Pulmonary sling
Images


Case Summary
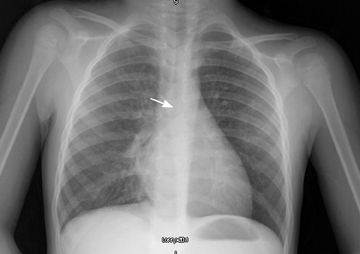
The patient was a 3-year-old girl with a long history of asthma, recurrent pneumonia, dysphagia, and bronchomalacia. She presented to the emergency department with an acute asthma exacerbation on top of her chronic barking cough. The patient had had multiple hospital admissions for asthma exacerbations, croup, and pneumonia, during which she underwent multiple imaging studies, including chest and neck x-rays, a computed tomography (CT) scan of the face as well as a barium swallow. Furthermore, tests for sweat chloride, CH50, immunoglobulin levels, and lymphocyte mitogen response were obtained and all were within the normal limits. During this admission, a chest radiograph (Figure 1) was ordered to rule out pneumonia.
Imaging Findings
The radiograph revealed an indentation of the right tracheobronchial angle and indicated leftward displacement of the trachea. While a right aortic arch was not the final diagnosis, the addition of a vascular anomaly to the differential diagnosis prompted additional diagnostic workup. An echocardiogram was ordered to evaluate the possibility of the right aortic arch, but the exam demonstrated normal aortic and cardiac anatomy. Given the persistent clinical suspicion of a vascular abnormality, a magnetic resonance (MR) study of the chest was obtained.
The MR angiography study showed an abnormal origin of the left pulmonary artery from the right pulmonary artery. The aberrant course of the left pulmonary artery was posterior to the distal portion of the trachea (Figure 2). The trachea measured approximately 5.5 × 5.5 mm in the transverse dimensions above the aberrant vessel, but only 2.5 × 4.6 mm below it. The proximal portions of the right and left main bronchi were narrow, but the distal portions had a normal diameter. The left pulmonary artery was of normal caliber proximally; however, posterior to the trachea the artery narrowed to 2.0 × 6.5 mm, which was best appreciated in the sagittal section (Figure 3). The internal diameter of the esophagus was found to be unaffected on a prior upper GI study, most likely as a result of the narrow diameter of the left crossing pulmonary artery anterior to the esophagus. The cardiovascular surgery team was consulted and subsequently the pulmonary sling was surgically repaired.
Diagnosis
Pulmonary sling
Discussion
Pulmonary vascular slings were first defined in 19581 by Contro et al and recognized as a rare cause of congenital bronchobstruction. They were found to make up only 4% (3/82) of congenital vascular anomalies in one case series.2 During the development of the adult arterial pattern, a sequence of aortic arches forms. The aortic arches then give rise to several major thoracic arteries. The formation of a pulmonary sling (anomalous left pulmonary artery) is the result of a failure of the proximal left 6th arch to properly involute.3 An anastomotic vessel, connecting the primitive pulmonary circulations, becomes the anomalous left pulmonary artery, arising from the right pulmonary artery.4 This vessel then travels above the main pulmonary bronchus to reach the left lung hilum by passing between the trachea and esophagus, often leading to a compression of these structures.5,6
There are two broad categories of pulmonary slings, designated as types I and II. Type I is defined by a normal tracheobronchial pattern, with the tracheal bifurcation at the fourth to fifth thoracic vertebral level.7 Type I slings are further divided into types IA and IB delineated by the presence (A) or absence (B) of a pre-eparterial tracheal bronchus.7 Type I malformations are less complex and are normally associated with tracheobronchomalacia.8 Symptomatic cases produce significant morbidity and mortality, and are usually managed by left pulmonary artery reimplantation surgery.8
Type II slings are subdivided into types IIA and IIB, which are both characterized by an abnormal bridging bronchus and a tracheal bifurcation at thoracic vertebral level 6.7 In type IIA, the bridging bronchus originates from the left main bronchus and supplies the right middle and upper lobe.7 Type IIB is defined by a complete absence of the right bronchial tree, with the right lung being supplied by a bridging bronchus from the left mainstem bronchus.7 The right lung is commonly hypoplastic. Type II is more common than type I, and is frequently complicated by long segment tracheal stenosis.8 Management of this type needs to address not only the aberrant pulmonary artery, but also the airway abnormality.8 Both types of slings, but especially type II, are associated with other cardiovascular, pulmonary, and congenital abnormalities.8
The most common presenting symptoms of pulmonary artery slings found in one case series were wheezing (3/8), stridor (2/8), and vomiting/feeding difficulties (2/8).9 Cough and recurrent infections are less common presentations.4,9 All of these symptoms are very common in children; however, in most cases they will have other, most commonly infectious, etiologies. Turner et al showed that the median age for the diagnosis of pulmonary sling is one month with the range being from birth to 3 months.9 However, for the diagnosis to be made expeditiously, a high level of clinical suspicion is required.
