Imaging in Stroke Diagnosis and Treatment: An Update
Images

The prevalence of stroke is steadily rising and annual stroke-related deaths (6.55 million) have increased by 43.3% since 1990, according to the global burden of diseases study 2019 data.1 Ischemic stroke contributed to 62.4% of the reported cases and remains a leading cause of morbidity and the second-most common cause of mortality.1
Management of ischemic stroke within the initial 4.5 to 6 hours of presentation may salvage the area of insufficiently perfused brain tissue that is still viable but “at risk” of ischemic injury if not treated. Research has been rapidly evolving with newer approaches to prevention and treatment.2,3 Despite medicine’s best efforts, preventative therapy may not be enough for some patients. Therefore, a prompt diagnosis with possible emergent treatment of an acute infarct becomes imperative to mitigate permanent neurological deficit and even death.
Cases amenable to endovascular treatment (EVT) require activation of the multidisciplinary stroke service, with rapid patient assessment, imaging, and treatment. The phrase “Time is Brain” forms the essence of acute ischemic stroke management and emphasizes the importance of early clinical recognition, establishing a diagnosis, and assessment severity based on radiological imaging and treatment. Through extensive studies on the multitude of patients who have suffered acute infarcts, guidelines and timelines have been developed to identify patients who will benefit most by medical therapy, or EVT, and a preventative strategy for future possible stroke.
It has been reported that for every 30-minute delay in treatment after the onset of an acute infarct, the likelihood of a favorable outcome decreases by approximately 11%.4 Acute ischemic strokes secondary to proximal large-vessel occlusion (LVO) carries a higher risk of morbidity and mortality.4 