Leontiasis ossea
Images
CASE SUMMARY
A 37-year-old female with a history of end-stage renal disease on hemodialysis presented with an eight-week history of slowly progressing maxillofacial pain and swelling. More recently, she claimed difficulty with phonation and dyspnea, which prompted her to seek medical attention. Physical examination was positive for maxillary and mandibular hypertrophy with splaying of the teeth and pain to palpation. Past medical history was pertinent for right femoral neck and supracondylar humeral fractures attributed to a diagnosis of renal osteodystrophy. Pertinent laboratory values included: parathyroid hormone 5,707 pg/mL (normal range 12-65) and alkaline phosphatase 2,784 units/L (normal range 0-120). During her hospitalization, a coordinated surgery with ENT and surgical oncology included tracheostomy, bone biopsy of the maxilla, and four-gland parathyroidectomy.
IMAGING FINDINGS
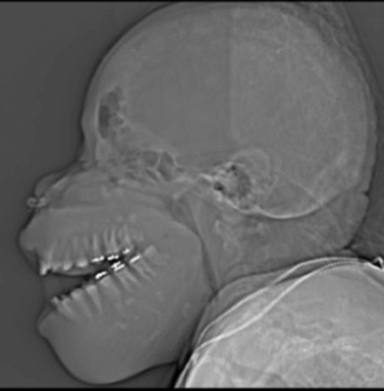
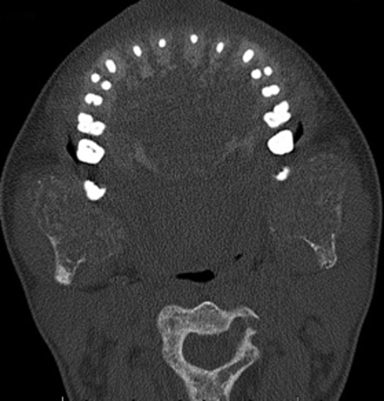
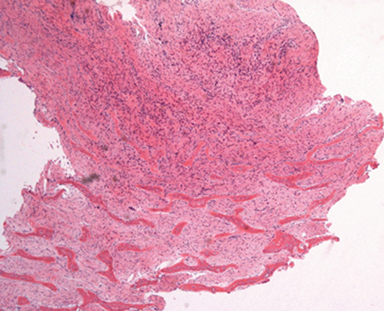
A preliminary CT scout image demonstrated hypertrophy of the maxilla and mandible with near complete obliteration of the maxillary sinuses and narrowing of the pharynx. Widening of the interdental spaces, and an expanded, heterogeneous appearing diploic space was also identified (Figure 1). Maxillofacial CT additionally demonstrated diffuse loss of corticomedullary differentiation with near complete, symmetric replacement of the osseous matrix of the mandible and maxilla with expansile soft tissue. This included tunneling of soft tissue through the residual osseous matrix (Figure 2, this image is available in a DICOM viewer powered by EXA-PACS). A heterogeneous lytic and sclerotic appearance to the calvarium and cervical spine was noted. The maxillary bone biopsy showed a fibrooseous lesion composed of cellular fibrous stroma with woven bone islands with many having osteoclastic rimming (Figure 3). No giant cells were present.
DIAGNOSIS
Renal osteodystrophy in the setting of hyperparathyroidism, leontiasis ossea pattern. Differentials include fibrous dysplasia, cherubism, giant cell tumor.
DISCUSSION
Renal osteodystrophy is described as the osseous sequelae of hyperparathyroidism secondary to renal failure where calcium from the bone is mobilized to replenish the body’s calcium requirements. Common locations include the ribs, inferior margin of the femoral neck, pubis, medial proximal tibia, calvarium, vertebrae, and distal aspects of the clavicles, phalanges, and ulna.
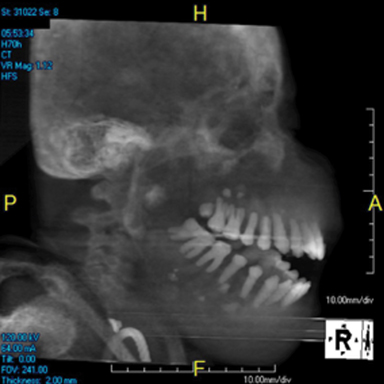
Facial involvement is rarer and often presents clinically with progressive swelling—sometimes painful due to nerve encroachment and tissue ulceration—leading to dysphagia, dysarthria, and airway compromise.1,2 The term “leontiasis ossea” has been generalized to describe the appearance of craniofacial hypertrophy in the setting of many diseases – paget disease, fibrous dysplasia, reactive inflammatory bone disease, renal osteodystrophy—which result in leonine facies.1,2 This manifests physically as widening of the interdental spaces, flattening of the nares and nasal bridge, and jaw enlargement (Figure 4).1,3 There is no sex or race predilection; however, some have proposed that younger individuals—age 10-30—have craniofacial bones more susceptible to parathyroid overstimulation since they are still normally undergoing active osseous development and remodeling.1 Laboratory abnormalities are those seen with secondary/tertiary hyperparathyroidism and include elevated parathyroid hormone, elevated alkaline phosphatase (indicating bone turnover), hypo- or normocalcemia, hyperphosphatemia, and decreased 1,25 dihydroxyvitamin D.