Primary means of diagnosis differ from institution to institution; however, the emphasis has shifted away from the barium swallow and angiography to CT scanning, MRI, and echocardiography.10,11 It has been demonstrated that chest X-ray films are the least sensitive for detecting vascular slings, but they are still the first line examination modality, occasionally revealing compression of the trachea and/or mainstem bronchus.9,10 The barium swallow and bronchoscopy have better sensitivity than chest X-ray, but not as high as echocardiography, angiography, and CT.9 Some institutions use echocardiography as the modality of choice, but as with all ultrasonographic studies, the sensitivity is operator dependent.12 Echocardiography is a relatively low cost, noninvasive, radiation-free modality, which may be used as an initial step in pediatric patients undergoing a work-up of vascular anomalies. MRI has been shown to be a viable means of diagnosing pulmonary slings, with the ability to reveal the aberrant vasculature without relying on contrast.13 However, it is associated with higher costs and requires the infants to be sedated.
Surgical treatment of patients with pulmonary slings is still a matter of some controversy. Current areas of debate include the surgical approach. Some centers prefer the median sternotomy, while others use a left thoracotomy.14 Cardiopulmonary bypass and reimplantation versus translocation with distal tracheal resection are also areas of debate.15 Furthermore, a number of surgical approaches are used to treat the tracheal stenosis, which often occurs concurrently.16 These techniques include tracheal resection, pericardial tracheoplasty, cartilage tracheoplasty, slide tracheoplasty, tracheal homograft, and tracheal autograft.11 Gikonyo et al reported a 90% mortality rate in patients with a pulmonary sling that was managed only medically.17
Conclusion
This case highlights the importance of clinical suspicion when determining which diagnostic test is warranted. When reviewing the patient’s complex medical history, with the final diagnosis in mind, it becomes clear that a vascular anomaly should have been high on the differential. However, because the patient had a normal barium swallow, the clinicians pursued other diagnoses. It was not until more than a year later that the possibility of a vascular anomaly was considered again. The delay in diagnosis in this case added significant morbidity, with the patient being hospitalized multiple times subsequent to the swallow study. Finally, without the astute observation of a mild displacement of the trachea on chest radiograph, this diagnosis would have continued to be delayed.
References
- Contro S, Miller RA, White H, Potts WJ. Bronchial obstruction due to pulmonary artery anomalies. I. Vascular sling. Circulation. 1958;17: 418-423.
- Woods RK, Sharp RJ, Holcomb GW 3rd, Snyder CL, Lofland GK, Ashcraft KW, Holder TM. Vascular anomalies and tracheoesophageal compression: A single institution’s 25-year experience. Ann Thorac Surg. 2001;72:434-438; discussion 438-439.
- Arey JB. Malformations of the aorta and aortic arches. In: Arey JB, editor. Cardiovascular pathology in infants and children. 1984:242–244.
- Davies M, Guest PJ. Developmental abnormalities of the great vessels of the thorax and their embryological basis. Br J Radiol. 2003;76;491-502. Review.
- Lincoln JC, Deverall PB, Stark J, et al. Vascular anomalies compressing the oesophagus and trachea. Thorax. 1969;24:295-306.
- Grover FL, Norton JB, Jr, Webb GE, Trinkle JK. Pulmonary sling. Case report and collective review. J Thorac Cardiovasc Surg. 1975;69:295–300.
- Wells TR, Gwinn JL, Landing BH, Stanley P. Reconsideration of the anatomy of sling left pulmonary artery: The association of one form with bridging bronchus and imperforate anus. Anatomic and diagnostic aspects. J Pediatr Surg. 1988;23:892-898.
- Newman B, Cho Y. Left pulmonary artery sling—anatomy and imaging. Semin Ultrasound CT MR. 2010;31:158-170. Review.
- Turner A, Gavel G, Coutts J. Vascular rings—presentation, investigation and outcome. Eur J Pediatr. 2005;104:266-270. Epub 2005 Jan 22.
- Backer CL, Mavroudis C, Rigsby CK, Holinger LD. Trends in vascular ring surgery. J Thorac Cardiovasc Surg. 2005;129:1339-1347.
- Backer CL, Mavroudis C, Dunham ME, Holinger LD. Pulmonary artery sling: Results with median sternotomy, cardiopulmonary bypass, and reimplantation. Ann Thorac Surg. 1999;67:1738-1744; discussion 1744-1745.
- Alboliras ET, Backer CL, Holinger LD, Mavroudis C. Pulmonary artery sling: Diagnostic and management strategy. Pediatrics. 1996;98:530.
- Di Cesare E, Manetta R, Paparoni S, Enrici RM. Pulmonary artery sling diagnosed by magnetic resonance imaging. Magn Reson Imaging. 1997;15:1107-1109.
- Campbell DN, Clarke DR, Lilly JR. Pulmonary artery sling. J Thorac Cardiovasc Surg. 1990;99:942-943.
- Jonas RA, Spevak PJ, McGill T, Castaneda AR. Pulmonary artery sling: Primary repair by tracheal resection in infancy. J Thorac Cardiovasc Surg. 1989;97:548-550.
- Berdon WE, Baker DH, Wung JT, et al. Complete cartilage-ring tracheal stenosis associated with anomalous left pulmonary artery: the ring-sling complex. Radiology. 1984;152:57-64.
- Gikonyo BM, Jue KL, Edwards JE. Pulmonary vascular sling: Report of seven cases and review of the literature. Pediatr Cardiol. 1989;10:81-89. Review.