Myofibroblastoma of the breast
Images
CASE SUMMARY
A 70-year-old woman presented with solitary round mass of right lateral breast. The patient underwent an ultrasound-guided core biopsy of this mass at an outside institution. The biopsy was originally misinterpreted as an invasive lobular carcinoma. She subsequently underwent excision of mass (lumpectomy) and sentinel node sampling.
IMAGING FINDINGS
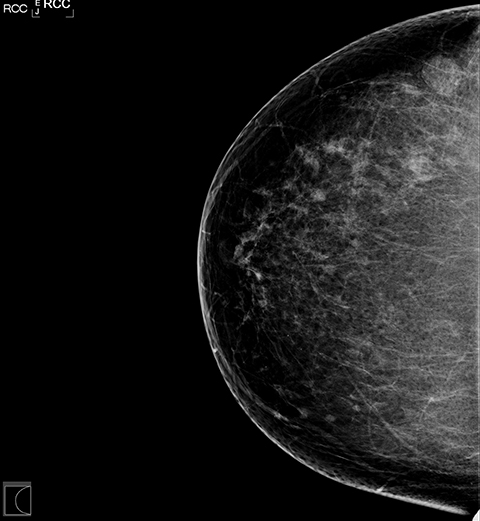
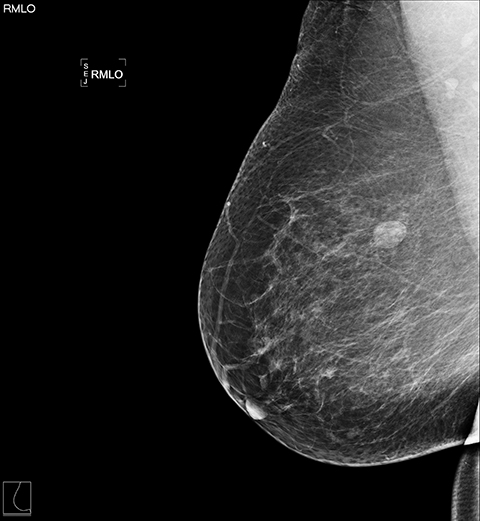
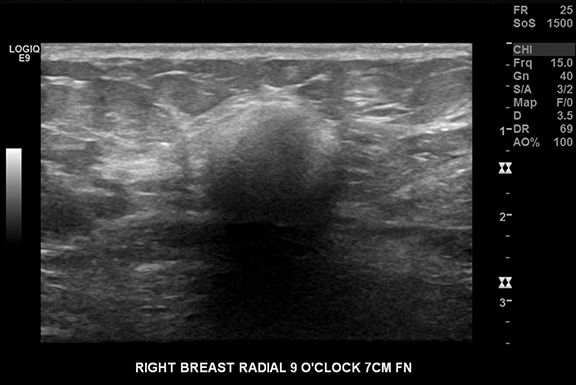
Standard mammographic views and diagnostic views of the right breast demonstrated heterogeneously dense breast tissue and a rounded mass in the lateral right breast (Figures 1A and 1B). Focused ultrasound of the lateral breast demonstrated a circumscribed hypoechoic indistinct shadowing mass, measuring 13 × 13 × 12 mm (Figure 2). Because of these suspicious imaging features, biopsy was performed for histologic diagnosis with 5 tissue cores obtained. A biopsy-marking clip was placed.
