Imaging the obstructed bowel and other intestinal emergencies





Dr. Kottler is a Postgraduate Year 5 Resident, Dr. Aguirre is a Research Assistant, Dr. Casola is the Vice Chair of Education, Chief of Body Imaging and Professor of Clinical Radiology, and Dr. Sirlin is an Assistant Clinical Professor, Department of Radiology, University of California, San Diego, CA.
Acute abdominal pain due to gastrointestinal (GI) tract pathology is one of the most frequent causes of patient presentation to the emergency room. Rapid diagnosis is essential to expedite appropriate management. In recent years, computed tomography (CT) has emerged as the most accurate noninvasive imaging tool in this setting. Distinct advantages of CT are its ability to directly visualize bowel wall and surrounding structures. This review discusses the CT evaluation of bowel obstruction and other nontraumatic, emergent GI tract pathologies. Imaging strategies, CT findings, differential diagnosis, and identification of potential complications are stressed.
Bowel obstruction
Bowel obstruction is a common, potentially devastating cause of acute abdominal pain, which occurs in approximately 20% of patients with an acute abdomen. 1 The frequency is even higher in patients with prior abdominal surgery. Important definitions are listed in Table 1. Imaging plays an important role in diagnosis and management.
Imaging strategies and techniques
To our knowledge, a formal cost-benefit analysis delineating the optimal imaging workup for patients with suspected bowel obstruction has not been performed. The imaging algorithm proposed here is based on the authors' anecdotal experience (Figure 1).
We believe that CT is the study of choice for suspected small-bowel obstruction (SBO). Although it may seem reasonable to begin with plain radiographs rather than with CT, CT has much higher accuracy for: 1) the diagnosis of SBO, 2) the recognition of its cause and potential complications, and 3) the identification of other entities with similar clinical presentations. 2,3 Thus, if clinical suspicion warrants, CT should be performed regardless of plain radiographic results. CT is particularly valuable in patients without prior laparotomy, as these patients are unlikely to have adhesions and may harbor an occult malignancy. CT is also invaluable in patients with suspected strangulation, known malignancy, or palpable abdomino-pelvic mass.
To help assess the bowel wall, intravenous (IV) contrast should be administered, unless its use is contraindicated. Enteric contrast is useful if symptoms are mild, as these patients may have partial obstructions or other causes for their pain. 4 We believe that enteric contrast should be avoided in patients with a more fulminant presentation and obvious intestinal distention. 4 In these patients, intraluminal fluid and gas provide effective negative contrast. Moreover, enteric contrast may unnecessarily delay CT or interfere with mural assessment.
In suspected large-bowel obstruction (LBO), a single preliminary radiograph of the abdomen may be helpful. 2 If the radiograph shows marked colonic dilatation, a fluoroscopic enema study, rather than a CT, should be considered, as the CT may be difficult to interpret in this setting. Patients with modest or no colonic dilatation should undergo CT. Rectal contrast may be necessary if the colon is tortuous.
Patients with suspected low-grade or intermittent SBO in whom CT is nondiagnostic may benefit from conventional or CT enteroclysis.
Small-bowel obstruction
Small-bowel obstruction is at least 3 times more common than LBO. 2 Luminal narrowing is usually severe; because small-bowel contents are liquid, mild or moderate narrowing may be insufficient to produce symptomatic obstruction. Small-bowel obstruction is usually caused by extrinsic rather than intrinsic factors, and benign causes are more common than malignant ones (Table 2, Figures 2 through 7). 1,2
Prompt diagnosis and management are necessary, as untreated SBO may be complicated by fluid shifts, electrolyte abnormalities, and strangulation. The clinical examination is usually nonspecific. Thus, imaging plays a crucial role. Imaging goals are summarized in Table 3.