The importance of emergent and reliable imaging for the diagnosis of LVO cannot be overstated. We aim to discuss the diagnosis and treatment of acute ischemic stroke in a series of two review articles. In this first article, we review various imaging modalities that are utilized to diagnose acute ischemic stroke. The second part, appearing in the November/December 2021 issue of Applied Radiology, will focus on treatment.
Noncontrast Computed Tomography
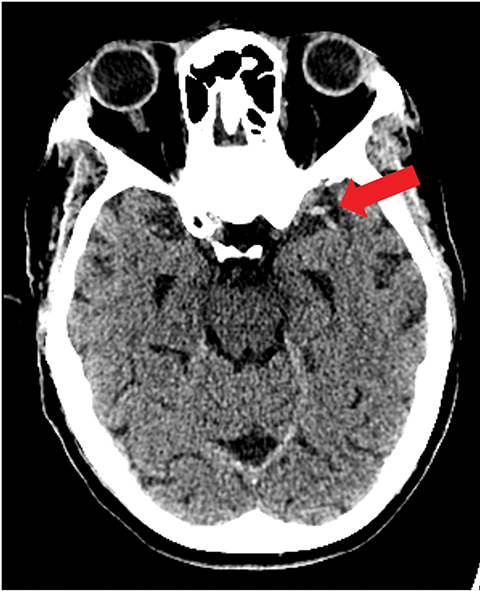
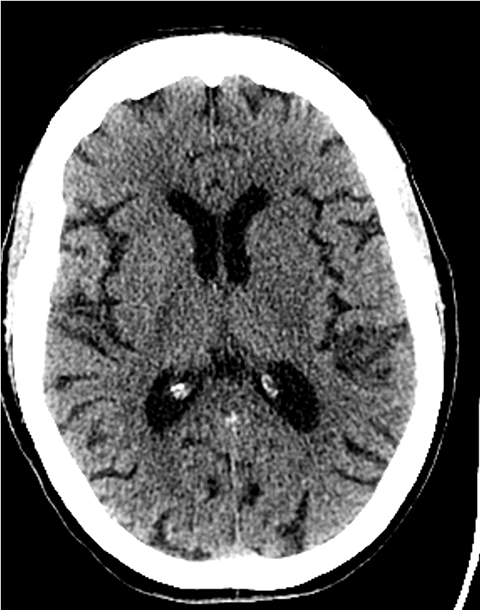
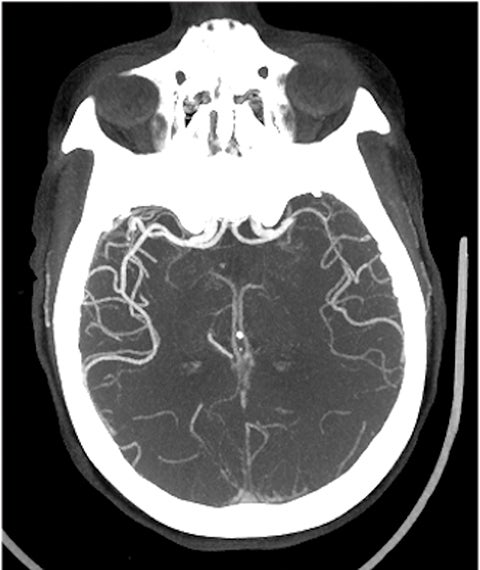
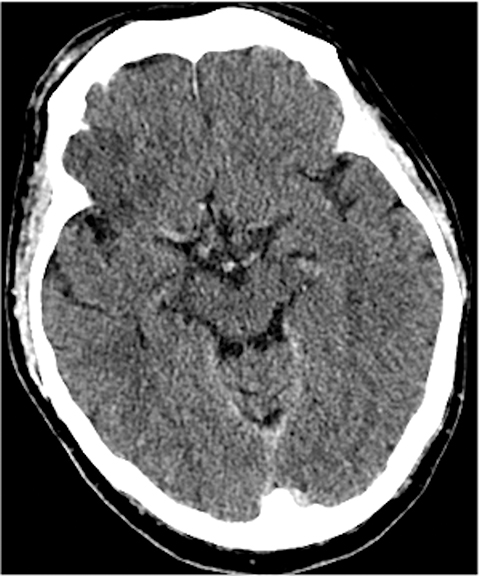
A noncontrast computed tomography (CT) head is the first step in the imaging workup of acute ischemic stroke, as it is widely available and can rapidly rule out a hemorrhagic stroke, which would be treated differently. With minimal risk of radiation, CT is indispensable to providing essential information regarding the size of an infarct, the affected territory, core infarct volume, and collateral supply (Figure 1). It is highly valuable in the setting of patients with implanted cardiac defibrillators or implants, where MRI is contraindicated.
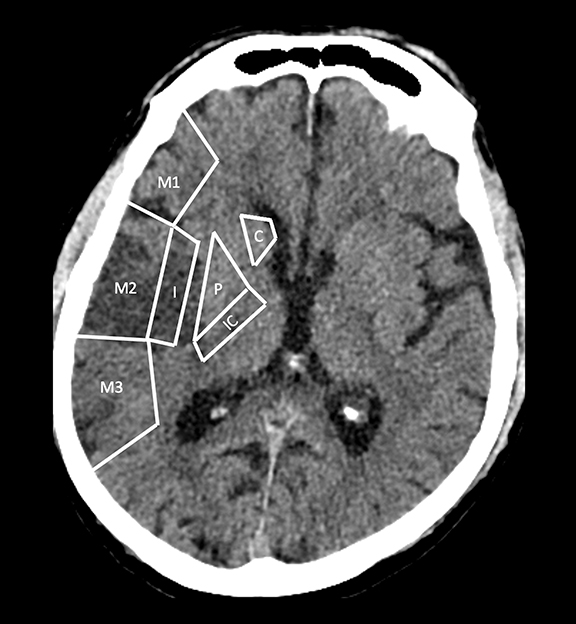
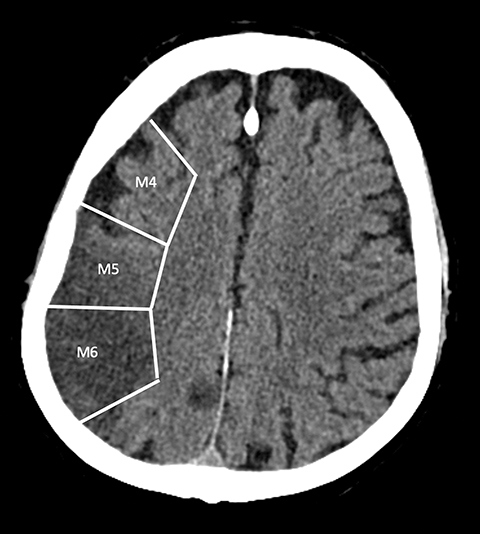
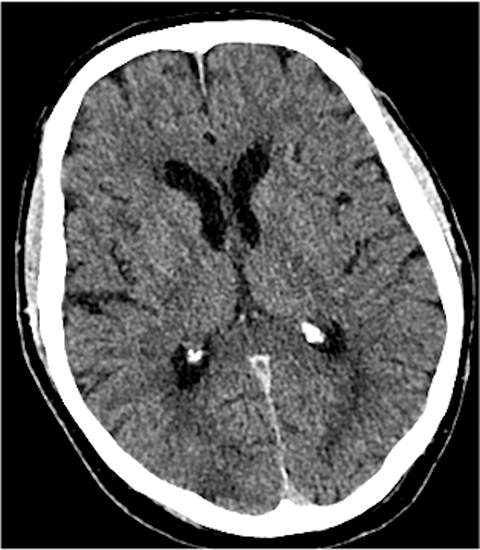
With regard to middle cerebral artery (MCA) territory occlusions/infarcts, the most commonly affected territory, the Alberta stroke program early CT score (ASPECTS) allows for an effective, semi-objective measure of the degree of infarct.5 ASPECTS is a 10-point scoring system and one point is deducted for each of ten predefined areas (caudate, putamen, internal capsule, insular cortex and six areas in MCA territory). Therefore, the system provides a simple and rapid measure of stroke burden that has been validated to prognosticate about the feasibility and likely outcome of reperfusion (Figure 2). ASPECTS 6 or more is the established cut-off criterion as per American Heart Association (AHA) guidelines to consider stroke patients for endovascular intervention.6
