Interventional uroradiology in the management of prostate cancer
Images
Prostate cancer is the most common cancer arising in men and the second most common cause of death among men. In 2011, nearly 210,000 new cases were diagnosed, with just over 27,000 deaths related to prostate cancer.1 For 2015, it is estimated that the rate of new cases will be approximately 220,000. The majority of patients continue to be 65 or older and often have co-morbid disease in other body systems. 2
As promoted throughout September—Prostate Cancer Awareness Month in the United States—routine screening through physical examinations and measurement of prostate-specific antibody are important for early detection. When detected early, most prostate cancers are confined to the gland without metastatic spread. Presentation as early, localized disease versus advanced and potentially metastatic disease will impact diagnostic and therapeutic considerations.
Depending upon the stage of disease and extent of metastatic spread, management may include radiation therapy, minimally invasive ablations, chemotherapy and/or surgery. Interventional radiologists can play a significant role along with urologists, medical oncologists and radiation oncologists in the care of these patients. The role of the interventional radiologist is enhanced when disease is confined to the prostate gland.
Image-guided urologic interventions for prostate cancer include diagnostic biopsies, fiducial marker placements for external beam radiation, ultrasound-guided and primary ablative therapies, and fluid collection drainages. Each procedure is performed in close collaboration with the management team. With correct patient selection and procedural preparation and technique, morbidity and mortality are minimized for optimum patient outcomes.
Direct prostate gland biopsies most commonly occur with transrectal ultrasound (TRUS) guidance. Alternatively, magnetic resonance imaging (MRI) may be utilized for transrectal guidance. With both modalities, patients are typically placed in a decubitus position. Self-limiting hematuria may frequently occur. TRUS guided biopsies are relatively easy to perform in an outpatient office setting. MRI guidance offers more precise tumor localization, but requires greater procedural intensity and time, leading to higher costs. In select cases when a transrectal approach is not feasible, a trans-gluteal approach with computed tomography guidance many be a consideration.3
Current state-of-the-art external beam therapy for prostate cancer utilizes hypofractionation, in which a reduced number of higher therapeutic radiation doses are delivered to the tumor. This therapy is termed stereotactic body radiotherapy (SBRT) when performed with five or fewer fractions. One of the challenges with hypofractionation is the unpredictable movement of the prostate gland during treatment. This motion occurs as x, y, and z orthogonal translation and rotation, in part secondary to bladder and rectal filling in addition to prostate interfraction transient inflammation.4 As SBRT uses smaller margins to allow for high daily radiation deposition, uncorrected prostate motion can result in suboptimal dose to the target and an excessive dose to surrounding nontarget structures. It is important therefore to track and correct prostate motion during SBRT.5
Under image guidance, fiducial markers can be placed into the prostate gland to track the prostate during treatment and then to correct for motion. Fiducial markers are particularly needed with the use of robotic accelerator radiation delivery SBRT systems, such as the Cyberknife (Accuray Inc., Sunnyvale, CA). The Cyberknife delivers isodose profiles directly to the prostate tumor, utilizing 1) a rapid dose fall-off for adjacent nontarget structures and 2) real-time image guidance during radiation delivery to correct for the translational and rotational changes in the prostate gland position. Real-time correction of motion is accomplished with two orthogonal digital x-ray monitoring of fiducial marker positions.
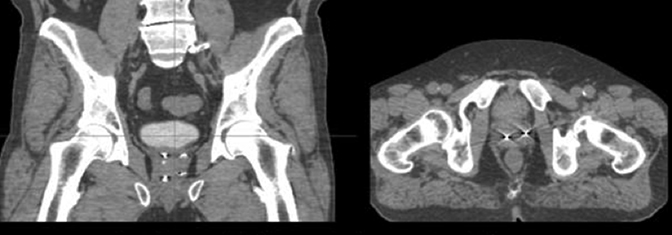
For image-guided fiducial placement, prior to Cyberknife planning gold fiducial markers are placed within the prostate gland, via a transperineal or transrectal approach under TRUS guidance. To employ 6-dimensional correction, at least 3 fiducials are required. Markers should be deposited with at least 2 cm of separation and at least a 15° angle between any fiducial triplets. A common methodology in our practice involves transperineal placement of the markers at the left base, left apex, right base, and right apex using double-fiducial loaded needles with a spacer between each fiducial (Figure 1). With optimal techniques, fiducial markers can be placed with minimal (<1mm) positional migration at 1 week, potentially affording planning on the same day of marker implantation.6
Alternative image-guided ablative uroradiologic interventions for prostate cancer include high intensity focused ultrasound (HIFU) and cryotherapy. HIFU utilizes 0.8 to 3.5MHz sound waves via transrectal probe, to cause hyerthermic tumor cell necrosis and destruction. MRI can be used during HIFU for contiguous temperature monitoring, thermal dose mapping, and intraprocedural adjustments. Cryotherapy results in tumor-cell destruction by freezing and thawing. Typically argon gas or liquid nitrogen is delivered for tumor freezing under TRUS guidance. A transrectal approach is used for delivering liquid nitrogen, while a trans-rectal or transperineal approach may be used for argon. Helium is utilized for warming, typically via the transurethral approach. 7
Following radiation therapy, ablative therapies, and surgical resections for prostate cancer, simple fluid collections and abscesses can occur within the pelvis. Diagnosis is typically made with CT, which is then utilized for procedural planning. Depending upon the location of the collection and adjacent viscera and vascular structures, drainage may occur via a transgluteal, transabdominal/pelvic, or transrectal approach under CT (transgluteal, transabdominal/pelvic) or ultrasound guidance (transrectal).
References
- Centers for Disease Control and Prevention. http://www.cdc.gov/cancer/prostate/statistics/. Accessed Aug. 24, 2015.
- American Cancer Society. http://www.cancer.org/cancer/prostatecancer/detailedguide/prostate-cancer-key-statistics. Accessed Aug. 24, 2015.
- Goenka AH, Remer EM, Veniero JC, et al. CT-guided transgluteal biopsy for systematic random sampling of the prostate in patients without rectal access. Am J Roentgenol. 2015; 205(3):578-583.
- Xie, et al. Int J Radiat Oncol Biol Phys. 2008;72(1):m236-246.
- Lovelock DM, et al. Int J Radiat Oncol Biol Phys. 2015;91(3): 588-594.
- Kumar KA, Wu T, Tonlaar N, et al. Image-guided radiation therapy for prostate cancer: A computed tomography-based assessment of fiducial marker migration between placement and 7 days. Pract Radiat Oncol. 2015;5(4):241-247.
- Nomura T, Mimata H. Focal therapy in the management of prostate cancer: An emerging approach for localized prostate cancer. Adv Urol. 2012; 2012:1-8.
Citation
Interventional uroradiology in the management of prostate cancer. Appl Radiol.
September 2, 2015