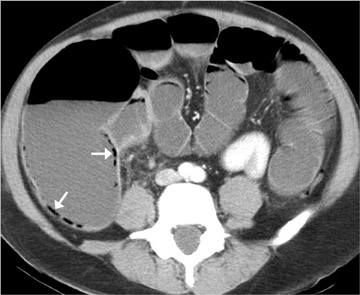
CT criteria for diagnosis of SBO are listed in Table 4. 1,2 The transition point itself is often not visualized, particularly if the obstruction is caused by adhesions. Some authorities recommend multiplanar reformatting to identify the transition point, but the diagnostic value of reformation has not been proven. An important ancillary finding is the "small-bowel feces" sign (Figures 2 and 3): Fecal-like material in the segment of small bowel just proximal to the obstruction. This sign suggests a relatively long-standing obstruction and should be differentiated from meconium equivalent in patients with cystic fibrosis (history and evaluation of the pancreas may be useful). In cases of adynamic ileus, both the transition point and small-bowel feces are absent, and the colon is dilated.
Table 5 lists CT findings that differentiate low- and high-grade obstructions. Findings favoring high-grade obstruction include total collapse of the small bowel and colon distal to the obstruction, marked dilatation proximal to the obstruction, and triangular fluid collections within the adjacent mesentery (Figure 4). Identification of high-grade obstruction is important because aggressive management is usually necessary.
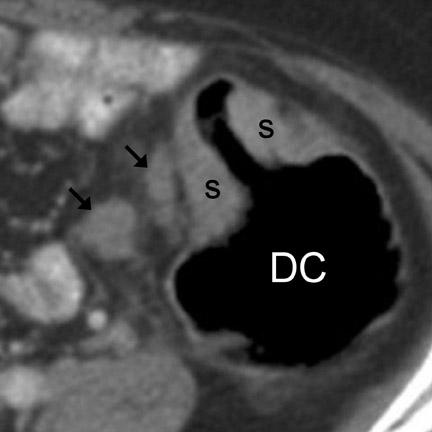
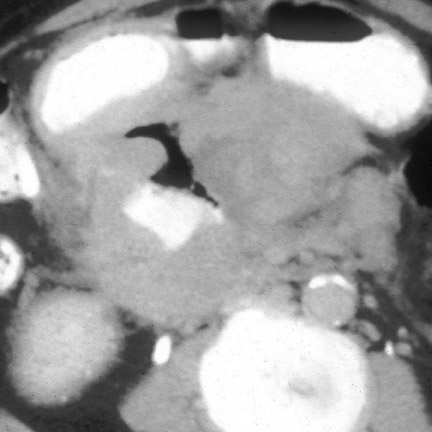
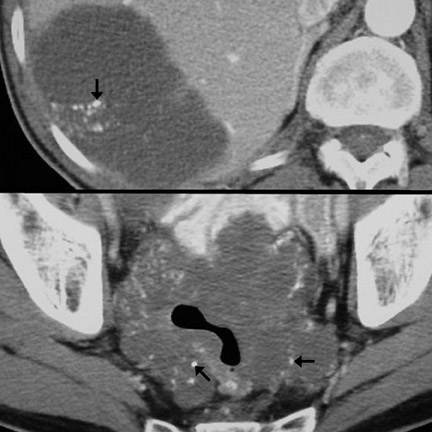
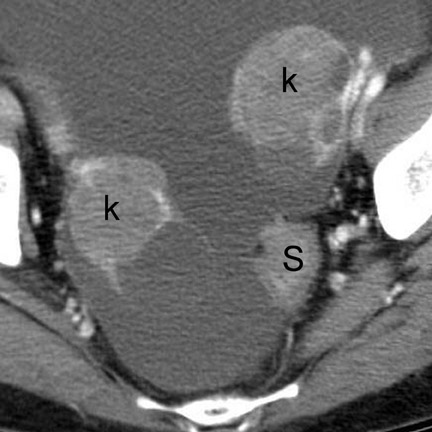
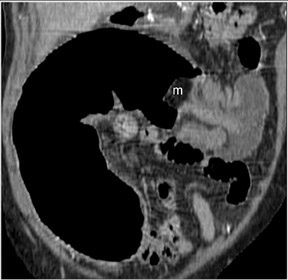
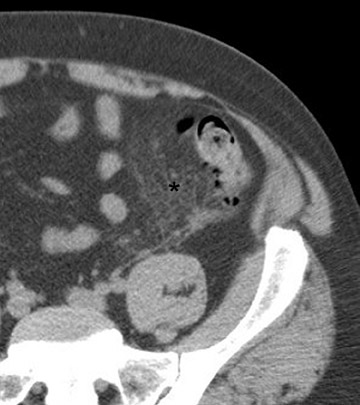
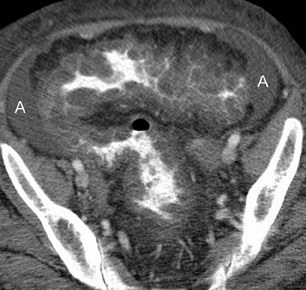
