansion, and induced hypertension. In this setting, emergency stent placement to restore cerebral perfusion may be considered, provided that irreversible infarction has not already occurred. Other endovascular techniques may be considered for management of expanding pseudoaneurysms and persistent thromboembolic events despite appropriate medical therapy.
References
- Cothren CC, Moore EE, Biffl WL, et al. Anticoagulation is the gold standard therapy for blunt carotid injuries to reduce stroke rate. Arch Surg. 2004;139(5):540-545.
- Biffl WL, Moore EE, Ryu RK, et al. The unrecognized epidemic of blunt carotid arterial injuries: early diagnosis improves neurologic outcome. Ann Surg. 1998;228(4):462-470.
- Biffl WL, Moore EE, Elliott JP, et al. The devastating potential of blunt vertebral arterial injuries. Ann Surg. 2000;231(5):672-681.
- Fabian TC, Patton JH, Croce MA, et al. Blunt carotid injury. Importance of early diagnosis and anticoagulant therapy. Ann Surg. 1996;223(5):513-522.
- Schievink WI. Spontaneous dissection of the carotid and vertebral arteries. N Engl J Med. 2001; 344(12):898-906.
- Redekop GJ. Extracranial carotid and vertebral artery dissection: a review. Can J Neurol Sci. 2008; 35(2):146-152.
- Pappas JN. The angiographic string sign. Radiology. 2002; 222(1):237-238.
- Zimmerman RA, Bilaniuk LT, Gennarelli T, et al. Cranial computed tomography in diagnosis and management of acute head trauma. AJR Am J Roentgenol. 1978;131(1):27-34.
- Munera F, Soto JA, Nunez D. Penetrating injuries of the neck and the increasing role of CTA. Emerg Radiol. 2004;10(6):303-309.
- Shahan CP, Magnotti LJ, Stickley SM, et al. A safe and effective management strategy for blunt cerebrovascular injury: Avoiding unnecessary anticoagulation and eliminating stroke. J Trauma Acute Care Surg. 2016;80(6):915-922.
- Biffl WL, Moore EE, Offner PJ, et al. Blunt carotid arterial injuries: implications of a new grading scale. J Trauma. 1999;47(5):845-853.
- Ramadan F, Rutledge R, Oller D, et al. Carotid artery trauma: a review of contemporary trauma center experiences. J Vasc Surg. 1995;21(1):46-55.
- Lucas C, Moulin T, Deplanque D, et al. Stroke patterns of the internal carotid artery dissection in 40 patients. Stroke. 1998;29(12):2646-2648.
- duToit DF, van Schalkwyk GD, Wadee SA, et al. Neurologic outcome after penetrating extracranial arterial trauma. Ann Vasc Surg. 2003; 38:257-262.
- Newhall K, Gottlieb DJ, Stone DH, et al. Trends in the diagnosis and outcomes of traumatic carotid and vertebral artery dissections among medicare beneficiaries. Ann Vasc Surg. 2016;36:145-152.
- Schniedereit N, Simons R, Nicoloau S, et al. Utility of screening for blunt vascular neck injuries with computed tomography angiography. J Trauma. 2006;60(1):209-215.
- CADISS trial investigators, Markus HS, Hayter E, et al. Antiplatelet treatment compared with anticoagulation treatment for cervical artery dissection (CADISS): a randomised trial. Lancet Neurol. 2015;14(4):361-376.
- Redekop G, Marotta T, Weill A. Treatment of traumatic aneurysms and arteriovenous fistulas of the skull base by using endovascular stents. J Neurosurg. 2001;95(3):412–419.
- Seth R, Obuchowski AM, Zoarski GH. Endovascular repair of traumatic cervical internal carotid artery injuries: a safe and effective treatment option. AJNR Am J Neuroradiol. 2013;34(6):1219-1226.
- Zoarski GH, Seth R. Safety of unilateral endovascular occlusion of the cervical segment of the vertebral artery without antecedent balloon test occlusion. AJNR Am J Neuroradiol. 2014; 35:856-861.
- Scott WW, Sharp S, Figueroa SA, et al. Clinical and radiological outcomes following traumatic Grade 1 and 2 vertebral artery injuries: a 10-year retrospective analysis from a Level 1 trauma center. J Neurosurg. 2014;121(2):450-456.
- Scott WW, Sharp S, Figueroa SA, et al. Clinical and radiological outcomes following traumatic Grade 3 and 4 vertebral artery injuries: a 10-year retrospective analysis from a Level I trauma center. The Parkland Carotid and Vertebral Artery Injury Survey. J Neurosurg. 2015; 122(5):1202-1207.
- Redekop G, Marotta T, Weill A. Treatment of traumatic aneurysms and arteriovenous fistulas of the skull base by using endovascular stents. J Neurosurg. 2001;95(3):41-419.
- Higashida RT, Halbach W, Tsai FY, et al. Interventional neurovascular treatment of traumatic carotid and vertebral artery lesions: results in 234 cases. AJR Am J Roentgenol. 1989;153(3):577-582.
Citation
D V, MKS H, G R. Imaging in acute trauma-induced dissection and ischemic stroke: A Review. Appl Radiol. 2019;(2):15-19.
March 28, 2019