Radiological Case: Non-papillary bladder metastasis from pancreatic adenocarcinoma
Images


CASE SUMMARY
A 50-year-old Caucasian woman, with significant history of hypothyroidism, hypertension, GERD, and tobacco use, presented with acute onset of right upper quadrant pain.
IMAGING FINDINGS AND DIAGNOSIS
Ultrasound revealed normal gallbladder and non-visualization of pancreas due to bowel gas. A hepatobiliary scan revealed that the gallbladder had an abnormal ejection fraction of 33%, suggestive of biliary dyskinesia. Labs were all within normal limits with the exception of low potassium of 2.9. Laparoscopic cholecystectomy with intraoperative cholangiogram was indicated at that time. Upon entrance into the intraperitoneal cavity there was evidence of peritoneal implants on at the dome of the right hemidiaphragm. There was also an omental nodule identified overlying the right transverse colon. Both lesions were positive for CA19-9, CEA, CK20, and CK7 immunohistochemical stains. The findings were consistent with moderately differentiated metastatic pancreatic adenocarcinoma. Intraoperative cholangiogram revealed normal filling of the intrahepatic ducts, common hepatic duct, common bile duct, and cystic duct with normal flow into the duodenum. Further exploration revealed one small nodule on the parietal peritoneum of the pelvic region. No evidence of primary neoplasm was discovered at the time.
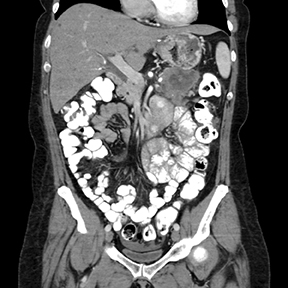
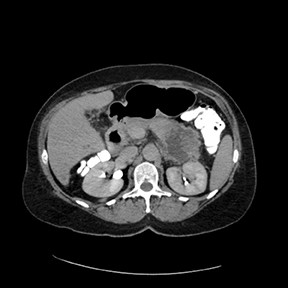
A CT scan on post-op day 9 (Figures 1 and 2) revealed a large, irregular, hypodense, heterogeneously enhancing lesion involving the tail of the pancreas. The lesion measured 5.4cm x 6.5cm. Central necrosis was appreciated within the lesion. The lesion extended to involve the anterior left pararenal fascia and was in close proximity to the posterior aspect of the greater curvature of the stomach. Partial involvement of the splenic vein was noted as well. Central retroperitoneal lymphadenopathy with a few prominent lymph nodes in the left periaortic window was also noted.
The patient initially received chemotherapy under clinical trial with gemcitabine, and trial drug LY2603618 after disease progression was noted 11 months after diagnosis. She was then treated with FOLFOX and was switched to Gemzar and Abraxane thereafter.
Twenty-one months after her initial diagnosis, the patient presented to the ED with complaints of fever, weakness, and passing blood clots in urine with dysuria. Labs revealed that the most recent CA-19-9 had increased from 500-2500 in the last month. Hemoglobin was 10.6, hematocrit: 31.5, WBC: 6,800, potassium: 2.9, BUN: 14, creatinine: 0.75, platelet count: 123,000.
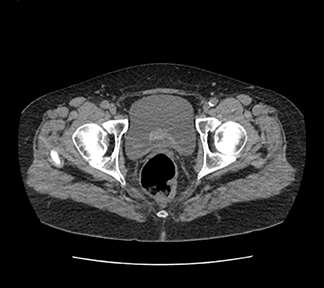
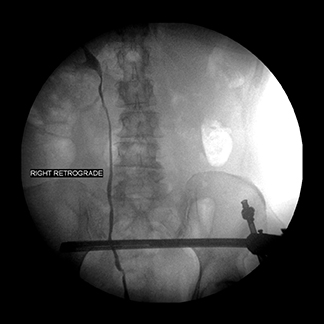
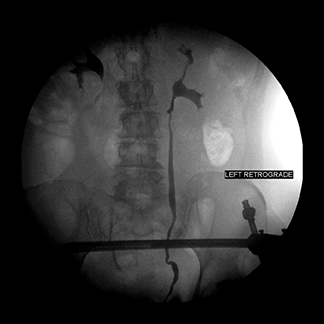
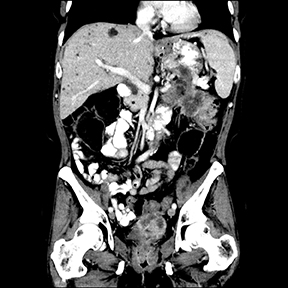
A CT scan demonstrated an irregular mass in the posterior aspect of the bladder (Figures 3 and 4). A digital vaginal exam demonstrated multiple masses on the anterior wall of the vagina. Cystoscopy noted a large, irregular, pedunculated mass in the midline of the bladder, starting at the trigone posteriorly, extending back to the posterior wall of the bladder. The mass bled heavily with gentle manipulation. Transurethral resection of the bladder tumor (TURBT) was initiated due to poor visualization of ureteral orifices. After resection, both ureteral orifices and retrograde pyelograms were performed (Figures 5 and 6). No evidence of any filling defects or abnormalities was present. Resected tumor specimen revealed metastatic pancreatic adenocarcinoma with extensive submucosal and muscularis involvement.