CT findings that suggest closed-loop obstruction are summarized in Table 6. 1-7 The appearance of the closed loop depends on its orientation and length. If horizontally oriented, the closed segment typically manifests as "C"-, "U"-, or comma-shaped loops. If vertically oriented, it appears as radially distributed loops around a central point (Figures 5 and 6). When long, a closed segment may appear as a cluster of multiple fluid-filled loops. Dilated mesenteric vessels (>3 mm diameter) characteristically converge to the point of obstruction or twist. The entry and exit loops of bowel may lie adjacent to one another and have a smoothly tapered, triangular, or beaked appearance (beak sign) (Figure 5). The closed loop is usually fluid-filled and gasless. The degree of dilatation and quantity of intraluminal fluid may be conspicuously disproportionate to that of more proximal segments; in the authors' anecdotal experience, this disproportion can be a helpful clue to the diagnosis. Closed-loop obstructions are usually treated as surgical emergencies because of the high risk of strangulation.
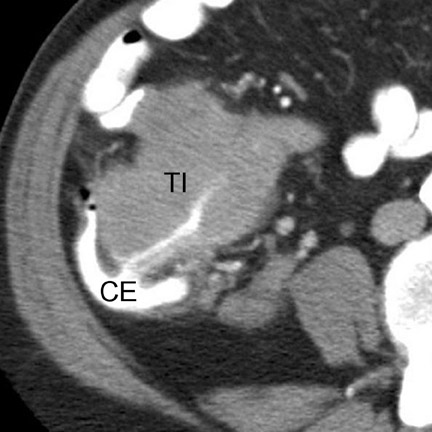
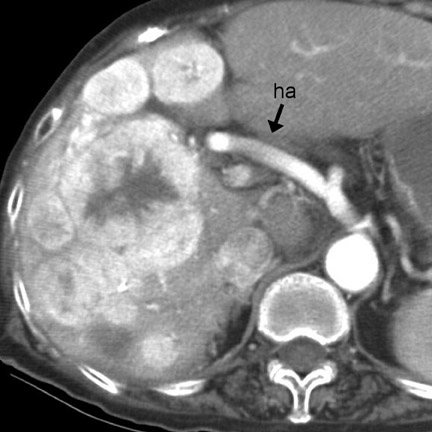
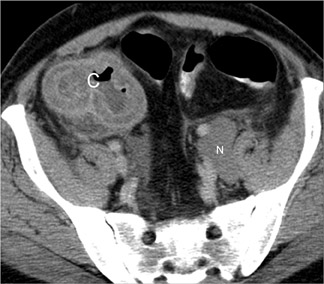
Strangulation can occur with simple or closed-loop obstruction but is more frequent in the latter. 2 CT findings suggesting strangulation can be classified as mural or extramural (Table 7). 1,2,4,7,8 The mural target or halo sign (indicating submucosal edema) is best seen with IV contrast (Figure 7), while mural hemorrhage (Figure 6) is best seen without IV contrast. Focal or segmental loss of mural enhancement, which some investigators believe is the most specific sign of strangulation (Figure 5), 2,8 suggests impaired arterial inflow. Persistent mural enhancement suggests impaired venous outflow. Pneumatosis and portomesenteric gas suggest advanced bowel ischemia. 4 Pneumatosis may be difficult to identify if luminal contents are heterogeneous. If pneumatosis is equivocal, identification of portomesenteric gas can be extremely helpful.