ASPECTS was conventionally considered applicable only to the anterior circulation.7 However, in a recently published study, Caruso, et al, have demonstrated the feasibility and validity of the posterior circulation Alberta stroke program early CT score (Pc-ASPECTS) in 50 patients with posterior circulation strokes. Pc-ASPECT MTT was found to be the most reliable parameter in predicting clinical outcome.8
ASPECTS scoring has certain limitations. These include modest interobserver variability owing to potential subjective bias in interpretation, unequal contribution of the included regions towards clinical outcomes, and difficulty in reproducibility.9,10 Recent advancements include the use of automated software to evaluate ASPECTS rapidly and potentially eliminate the interobserver variability.11 When combined with perfusion imaging using an intravascular tracer, a noncontrast CT head may yield information to help detect salvageable brain parenchyma in patients with LVO.12
CT Angiography
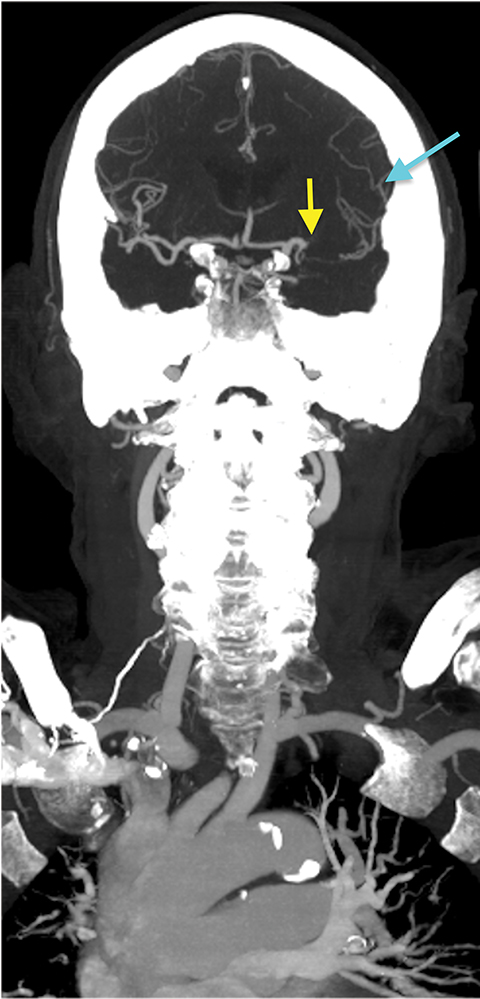
Requiring a rapid, well-timed intravenous administration of contrast material, CT angiography (CTA) is an effective method for imaging cerebrovascular anatomy. CTA is recognized to be a highly sensitive (92-100%) and specific (82-100%) method for detecting LVO.13 Additionally, the modality allows assessment of the aortic arch anatomy, the branching of large vessels, and the presence of calcifications, atherosclerotic plaques, and/or stenoses — an assessment that is crucial to identifying patients for endovascular intervention (Figure 3). CT angiography also serves an important role in the identification of other conditions that may present with clinical features similar to those of stroke.