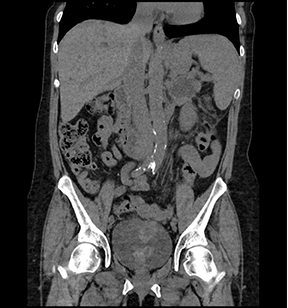
Six weeks after the patient’s original TURBT (22 months after initial diagnosis), she presented back to the hospital for follow-up imaging. A CT scan showed splenomegaly, locally invasive pancreatic tail malignancy involving the adjacent splenic flexure and perirenal fascia of the kidney (Figure 7). Multiple foci of peritoneal carcinomatosis were scattered along abdomen and pelvis under the diaphragm. Left hydronephrosis and hydroureter noted secondary to obstruction of the ureter due to peritoneal carcinomatosis (Figures8 and 9), which had significantly increased since the prior CT, in the peritoneal recess. The patient was taken to surgery for possible ureter stent placement to manage hydronephrosis. Upon cystoscope insertion, the trigone was noted to be elevated. The ureteral orifices could not be identified even with the use of indigo carmine. She was noted to have regrowth of the bladder tumor posteriorly outside of the trigone and it was very friable, and would bleed profusely on contact. The tumor was resected to the base and the ureteral orifices were never appreciated. Following surgery, the patient declined further intervention with percutaneous nephrostomy tubes and was discharged home where she would be comfortable.
DISCUSSION
Pancreatic cancer is the fourth-leading cause of death in the USA among men and women.1 It accounts for 6% of all cancer mortalities in the U.S. in males, and 7% in females.1 In 2013, an estimated 45,220 new cases of pancreatic cancer will be diagnosed nationwide.1 Of those new cases, 38,460 people are expected to die.1 Overall, pancreatic cancer accounts for 7% of all cancer deaths.1 Clinically, the initial presentation of pancreatic cancer depends on location of the tumor. Approximately 60-70% of pancreatic tumors are located at the head of the pancreas, 20-25% are found in the body/tail, and infrequently, the entire organ is involved.2 Tumors found in the head of the pancreas usually present with jaundice, steatorrhea, and weight loss.3 This case represents a pancreatic carcinoma located in the tail of the pancreas which was found incidentally with no other signs and symptoms traditionally associated with the disease.
Urological manifestations usually occur in the late stages of pancreatic carcinoma.4 When the urinary tract is involved, the left side usually predominates.4 The most common manifestations of the urinary tract are ureteral obstruction and renal displacement.4 Hematuria in pancreatic metastasis is rare unless there is mucosal ulceration.6 The only 2 previously reported cases in American literature of metastatic pancreatic adenocarcinoma with hematuria were Kiefer in 19276 and Chiang in 1992.5 In addition, the lesions presented as papillary bladder lesions different in character from ours. The case we have presented is similar to the aforementioned cases, but the bladder metastasis presented as a large, irregular, pedunculated mass invasive to the bladder.
CONCLUSION
Genitourinary metastasis of pancreatic adenocarcinoma is exceedingly rare. Bladder metastases are far less common early in this disease,with only a few cases ever reported. Our case involved a 52-year-old female presenting with new onset hematuria, who was found to have a bladder mass on follow-up imaging almost 2 years after diagnosis of pancreatic carcinoma.
REFERENCES
- Cancer Facts & Figures 2013. American Cancer Society. 2013. http://www.cancer.org/acs/groups/content/@epidemiologysurveilance/documents/document/acspc-036845. pdf. Accessed October 2014.
- Modolell I, Guarner L, Malagelada JR. Vagaries of clinical presentation of pancreatic and biliary tract cancer. Ann Oncol. 1999;10 Suppl 4:82-84.
- Porta M, Fabregat X, Malats N, et al. Exocrine pancreatic cancer: Symptoms at presentation and their relation to tumour site and stage. Clin Transl Oncol. 2005;7:189-197.
- Warden SS, Fiveash JG, Tynes WV II, Schellhammer PF. Urologic aspects of pancreatic adenocarcinoma. J Urol. 1981;125:265-267.
- Chiang KS, Lamki N, Athey PA. Metastasis to the bladder from pancreatic adenocarcinoma presenting with hematuria. Urol Radiol. 1992;13: 187-189.
- Kiefer ED. Carcinoma of the pancreas. Arch Internal Medicine. 1927;40:1-29.