Large-bowel obstruction
Large-bowel obstruction is less common than SBO. Because large-bowel contents are solid or semi-solid, mild to moderate luminal narrowing may cause symptomatic obstruction. In contrast to SBO, LBO is usually secondary to intrinsic rather than extrinsic factors and malignant causes are more common than benign ones (Table 2, Figures 8 through 11). 9
Partly because of the different causes and partly because they produce fewer fluid and electrolyte abnormalities, LBOs tend to present more insidiously than do SBOs. The major risk of LBO is perforation. Perforation tends to occur at the site of greatest dilatation, usually the cecum. 1,2 In cases of malignant obstruction, the perforation may occur at or adjacent to the cancer site rather than in the cecum.
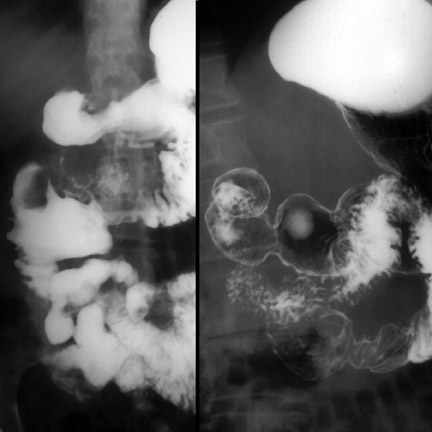
Whereas CT is the imaging study of choice for SBO, plain radiographs and enema studies continue to play an important role in the evaluation of LBO (Figure 8). 2 Radiographic findings depend on the site and cause of obstruction and the competency of the ileocecal valve. If the ileocecal valve is competent, the colon, and particularly the cecum, may markedly dilate without significant small-bowel distention. 2 If incompetent, diffuse gaseous distention of both the small and large bowel results, making differentiation from adynamic ileus difficult. Distal LBOs may also mimic adynamic ileus. In this case, films with the patient in the right-side-down decubitus or prone positions may be helpful; gas entering the rectum and rectosigmoid after repositioning excludes obstruction, while nondistention of the rectum suggests obstruction. Cecal volvulus classically manifests as a kidney-shaped mass in the left upper quadrant or epigastrium 10 and sigmoid volvulus as an inverted "U" arising vertically or obliquely from the pelvis or as three curved lines converging toward the pelvis. 10
The CT technique for LBO is identical to that for SBO except that rectal contrast or air insufflation may be necessary. Rectal contrast should not be given, however, if colonic inflammation is severe, or if peritoneal signs or perforation is present.
Other entities
Other causes of acute, nontraumatic GI pain include appendicitis, diverticulitis, ischemia, inflammatory bowel disease (IBD), and infection. Each of these topics is complex, and thorough reviews have been published. 11-14 Salient features are discussed below.