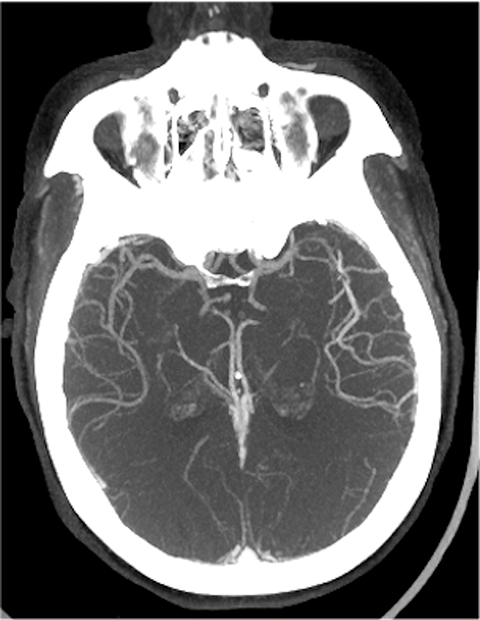
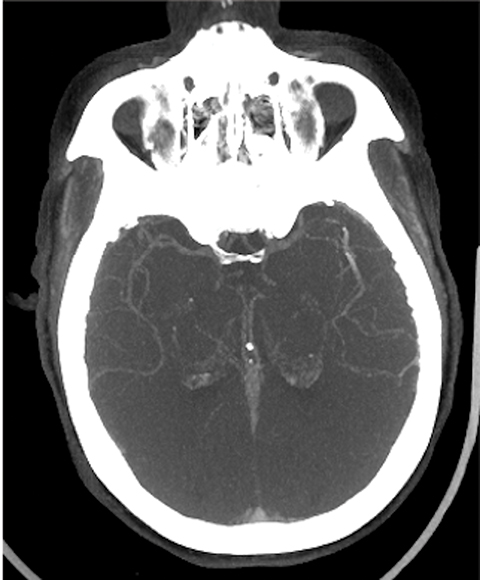
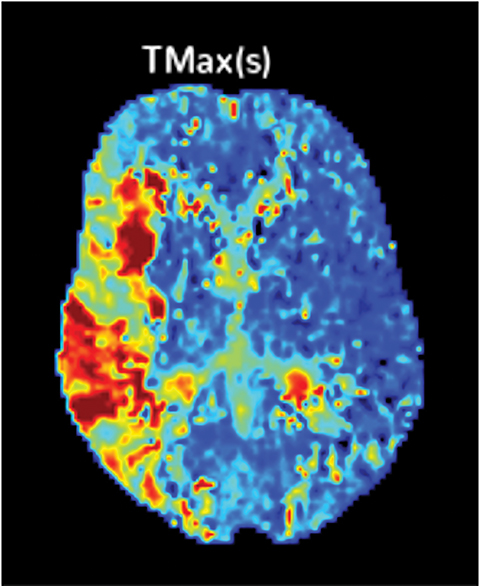
The cerebral circulation may have multiple subsidiary vascular channels, called “collateral circulation,” which serve as a backup supply to the brain parenchyma in occlusions of the primary arterial supply. Collateral circulation is recognized as an independent predictor of a good outcome in ischemic stroke (Figure 3).14 Multiphase CTA helps to identify the collateral supply and may prove highly useful regarding treatment decisions (Figure 4).
