Nonketotic hyperglycemia-induced hemichorea-hemiballism
Images


CASE SUMMARY
A 57-year-old woman presented to the emergency department complaining of headache, nausea and vomiting. Relevant medical history included hypertension and insulin dependent type 2 diabetes with poor to nonexistent medical compliance over the preceding year. Headaches were intermittent by patient report with accompanying vague reported coordination loss in the left upper extremity. Review of systems was positive for mild dysmetria in the left upper extremity. Blood glucose levels were elevated at 574 mg/dl, but secondary laboratory findings were negative for diabetic ketoacidosis. Blood pressure, glucose control and supportive care was initiated. The patient’s symptomatology normalized with supportive care and the subsequent short hospital course and discharge were uneventful.
IMAGING FINDINGS
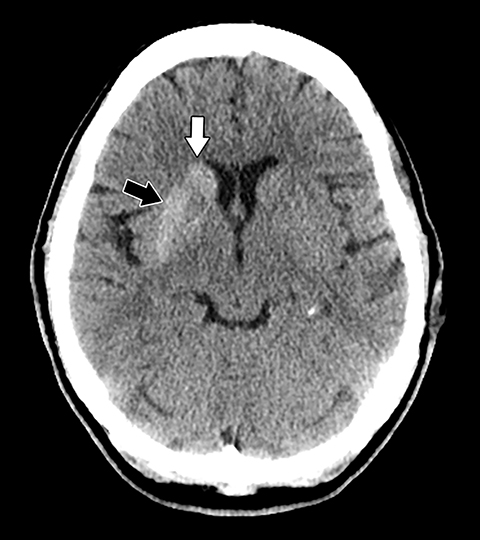
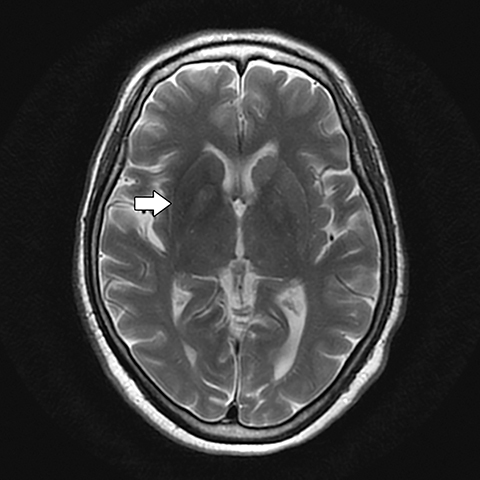
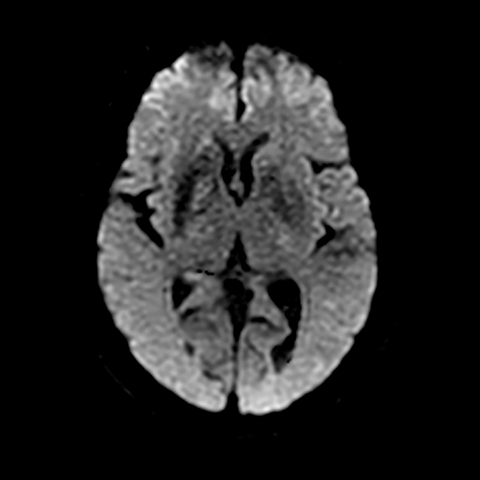
Noncontrast head CT obtained at admission was negative for focal hemorrhage or mass. However, the scan did demonstrate mildly increased density within the right basal ganglia, involving the caudate and lentiform nuclei and to a lesser extent globus pallidus (Figure 1). Subsequently, a cranial MRI scan was performed, demonstrating corresponding increased T1 signal and minimally decreased T2 signal within the right corpora striatum and globus pallidus. Contrast enhancement, diffusion restriction and mass effect were notably absent (Figures 2-5).
DIAGNOSIS
The clinical history of poor glycemic control, dysmetria and coordination loss within the left upper extremity together with the characteristic imaging findings of contralateral basal ganglia hyperdensity on CT and hyperintensity on T1-weighted imaging are characteristic for the diagnosis of nonketotic hyperglycemia-induced hemichorea-hemiballism (NKHH).
The unilaterality of involvement would be atypical for microcalcification, Wilson’s disease or excessive manganese deposition. Additionally, hemorrhage was excluded by the absence of supportive signal changes on MRI and the close conformation of the findings to the anatomic confines of the basal ganglia nuclei, sparing the surrounding internal and external capsules and devoid of mass effect.