Appendicitis
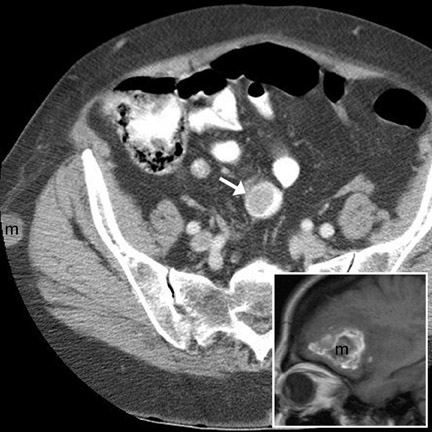
The optimal CT imaging technique for appendicitis is controversial. We obtain 5-mm slices without oral or IV contrast on a multidetector scanner from the liver dome (to visualize the lung bases) to the symphysis pubis and then retrospectively reconstruct overlapping 2.5-mm slices, if necessary. If the findings are ambiguous or if the appendix is not visualized (about 25% of cases), we give IV contrast and rescan the lower quadrants (Figure 12). In the setting of prolonged symptoms (eg, >72 hours), pre-existing bowel disease, pregnancy, or young patient age, both IV and oral contrast are administered initially. Other institutions routinely give IV and/or enteric contrast in all cases. 3,15-17
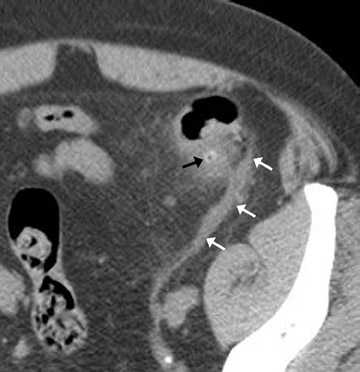
The CT findings of appendicitis are summarized in Table 8 4 and include primary and secondary signs. Although most authors recommend a 6mm outer-to-outer wall threshold measurement for appendiceal dilatation, up to 42% of appendices 6- to 10-mm in diameter are normal. 18 Thus, in the absence of other findings, a diameter of 6 to 10 mm does not clinch the diagnosis. Periappendiceal fat stranding is usually, but not invariably, present and may be absent in mild or early appendicitis. In our anecdotal experience, thickening of the right lateral conal fascia may be obvious even if fat stranding is subtle. Thus, we feel that thickening of the lateral conal fascia is a helpful finding. In equivocal cases, we recommend close clinical observation and repeat imaging.
Complications of appendicitis occur more frequently with prolonged symptoms and include perforation, peritonitis, abscess, and sinus tract/fistula formation.
Diverticulitis
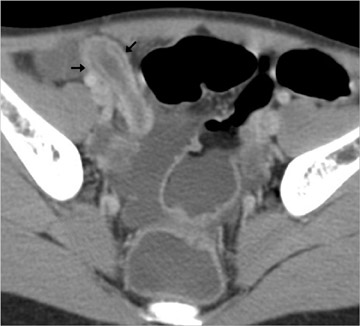
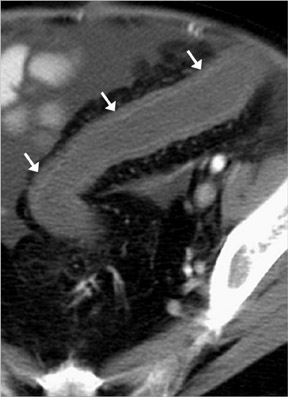
Diverticulitis occurs when a diverticulum becomes obstructed and focally inflamed and then perforates. The perforation is typically contained and causes a local inflammatory process that is predominantly extracolonic or "peridiverticular." The greatest concentration of diverticula is in the distal descending andsigmoid colon; hence, most cases (95%) of diverticulitis are left-sided (Figure 13) 4 ; a minority of cases (5%) are right-sided. 14 For unknown reasons, right-sided diverticulitis has a predilection for patients of Asian descent. 14 Diverticulitis of the small bowel is uncommon.