The ischemic core of the infarct---the irreversibly damaged brain tissue---and surrounding, potentially salvageable brain parenchyma or “penumbra” in the affected vascular territory may be identified by hypodensity or absence of contrast opacification as compared to normally perfused regions of parenchyma. Therefore, CTA can indirectly help estimate perfusion in a vascular territory. A study by Camargo, et al, demonstrated a higher sensitivity with CTA in comparison with noncontrast CT for detecting ischemic changes and predicting final infarct volume.15
Mismatch Imaging
The concept of mismatch imaging relies on differentiating between the infarct core and the surrounding penumbra based on their imaging characteristics. Identifying a large penumbra may help to salvage it with timely intervention, subsequently reducing the final volume of infarct.
The standard treatment for acute ischemic stroke within 6 hours of presentation is mechanical thrombectomy. However, thrombectomy and revascularization may still be beneficial in patients beyond this time window, especially if the ischemic penumbra is large.16 Both CT and MRI can be used to assess this mismatch.16
In acute cerebral ischemia, cell edema occurs first, followed by angioedema. Diffusion weighted imaging (DWI) is the most sensitive noninvasive method for detecting cytotoxic edema associated with ischemic injury.16 A fluid-attenuated inversion-recovery (FLAIR) sequence can detect vasogenic edema caused by cerebral ischemia and hypoxia in tissue that is not completely beyond viability. The amount of FLAIR hyperintensity beyond areas of reduced diffusivity is called the DWI-FLAIR mismatch, and it is used to quantify the extent of penumbra. DWI-FLAIR mismatch plays an important role in treatment decisions for patients who present within 6 to 24 hours of onset and for those with “wake-up strokes,” where the timing of onset is unknown.16
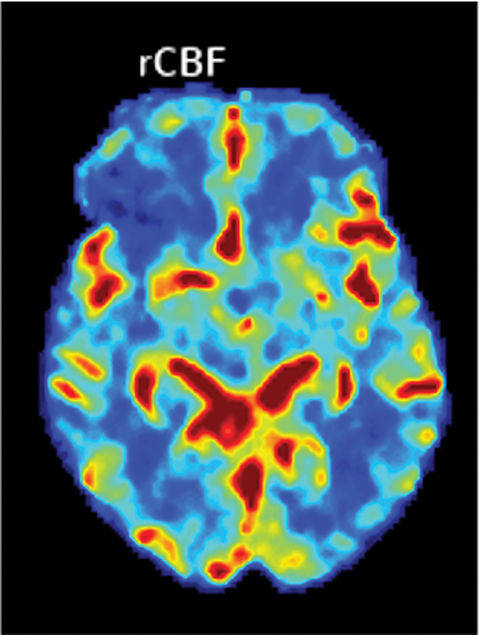
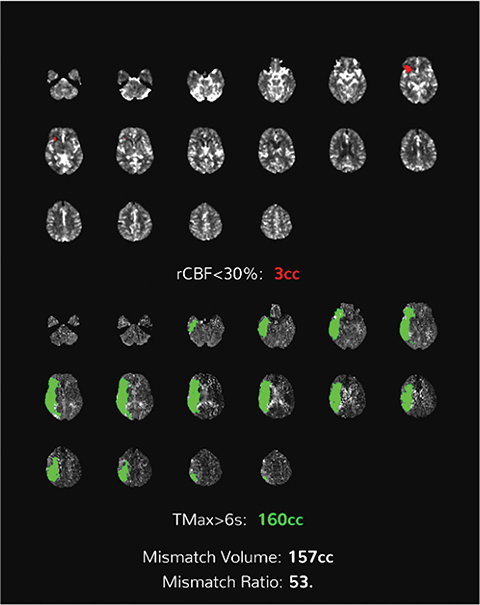
Perfusion-based quantitative assessments can also provide valuable information, including such data as cerebral blood flow (CBF), defined as the flow rate of blood within a region of brain. Cerebral blood flow drops in cases of compromised arterial supply.17 The time to perfuse a territory with compromised arterial supply is higher than that of normally perfused tissue. This phenomenon can be quantified by the time-to-peak (TTP), a measurement of time required for a given region of brain to reach maximal attenuation.17
In viable but ischemic tissue, cerebral blood volume (CBV), the total amount of blood within a given region of the brain, may be normal or slightly increased as a result of autoregulatory vasodilation. However, in irreversibly injured tissue, CBV is low owing to loss of autoregulation.17 Mean transit time (MTT) is the length of time a certain volume of blood spends in the capillary phase. Mean transit time can be calculated by dividing the cerebral blood volume by cerebral blood flow.