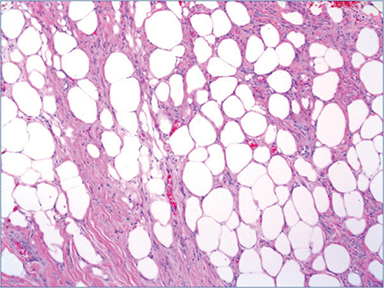
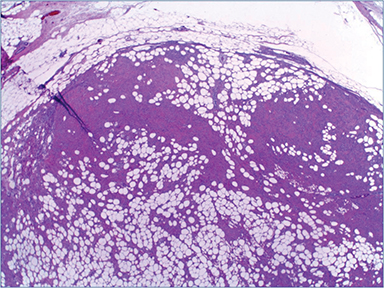
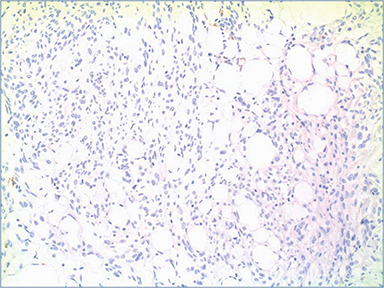
Pathologic evaluation of the ultrasound-guided core biopsy showed a nodular proliferation of uniform, bland ovoid to spindle-shaped cells separated by prominent adipose tissue and hyalinized collagen bundles, with no significant atypia or mitotic activity (Figure 3). Immunostains performed by the outside institution revealed that these cells were positive for estrogen (ER) and progesterone receptors (PgR), and were negative for E-cadherin. Excisional biopsy (lumpectomy) showed a similar appearing neoplasm (Figure 4), but this proliferation was very well circumscribed (as seen on initial imaging). Although the lipomatous component was prominent on the initial core biopsy, on excision this component comprised less than 50 percent of the lesion; the majority of the lesion displayed a predominance of spindled cells and hyalinized collagen bands. The two sentinel lymph nodes examined on routine hematoxylin and eosin and cytokeratin (AE1/AE3) immunostains were benign.
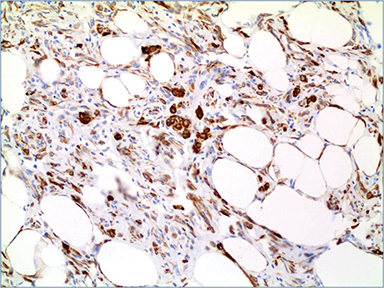
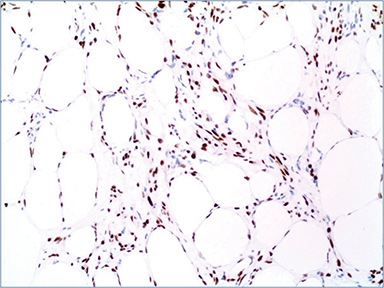
Additional immunostains performed at our institution revealed negative staining of the lesional cells for wide spectrum cytokeratin (CK), CK OSCAR (Figure 5A), epithelial membrane antigen (EMA), S100, HMB-45, smooth muscle actin and positive staining for CD34, desmin (Figure 5 B), estrogen receptor (Figure 6), and BCL-2. The overall histopathological and immunostaining patterns were interpreted as a myofibroblastoma with prominent adipocytic component.
DIAGNOSIS
Myofibroblastoma of breast with prominent adipocytic component
DISCUSSION
Myofibroblastoma (MFB) is a rare benign spindle cell tumor of the breast. First described by Wargotz et al in 1987,1 MFB typically presents as a slow growing, well circumscribed, solitary mass ranging from 1-4 cm in size. Although it has been reported in adults of either gender, it is most frequently seen in older males and in postmenopausal women.2 This lesion is composed of stromal cells showing fibroblastic and myofibroblastic differentiation at the morphological, immunohistochemical and ultrastructural levels.1,2 Histologically, the spindled to ovoid cells of MFB are arranged in interweaving fascicles separated by collagen bands, occasionally with entrapped breast glands.1,2 Typically, the lesional cells of MFB are immunoreactive for muscle markers, including desmin and smooth muscle actin due to their myofibroblastic origin. Additionally these lesions usually stain for CD34, desmin, BCL2, CD99, CD10 and WT1.3 There is variable positivity for ER and PgR.4
Multiple variants of MFB have been described, including cellular, infiltrative, epithelioid, deciduoid-like, lipomatous, collaginized/fibrous and myxoid. The term “lipomatous myofibroblastoma” has been used to designate those cases of MFB containing a significant mature fatty component (approximately 75 percent of the entire tumor).3,6 In the lipomatous variant of MFB, the presence of infiltrative epithelioid appearing cells with admixed adipose tissue, may lead to the possibility of misdiagnosing these lesions as an invasive carcinoma with lobular growth pattern infiltrating adipose tissue.5
Needle core biopsy of MFB can be difficult to interpret based on small sampling, especially if one is faced with an unusual variant.3 In our case, although the lipomatous component did not ultimately fulfill the definition of “lipomatous myofibroblastoma” on excision, the presence of a significant adipose component intermixed with the plump spindled cells created the false impression of an infiltrating carcinoma. Moreover, the positive immunohistochemical stains for hormone receptors (ER and PgR) and absence of E-cadherin staining further reinforced the mistaken impression of invasive carcinoma.4 Thus, additional immunohistochemical staining and awareness of breast stromal lesions are essential in determining the correct diagnosis, along with close collaboration between radiologist and pathologist in correlating imaging with pathologic findings.