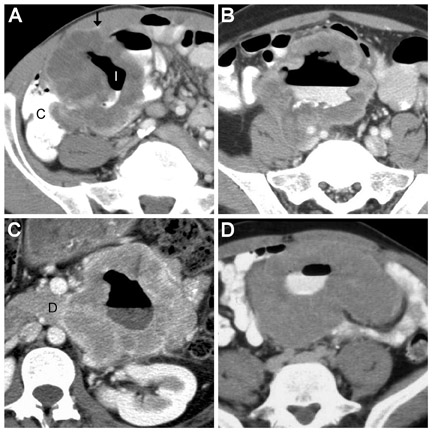
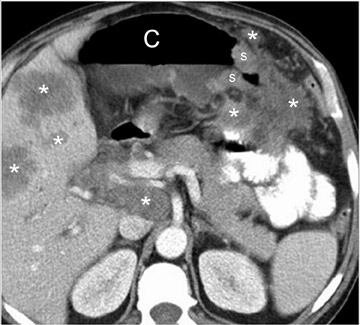
CT criteria for the diagnosis of diverticulitis are listed in Table 9. 4,13,14,19 The most common and usually striking finding is pericolonic fat stranding. Fat stranding is characteristically disproportionate to the degree of wall thickening, 20 which tends to be mild (4 to 5 mm). Disproportionate fat stranding is an important finding, as it may help suggest the diagnosis in atypical presentations. At least one diverticulum is visualized in >80% of cases of diverticulitis 19 ; definitive CT diagnosis of diverticulitis may be difficult if diverticula are not identified. The comma sign refers to comma-shaped thickening of the root of the sigmoid mesocolon. The centipede sign refers to a multitude of engorged, parallel mesenteric vessels supplying the affected segment of bowel. Based on anecdotal experience, we also rely on an unpublished "fuzzy diverticulum" sign (Figure 13A). This sign refers to an ill-defined diverticulum, larger than other visible diverticula, centered where the fat stranding is most pronounced. This "fuzzy diverticulum" presumably represents the causative diverticulum.
The most important differential consideration for left-sided diverticulitis is adenocarcinoma, as it affects a similar demographic group (in whom incidental diverticula may be present) and causes relatively focal colonic involvement. Differentiation is aided by ancillary findings (Table 10). 3,9,21 In particular, colonic thickening disproportionate to pericolonic fat stranding and the presence of regional nodes suggest malignancy. However, clinical and CTfindings may overlap (Figure 10). 3 If findings are equivocal and tissue sampling is not planned, a follow-up examination should be performed after resolution of acute manifestations (usually approximately 4 weeks). Other differential considerations include appendicitis, omental infarction, and epiploic appendagitis. 20
Complications of diverticulitis include intra- and extramural abscesses, sinus tract and fistula formation (especially colovesical fistula), bowel and ureteral obstruction, gross intraperitoneal or retroperitoneal perforation, peritonitis, septic thrombosis of portomesenteric veins, and liver abscess. Gross perforation with free gas in the peritoneal or retroperitoneal spaces usually requires immediate laparotomy. Contained perforations with extramural abscesses >3 cm are usually treated by drainage followed by surgery; abscesses <3 cm typically resolve with conservative management.
Bowel ischemiaBowel ischemia can be divided into occlusive and nonocclusive causes. Occlusive disease refers to obstruction of large arteries (embolus, thrombus, trauma, compression, or infiltration) or veins (thrombus). Nonocclusive ischemia is due to low-flow states, small-vessel disease, splanchnic vasoconstriction, or a combination of these factors. Nonocclusive ischemia tends to resolve spontaneously and has a better prognosis than occlusive ischemia.
Small-bowel ischemia is usually due to occlusion of the superior mesenteric artery or vein (Figure 14) 22 or their branches and tributaries. The distribution and length of involvement depend on the site of vascular obstruction. The jejunum, ileum, and right colon may be affected and the duodenum (supplied by the celiac axis) spared. Morbidity and mortality are high, especially for arterial occlusions.
Nonocclusive ischemia of the small bowel is less common. Possible causes include hypovolemia (Figure 15, "shock bowel"), drugs, and vasculitis. These usually result in diffuse small-bowel involvement without sparing the duodenum. Long segments of colon may also be affected. Radiation enteritis, another cause of nonocclusive ischemia, is not as common as radiation colitis, due in part to the greater mobility of small-bowel loops.