Radiologically, the maxillofacial changes of renal osteodystrophy have previously been broken down into three separate patterns.3 Osteitis fibrosa is a constellation of osteolytic lesions, cortical thinning, and coarsened trabeculation.4,5 Microlunar osteopenia in the skull gives the classic “salt-and-pepper” appearance. Cystic brown tumors are also seen in this pattern. These are expansile, eccentric, lytic lesions with occasional internal trabeculation, cortical thinning, and scarce periosteal reaction.4,6 The second pattern is a more generalized fibrotic appearance with replacement of the normal medullary space by ground glass attenuation. Poor corticomedullary differentiation is the hallmark of this stage and remains an important differentiator from fibrous dysplasia.3,6 The rarest pattern—and that most representative of an advanced disease state— is uremic leontiasis ossea which presents as marked expansion of the mandible and maxilla, occasionally with obliteration of the maxillary sinuses and splaying of the teeth.1,6 There is poor visualization of cortical bone with intermixed serpentine tunneling through the residual matrix.3 In the cranium, widening of the diploic space is demonstrated with a heterogeneous appearance.
On histological analysis, curved spicules of woven bone are seen amongst a background of extensive fibrosis, sometimes with large resorptive lacunae walled with osteoclasts.1,3,7 An important element on analysis is the presence or absence of multinucleated giant cells. In the current case, no giant cells were visualized. This provides differentiation from additional entities that typically include giant cells: cherubism, central giant cell granuloma, giant cell tumor in the setting of paget’s disease, and aneurysmal bone cyst.8-10 It is important to note; however, that giant cells are present in some manifestations of renal osteopathy, such as brown tumor.4,6
Patient age and sex should also be considered. Entities such as cherubism usually present at 2-3 years of age and spontaneously resolve after puberty.8Paget’s disease typically presents later than the current patient’s age and has a male predilection. Fibrous dysplasia is usually quiescent once skeletal maturity is achieved; however, it can be seen in adults.
Lesion location and distribution within the jaw is also important, as odontogenic tumors are in approximation with the root apex or crown of a tooth and are more focal.6 Entities such as Paget’s and fibrous dysplasia can have similar leonine facies but tend to asymmetrically involve the maxilla and mandible. Often times, histological findings need to be supplemented with radiographic, biochemical, and clinical findings to confidently differentiate these entities.
Treatment usually entails parathyroidectomy with monitoring of serum calcium, phosphate, and vitamin D levels while on appropriate replacement (calcium/vitamin D) or binding (phosphate) pharmacotherapy.4 The prognosis is usually favorable. In the instance that the facial deformities of advanced renal osteopathy do not regress, additional surgery is sometimes indicated for further alleviation of symptoms and cosmesis.2,7
CONCLUSION
Several radiologic patterns of maxillofacial involvement have been described in the setting of renal osteodystrophy. Leontiasis ossea is believed to be the rarest and manifests clinically as widening of the interdental spaces, flattening of the nares and nasal bridge, and jaw enlargement. Soft tissue tunneling through the residual osseous matrix remains a key imaging finding. Histological and biochemical testing can provide further differentiation from similar entities such as giant cell tumors and fibrous dysplasia.
REFERENCES
- Lee VS, Webb MS, Martinez S, McKay CP, Leight Jr GS. Uremic leontiasis ossea: “Bighead” disease in humans? Radiologic, clinical, and pathologic features. Radiology. 1996;199:233-240.
- Haroyan H, Bos A, Ginat DT. Uremic leontiasis ossea. Am J Otolaryngol. 2015;36: 74-76.
- Chang JI, Som PM, Lawson W. Unique imaging findings in the facial bones of renal osteodystrophy. Am J Neuroradiol. 2007;28:608-609.
- Antonelli JR, Hottel TL. Oral manifestations of renal osteodystrophy: case report and review of the literature. Spec Care Dentist. 2003;1:28-34.
- Yang Q et al. Skeletal Lesions in primary hyperparathyroidism. Am J Med Sci. 2015;349(4):321-327.
- Cure JK, Vattoth S, Shah R. Radiopaque jaw lesions: an approach to the differential diagnosis. Radiographics. 2012;32:1909-1925.
- Michiwaki Y, Michi K, Yamaguchi A. Marked enlargement of the jaws in secondary hyperparathyroidismd- a case report. Int J Oral Maxillofac Surg. 1996;25:54-56.
- Jain V, Sharma R. Radiographic, CT and MRI features of cherubism. Pediatr Radiol. 2006;36:1099-1104.
- Theodorou DJ, Theodorou SJ, Sartoris DJ. Primary non-odontogenic tumors of the jawbones an overview of essential radiographic findings. Clin Imaging. 2003;27: 59-70.
- Sun ZJ, Cai Y, Zwahlen RA, Zheng YF, Wang SP, Zhao YF. Central giant cell granuloma of the jaws: clinical and radiological evaluation of 22 cases. Skeletal Radiol. 2009;38: 903-909.
Citation
NS C, A B. Leontiasis ossea. Appl Radiol. 2017;(12):25-27.
October 19, 2017