Both MTT and TTP can be used to determine whether a significant amount of salvageable tissue remains after an ischemic event. The TTP threshold of 6 seconds is commonly used to define hypoperfused territory, which includes salvageable penumbra and ischemic core. The core is commonly defined as a decrease in CBF <30% of the contralateral territory (Figure 5).17
One study that compared the predictive clinical outcomes from CT ASPECTS and CT perfusion-based quantitative assessment of the core volume in 508 patients with acute, emergent, large-vessel occlusion did not demonstrate any statistical difference between the two methods.7
Dual-layer Spectral CT
An advanced imaging technique, dual-layer spectral CT (DLSCT) is increasingly being utilized by leading stroke centers worldwide. The technique is based on an advanced detector system that can capture the photons of low and high energy simultaneously without requiring a change in the X-ray tube parameters.18
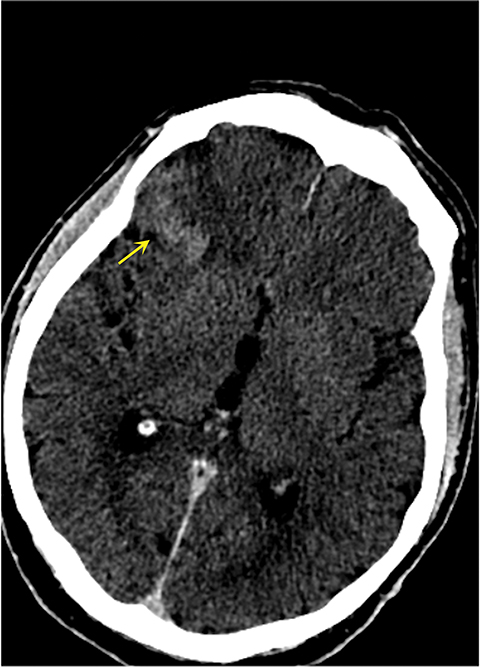
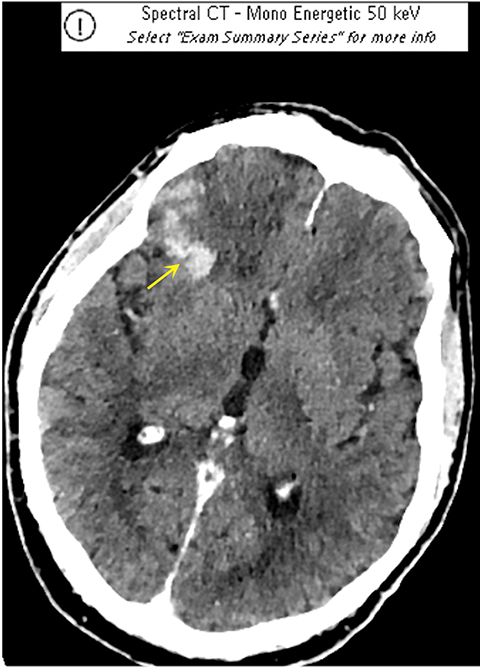
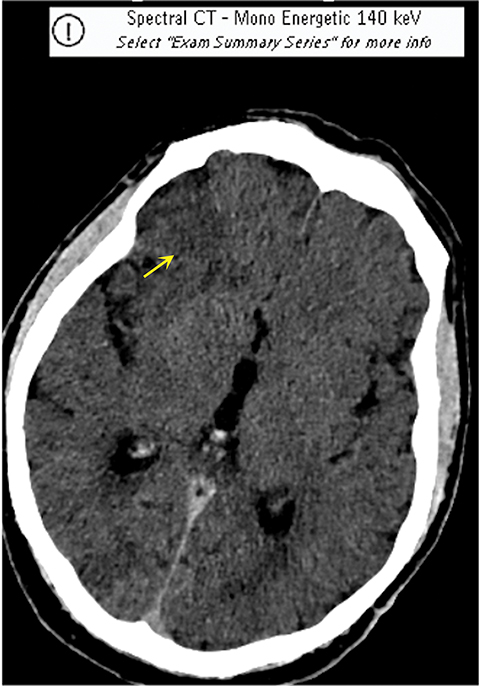
It has been well established that parenchymal hyperdensity may be observed on post-thrombectomy noncontrast CT scans as result of iodinated contrast injection during the procedure. Therefore, differentiating this hyperdensity from hemorrhage is highly pertinent (Figure 6). The dual-layer detection system in DLSCT can differentiate between hemorrhage and extravasation of iodinated contrast based upon differential K-edge, which proves highly useful in acute stroke management, especially after mechanical thrombectomy.18-20
Such information is crucial in deciding whether to use anticoagulants and antiplatelet agents after mechanical thrombectomy and in stroke patients receiving iodinated contrast prior to transfer to a thrombectomy-capable center. A recently published study by Riederer, et al, demonstrated 100% sensitivity and specificity for DLSCT in the detection of blood.19