As opposed to small-bowel ischemia, colonic ischemia is usually due to nonocclusive factors. 9 Watershed areas (splenic flexure to descending colon and rectosigmoid) are most frequently involved. However, any portion of the colon can be affected, especially with embolic sources. Radiation colitis affects bowel within the radiation field, most commonly the rectosigmoid. 9
CT findings of bowel ischemia depend upon the presence of reperfusion. In ischemia without reperfusion (active ischemia), the bowel wall is hypo- or nonenhancing (Figure 5) and may be thin, and the mesenteric vascular structures are diminutive. In ischemia with reperfusion (Figure 14), the wall is thick, the mucosa and muscularis/serosa are hyperemic (striated, halo, or target pattern) and the mesenteric vessels are engorged. Engorged, hyperenhancing vasa rectae may penetrate the involved bowel wall and stand out against the edematous submucosa ("starry sky," Figure 14). Ancillary findings suggestive of bowel ischemia include pneumatosis and portomesenteric gas. 4,22
Inflammatory bowel disease (IBD)
Crohn's disease (CD) or ulcerative colitis (UC) may cause acute abdominal pain. Although a history of IBD is usually known, new presentations occur. In these cases, a combination of direct and ancillary findings on CT often permits correct diagnosis.
The primary CT feature of acute IBD is mild-to-moderate bowel wall thickening (mean of 10 to 11 mm for CD and 7 to 8 mm for UC). 3 The involved wall may hyperenhance. Typically, UC manifests a stratified pattern (Figure 16), 3 whereas CD may manifest a stratified or homogeneous pattern (Figure 17). Mesenteric vessels may be engorged with either CD or UC. 3 Vascular engorgement in the setting of active CD has been termed the comb sign and is analogous to the centipede sign of diverticulitis.
The distribution of wall thickening is important. Crohn's disease affects the small bowel alone in 20% of cases, the small bowel and colon in 60%, and the colon alone in 20% of cases (right more commonly than left colon) 23 ; terminal ileal involvement and skip lesions are characteristic. In contrast, UC causes continuous involvement proximally from the rectum (Figure 17) 23 ; the terminal ileum may be abnormal in a minority of cases of long-standing diffuse UC ("backwash ileitis"). 23 Fibrofatty proliferation around the terminal ileum ("creeping fat") is diagnostic of CD (Figure 16) and helps differentiate CD from UC and any disease of the terminal ileum. Fibrofatty proliferation around the rectum also occurs but is nonspecific and can develop with any chronic proctitis (UC, CD, or other). Sacroiliitis is more characteristic of UC. Other extraluminal complications (fistula, sinus tract, abscess, septic thrombosis of portomesenteric veins) are distinctly more common in CD.
Infection
Gastrointestinal infection can cause severe abdominal symptoms. Acute infections of the small bowel are usually subtle on CT and may have no CT imaging abnormalities. Colonic infections are usually obvious because mucosal ulceration and submucosal edema cause detectable colonic thickening. If thickening is severe, the colon may assume an accordion-like configuration (Figure 18). Transmural inflammation leads to adjacent fat stranding and/or ascites. Although obvious, CT findings are usually nonspecific and overlap with noninfectious causes of colitis. However, some features aid in differential diagnosis. For example, Yersinia, myco-bacteria, and amebiasis have a predeliction for the ileocecal region. Cytomegalo- virus (CMV) and Clostridium difficile (Figure 18) often cause diffuse colonic edema, and the degree of mural thickening may be severe.
Although immunocompromised patients are susceptible to the normal spectrum of GI infections, some infections are particularly characteristic in this population. These opportunistic pathogens include mycobacteria (tuberculosis and Mycobacterium avium intracellulare), viruses (CMV), parasites (Cryptosporidium) and, in the setting of neutropenia, typhlitis (neutropenic colitis, Figure 19).
Conclusion
CT plays an increasingly important role in the evaluation of the acute abdomen. Characterization of the primary abnormality and the identification of ancillary findings permit accurate diagnosis and help guide and expedite appropriate management.