DISCUSSION
Hemichorea-hemiballism is a rare hyperkinetic movement disorder characterized by typically continuous unilateral involuntary and nonrhythmic movements of one or both limbs.1 Though basal ganglia infarction accounts for most cases, uncontrolled and prolonged hyperglycemia now represents the second-most common etiology, designated by the clinical acronyms NKHH (nonketotic hemichorea-hemiballism) or HIHH (hyperglycemia-induced hemichorea-hemiballism).2 Elderly type II diabetics with poor medical compliance represent the largest patient cohort, with a slight Asian and female demographic predilection described in the literature.3 Clinical symptomatology may vary widely in severity and tends to wane rapidly and normalize with restoration of glucose control, though occasionally the course may be more protracted, lasting weeks to months. In addition to glucose control, pharmacologic therapy with neuroleptic medications has shown proven utility when necessary.4
The imaging features are relatively characteristic, typified by increased T1 signal intensity on MRI within the basal ganglia contralateral to the symptomatic extremity. There is a predilection for involvement of the putamen. Rarely, bilateral involvement may be observed. Mass effect is conspicuously absent, and the signal changes closely conform to the anatomic confines of the globus pallidus, caudate, and putamen. Slight corresponding T2 shortening may be observed.2,5 Signal loss on gradient echo and susceptibility weighted imaging has been inconsistently reported as has diffusion restriction.5,6 Contrast enhancement is typically minimal to absent.7 CT imaging demonstrates correlative increased density within the affected basal ganglia, again unilateral in distribution and contralateral to the affected extremity.5
The neuroanatomic etiology underlying the imaging pattern has been a source of some contention. Though recent histological reports support T1 shortening due to increased cytoplasmic protein content within reactive gemistocytic astrocytes, likely precipitated by hyperosmolar injury and potential microcirculatory hyperviscosity, other cofactors would likely underlie the density changes on CT.8 Depletion of gamma-aminobutyric acid (GABA), a primary inhibitive neurotransmitter within the basal ganglia in conjunction with non-ketotic hyperglycemia is felt to potentially account for the choreiform and ballistic kinetic movements, though the typical unilateral involvement is yet to be explained.2,8
Differential diagnostic considerations for increased T1 signal within the basal ganglia include early microcalcification, either senescent, metabolically derived, or dystrophic from prior deep gray matter anoxic, toxic or hypoglycemic injury, or Fahr’s disease. Hyperdensity on CT could also be correspondingly expected dependent upon the extent of mineralization.9 Additionally, excessive manganese deposition in conjunction with chronic hepatic encephalopathy or total parenteral nutrition, as well as Wilson’s disease, can present with T1 shortening and basal ganglia hyperintensity.10 A key discriminating factor, however, is the typically bilateral involvement seen with the above listed entities, in contrast to the classically asymmetric and unilateral changes observed in NKHH.11
CONCLUSION
Though rare, the clinical syndrome of hemichorea-hemiballism in conjunction with hyperglycemia displays relatively characteristic imaging features typified by unilateral increased density within the putamen and, to a secondary extent, caudate nucleus and globus pallidus, accompanied by T1 shortening and hyperintensity on MR imaging. The clinical features vary in severity, typically waning rapidly with restoration of glucose control. Familiarity with the imaging features may allow the interpreting radiologist to first suggest the correct clinical diagnosis and expedite timely and appropriate patient care.
REFERENCES
- Ozgur A, Esen K, Kaleagasi H, et al. Diabetic striatopathy in a patient with hemiballism. Emerg Radiol. 2015; 22: 347-349
- Lai P, Tien R, Chang M, Teng M, et al. (1996) Chorea-ballismus with nonketotic hyperglycemia in primary diabetes mellitus. AJNR. 1996;17(6): 1057–1064
- Oh S, Lee K, Im J, et al. Chorea associated with non-ketotic hyperglycemia and hyperintensity basal ganglia lesion on T1 weighted brain MRI study: a meta-analysis of 53 cases including four present cases. J Neurol Sci. 2002; 200:57-62
- Hawley J, Weiner W. Hemiballismus: current concepts and review. Parkinsonism Relat Disord. 2012; 18:125-9
- Bathla G, Policeni B, Agarwal A. Neuroimaging in patients with abnormal blood glucose levels. AJNR. 2014; 35: 833-840
- Chu K, Kang D, Kim D, et al. Diffusion-weighted and gradient echo magnetic resonance findings of hemichorea-hemiballismus associated with diabetic hyperglycemia: a hyperviscosity syndrome? Arch Neurol. 2002; 59: 448-452
- Iwata A, Koike F, Arasaki K, et al. Blood brain barrier destruction in hyperglycemia chorea in a patient with poorly controlled diabetes. J Neurol Sci. 1999; 163(1): 90-93
- Cherian A, Thomas B, Baheti N, et al. Concepts and controversies in nonketotic hyperglycemia-induced hemichorea: further evidence from susceptibility-weighted MR imaging. J Magn Reson Imaging. 2009; 29: 699-703
- Bekiesinska-Figatowska M, Mierzewska H, Jurkiewicz E. Basal ganglia lesions in children and adults. Eur J Radiol. 2013; 82: 837-849
- Rovira A, Alonso J, Cordoba J. MR imaging findings in hepatic encephalopathy. AJNR. 2008; 29: 1612-1621
- Yu F, Steven A, Birnbaum L, et al. T2*-based MR imaging of hyperglycemia-induced hemichorea-hemiballism. J Neurorad. 2016; 44(1): 24-30.
Citation
S C, M R, D L, J B. Nonketotic hyperglycemia-induced hemichorea-hemiballism. Appl Radiol. 2018;(6):24-26.
June 7, 2018