New Technologies for Analysis
New telemedicine platforms permit more efficient workups to be performed even before patients present in the hospital.21 With advances in personal mobile device hardware, radiological imaging can be viewed remotely in almost any situation.21
Artificial intelligence (AI) and machine learning (ML) software tools are increasingly being used for stroke diagnosis. For instance, Viz LVO (Viz.ai, San Francisco California) uses an algorithm to detect LVOs on CT angiography. The software is able to alert stroke specialists within minutes of image completion, a crucial component when “Time is Brain.”21
In summary, rapid, effective, and reliable medical imaging plays a crucial role in the diagnosis and treatment of acute ischemic stroke. Applied on a case-by-case basis, imaging modalities such as MRI and CT provide for effective approaches to acute stroke management.
References
- Roth GA, Mensah GA, Johnson CO, et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: Update from the GBD 2019 Study. J Am Coll Cardiol. 2020; 76(25) 2982–3021.
- Zerna C, Hegedus J, Hill MD. Evolving treatments for acute ischemic stroke. Circ Res. 2016;118(9):1425-1442. doi:10.1161/CIRCRESAHA.116.307005
- Serulle Y, Khatri D, Sy H, Yerneni S, Langer D, Ortiz R. Use of quantitative magnetic resonance angiography in patients with symptomatic intracranial arterial stenosis who undergo stenting: presentation of three cases. J Cerebrovasc Endovasc Neurosurg. 2021;23(2):136-144. doi:10.7461/jcen.2021.E2020.10.005
- Waqas M, Vakharia K, Munich SA, et al. Initial emergency room triage of acute ischemic stroke. Neurosurgery. 2019;85(suppl_1):S38-S46. doi:10.1093/neuros/nyz067
- Pexman JH, Barber PA, Hill MD, et al. Use of the Alberta Stroke Program Early CT Score (ASPECTS) for assessing CT scans in patients with acute stroke. AJNR Am J Neuroradiol. 2001;22(8):1534-1542.
- Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association [published correction appears in Stroke. 2019 Dec;50(12):e440-e441]. Stroke. 2019;50(12):e344-e418. doi:10.1161/STR.0000000000000211
- Raza SA, Barreira CM, Rodrigues GM, et al. Prognostic importance of CT ASPECTS and CT perfusion measures of infarction in anterior emergent large vessel occlusions. J Neurointerv Surg. 2019;11(7):670-674. doi:10.1136/neurintsurg-2018-014461
- Caruso P, Ridolfi M, Lugnan C, et al. Multimodal CT pc-ASPECTS in infratentorial stroke: diagnostic and prognostic value [published online ahead of print, 2021 Feb 16]. Neurol Sci. 2021;10.1007/s10072-021-05072-x. doi:10.1007/s10072-021-05072-x
- Shin DH, Shin DJ, Kim JR. Do all ASPECT Score regions have the same predictive power for functional outcomes? J Stroke Cerebrovasc Dis. 2020;29(2):104516. doi:10.1016/j.jstrokecerebrovasdis.2019.104516
- Schröder J, Thomalla G. A critical review of Alberta Stroke Program Early CT Score for evaluation of acute stroke imaging. Front Neurol. 2017;7:245. Published 2017 Jan 12. doi:10.3389/fneur.2016.00245
- Sundaram VK, Goldstein J, Wheelwright D, et al. Automated ASPECTS in acute ischemic stroke: A comparative analysis with CT perfusion. AJNR Am J Neuroradiol. 2019;40(12):2033-2038. doi:10.3174/ajnr.A6303
- Wang T, Chen L, Jin X, et al. CT perfusion-based ASPECTS improves the diagnostic performance of early ischemic changes in large vessel occlusion. BMC Med Imaging. 2021;21(1):67. Published 2021 Apr 12. doi:10.1186/s12880-021-00593-5
- Latchaw RE, Alberts MJ, Lev MH, et al. Recommendations for imaging of acute ischemic stroke: a scientific statement from the American Heart Association. Stroke. 2009;40(11):3646-3678. doi:10.1161/STROKEAHA.108.192616
- Goyal M, Demchuk AM, Menon BK, et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med. 2015;372(11):1019-1030. doi:10.1056/NEJMoa1414905
- Camargo EC, Furie KL, Singhal AB, et al. Acute brain infarct: detection and delineation with CT angiographic source images versus nonenhanced CT scans. Radiology. 2007;244(2):541-548. doi:10.1148/radiol.2442061028
- Cao S, Dong H. Predictive value of DWI-FLAIR Mismatch in patients with ischemic stroke and receiving endovascular treatment beyond time window. Pak J Med Sci. 2021;37(2):466-471. doi:10.12669/pjms.37.2.3293
- Bonney PA, Walcott BP, Singh P, Nguyen PL, Sanossian N, Mack WJ. The continued role and value of imaging for acute ischemic stroke. Neurosurgery. 2019;85(suppl 1):S23-S30. doi:10.1093/neuros/nyz068
- Bernsen MLE, Veendrick PB, Martens JM, Pijl MEJ, Hofmeijer J, van Gorp MJ. Initial experience with dual-layer detector spectral CT for diagnosis of blood or contrast after endovascular treatment for ischemic stroke. Neuroradiology. 2021 May 27. doi: 10.1007/s00234-021-02736-5. Epub ahead of print. PMID: 34046731.
- Riederer I, Fingerle AA, Zimmer C, Noël PB, Makowski MR, Pfeiffer D. Potential of dual-layer spectral CT for the differentiation between hemorrhage and iodinated contrast medium in the brain after endovascular treatment of ischemic stroke patients [published online ahead of print, 2021 Apr 29]. Clin Imaging. 2021;79:158-164. doi:10.1016/j.clinimag.2021.04.020
- Gulko E, Ali S, Gomes W, Gandhi C, Mehta H. Differentiation of hemorrhage from contrast enhancement using dual-layer spectral CT in patients transferred for acute stroke. Clin Imaging. 2021;69:75-78. doi:10.1016/j.clinimag.2020.06.046
- Yaeger KA, Shoirah H, Kellner CP, Fifi J, Mocco J. Emerging Technologies in Optimizing Pre-Intervention Workflow for Acute Stroke. Neurosurgery. 2019;85(suppl_1):S9-S17. doi:10.1093/neuros/nyz058
This article is accredited for one SA-CME credit. Visit appliedradiology.org/SAM2 for full SA-CME information.
Editor’s note: This is the first of a two-part series. The second part will appear in the November-December 2021 issue of Applied Radiology.
Related Articles
References
Citation
S B, R H, R Z, A B, J H, AL B, D K. Imaging in Stroke Diagnosis and Treatment: An Update. Appl Radiol. 2021; (5):8-15.
September 10, 2021