New PET-CT, SPECT, amyloid-imaging technologies unveiled at SNMMI
June 10, 2013 – Siemens Healthcare is featuring its new Biograph mCT Flow is a new positron emission tomography/computed tomography (PET/CT) system and a new high sensitivity single-photon emission computed tomography (SPECT) system at the 2013 annual meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI), June 8-12, in Vancouver, B.C., Canada.
The Biograph mCT Flow overcomes the limitations of conventional bed-based PET/CT with FlowMotion, a new technology that moves the patient smoothly through the system’s gantry, while continuously acquiring PET data. FlowMotion of Biograph mCT Flow is designed to enable physicians to conduct customized scans tailored to the patient’s unique anatomy for the finest3 detail in every organ. Additionally, FlowMotion provides accurate and reproducible quantification in all dimensions for precise disease characterization in therapy monitoring. Finally, Biograph mCT Flow eliminates radiation due to overscanning, while its continuous motion and wide 78 cm bore provides the patient with a more relaxing diagnostic experience. For more information on Biograph mCT Flow, please refer to the accompanying press release on the new system.
The company is also
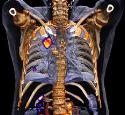
displaying Symbia Intevo, an xSPECT system, which combines the high sensitivity
of single-photon emission computed tomography (SPECT) with the high specificity
of CT, completely integrating the data from the 2 modalities, to generate high
resolution and for quantitative images.1 Siemens’ Symbia
Intevo™1 completely integrates SPECT and CT enabling a new level of
image quality and quantification
In traditional SPECT/CT Symbia Intevo’s precise alignment of SPECT and CT provides physicians with
volumetric information from the CT scan, enabling accurate and consistent
quantitative assessment—a numerical indication of a tumor’s level of metabolic
activity. With accurate quantitative assessment, the physician can apply
quantitative information to assess whether a patient's course of treatment has
regressed, stabilized or grown—something that is difficult to do with a purely
visual assessment of the tumor.
While Symbia Intevo uses more CT data than ever before, Siemens is still able
to limit patient dose by offering combined applications to reduce exposure
(CARE). These applications, which are unique to Siemens, include the CARE
Dose4D™ technique, which can reduce patient CT radiation dose by up to 68%.4
Symbia Intevo also offers applications to improve productivity and patient throughput.
For example, Siemens’ AUTOFORM collimator—which is standard on Symbia
Intevo—captures up to 26 more counts, or photons that are generated from
radiotracer activity.4 This increased number of counts
potentially boosts image acquisition time and patient throughput.
Additionally at this year’s SNMMI annual meeting, Siemens Molecular Imaging
will highlight a comprehensive amyloid imaging solution5 for
the visualization and quantification of amyloid plaques in order to evaluate
the pathology of Alzheimer’s disease in the living brain. This comprehensive
amyloid imaging solution includes the Biograph mCT PET•CT family of scanners,
syngo.PET Amyloid Plaque software5—which enables the physician to
evaluate amyloid plaque density in the living brain—and the radiopharmaceutical
production and distribution expertise of PETNET Solutions, a wholly owned
Siemens subsidiary. This unique combination of scanner, software and PET
radiopharmaceutical expertise is a valuable tool for the evaluation of
Alzheimer’s disease and other causes of cognitive decline.
Siemens will also introduce the new Explora One™ chemistry box, a fully
integrated cassette-based solution for the production of commercially available
and newly released 18F PET imaging biomarkers, as well as imaging biomarkers
under development or in clinical trials. A fully integrated hardware design
aids compliance with a wide range of imaging biomarker production scenarios,
with the benefit of reduced lab space, which in turn reduces initial setup
costs. Explora One also virtually eliminates user error by providing an
automated and integrated workflow for easy imaging biomarker production,
improving production output efficiency and quality and decreasing the risk of
loss of revenue due to human error and contamination.
For information: www.healthcare.siemens.com and www.appliedradiology.com
Related Articles
Citation
New PET-CT, SPECT, amyloid-imaging technologies unveiled at SNMMI. Appl Radiol.
June 10, 2013