Mammography of MFB typically shows a round to oval mass without microcalcifications. Ultrasonography usually demonstrates a solid circumscribed tumor, although a variable and mixed echo pattern can be expected, sometimes with more distal acoustic attenuation as a result of fat tissue and other types of tissue in tumor. Doppler modality may show a slight peripheral hypervascularization of the tumor. According to the imaging, the differential diagnosis includes hamartoma, fibroadenoma and lipoma; however in this case, the lesion did not demonstrate clearly benign features by mammography or ultrasound thus prompting core needle biopsy.6-8
Surgery is the recommended treatment for MFB since these lesions may recur if not entirely excised.9 Due to its circumscription with good cleavage planes; these tumors are usually easily excised.
CONCLUSION
This case report highlights the diagnostic pitfall of mistaking myofibroblastoma for invasive lobular carcinoma in the setting of core biopsy with hormone receptor positivity. And it emphasizes the importance of radiologic-pathologic correlation in assuring a correct diagnosis, particularly when faced with a challenging radiologic and pathologic entity.
REFERENCES
- Wargotz ES, Weiss SW, Norris HJ. Myofibroblastoma of breast: 16 cases of distinctive benign mesenchymal tumor. AJ Surg Pathol.1987; 11:493-502.
- Lerwill MF, Koener FC, Benign mesenchymal neoplasms. In: Hoda SA, Brogi E, Rosen’s breast pathology, 4th edition Philadelphia PA. WoltersKluwer. 2014:1019-1094.
- Magro G. Mammary fibroblastoma: A tumor with a wide morphologic spectrum. Arch Pathol lab Med. 2008;132(11):1813-1820.
- Magro G. Epithelioid-cell myofibroblastoma of the breast: Expanding the morphologic spectrum. Am J Surg. Pathol. 2009;33(7): 1085-1092.
- Wahbah MM, Gilcrease MZ, Wu Y. Lipomatous variant of Myofibroblastoma with epithelioid features: a rare and diagnostically challenging breast lesion. Ann Diagn Pathol. 2011;15(6):454-458.
- Magro G, Michal M, Vasquez E, Bisceglia M. Lipomatous myofibroblastoma: a potential diagnostic pitfall in the spectrum of the spindle cell lesions of the breast. Virchow’s Arch. 2000; 437(5):540-544.
- Dockery WD, Singh HR, Wilentz RE. Myofibroblastoma of the male breast: imaging appearance and ultrasound-guided core biopsy diagnosis. Breast J. 2001;7(3):192-194.
- Greenberg JS, Kaplan SS, Grady C. Myofibroblastoma of the breast in women: imaging appearances. AJR Am J Roentgenol. 1998;171(1):71-72.
- Mele M, Jensen V, Wronecki A, Lelkaitis G. Myofibroblastoma of the breast. Case report and literature review. Int J Surg Case Rep. 2011; 2(6):93-96.
Citation
P R, TJ L, A C. Myofibroblastoma of the breast. Appl Radiol. 2017;(3):42-44.
March 4, 2017