Radioactive antibodies: Selective targeting and treatment of cancer and other diseases




Dr. Goldenberg is an Experimental Pathologist, Immunologist, and Clinical Investigator, and he is President, Garden State Cancer Center at the Center for Molecular Medicine and Immunology, Belleville, NJ. Dr. Sharkey is a Tumor Biologist and Immunologist, Garden State Cancer Center, Belleville, NJ. Dr. Barbet is a Researcher in the use of antibodies in diagnosis and therapy, Cancer Research Department of Inserm, Nantes, France. Dr. Chatal is a Professor of Nuclear Medicine, and he directs the Nuclear Medicine Departments at the Rene Gauducheau Cancer Center and the University Hospital, Nantes, France.
For several decades, radioactive antibodies have been used as imaging and therapeutic agents, but recent developments raise enthusiasm that diverse imaging agents for various diseases as well as a new generation of cancer therapeutics are more likely than ever before to be used in the near future.
Representative cases
Case 1
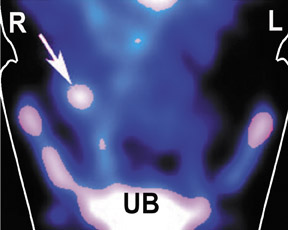
A 16-year-old boy came to the emergency room with abdominal pain, nausea, and vomiting. His doctors decided to use an investigational agent consisting of an antibody fragment that binds to activated neutrophils, a form of white blood cells accumulating in sites of infection, and is labeled with technetium-99m ( 99m Tc), an isotope that is routinely used for nuclear scans that are used as diagnostic imaging studies. An hour after injecting this radioactive antibody, a nuclear scan showed a focus of radioactivity in the right lower abdomen, in the region of the appendix, which suggested that there was a focus of infection (Figure 1) because of an accumulation of neutrophils. He then went to surgery, where an inflamed appendix (7.5 cm in length and 1.5 cm in diameter) was removed. 1
Case 2
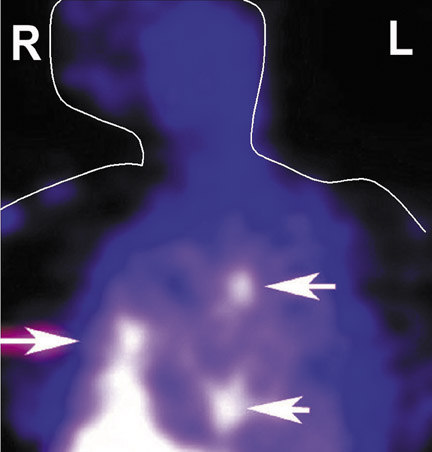
A 26-year-old woman with aquired immunodeficiency disease (AIDS) was suffering from progressive breathing difficulty, congestion, cough, and other symptoms of pneumonia, which was confirmed by chest X-ray. An experimental antibody against Pneumocystis carinii was labeled with 99m Tc and injected into the patient. Twenty-four hours later, her body was scanned with a nuclear camera, which showed accumulation of radioactivity in the right lower lung lobe and in the left lung (arrows), coinciding with the pneumonia seen on the chest X-ray (Figure 2). A bronchial fluid aspirate revealed Pneumocystis organisms, which can infect immunocompromised patients. 2
Case 3
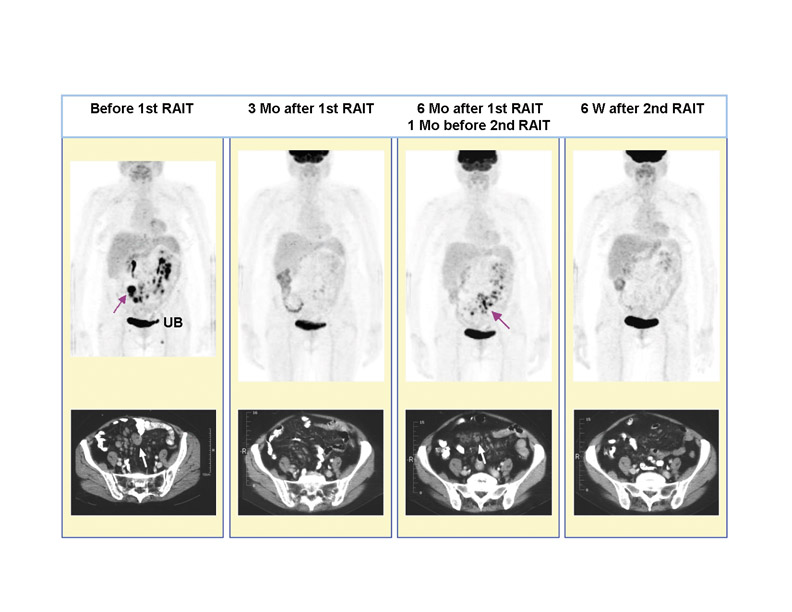
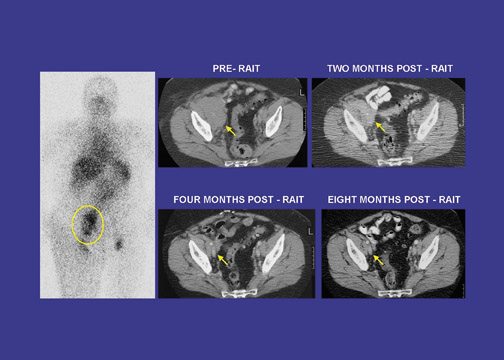
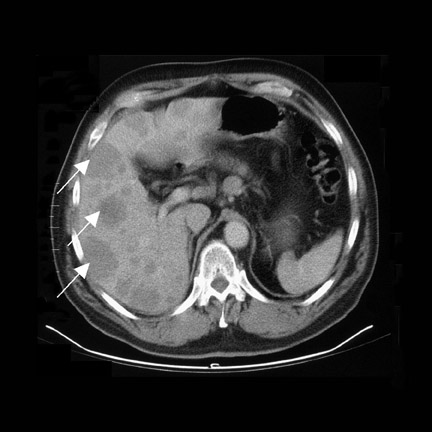
A 67-year-old woman was in relapse with a non-Hodgkin's lymphoma (NHL) after receiving 3 cycles of chemotherapy, irradiation to her abdomen, and 2 immunotherapy treatments with an anti-CD20 antibody. Tumors were found in the abdomen by axial computed tomography (CT) and by positron-emission tomography (PET) scans using fluorine-18 deoxyglucose ( 18 FDG), as shown in Figure 3. Epratuzumab, a humanized antibody against a surface marker of NHL (CD22), was labeled with a beta-emitting radionuclide, yttrium-90 ( 90 Y) and given in weekly fractionated doses over 3 weeks. Three months later, scans showed that all disease had disappeared, but by 6-month follow-up there was recurrence at the same sites in the abdomen. A second treatment course of 3 weekly injections was given, and, again, all disease responded well at the 6 week evaluation. Based on the results of PET scans, the patient enjoyed a complete disappearance of disease for 32 weeks, without experiencing the side effects usually encountered with cytotoxic drugs. 3
These cases are a few examples of how radioactive antibodies are being studied and introduced into medical practice, as more specific diagnostic or therapeutic agents. This article discusses this new technology and its development, current uses, and future prospects.
Antibodies are used to target and image disease
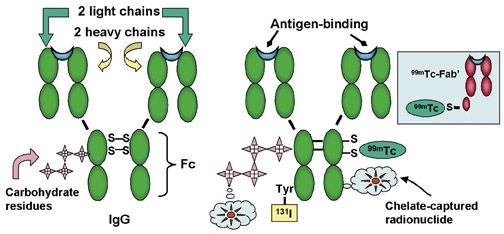
At the beginning of the 20th century, German chemotherapist Paul Ehrlich coined the term Zauberkugeln ( or magic bullets ) to elucidate the idea of using the body's immune system to selectively target receptors on microbes. 4 As early as the 1950s, scientists such as David Pressman, PhD (Sloan-Kettering Cancer Center, New York, NY, and then Roswell Park Cancer Institute, Buffalo, NY) and William Bale, PhD, and Irving Spar, PhD (University of Rochester, NY) investigated this idea of selective tumor localization by making antibodies against receptors on rodent tumors. 5,6 They radiolabeled these antibodies with iodine-131 ( 131 I) and were then able to show selective tumor targeting by measuring the higher radioactivity in the tumor (using a radiation counter) and comparing the measurements with those of normal tissues. Antibodies are glycoproteins produced by lymphocytes to combat infection and foreign organisms with a system that has evolved over millions of years to protect animals from their environment. As part of the immune regulatory system, antibodies are also used as vaccines for treating or preventing infections and are being developed for use against other diseases, including cancers. They are depicted as Y -shaped molecules, where the 2 arms of the Y are the binding sites to receptors, or specific "antigens" that evoke these antibodies and permit them to attach to the same antigens when present in the body (Figure 4). The tail of the antibody recruits immune cells and factors, such as lymphocytes and complement, which join forces with these circulating antibodies to overcome infection or expel foreign invaders. Until the early 1980s, studies using radiolabeled antibodies were confined to the use of radioiodine (eg, 131 I and 125 I), but this field has greatly benefited from the contributions of many chemists, such as Donald Hnatowich, PhD (University of Massachusetts, Worcester), Claude Meares, PhD (University of California at Davis), and Otto Gansow, PhD and Martin Brechbiel, PhD (National Institutes of Health, Bethesda, MD), who developed chelates, or metal-binding chemicals, that could be coupled to antibodies, which greatly expanded the number and types of radionuclides that could be attached to antibodies for detection or therapy, such as indium-111 (111In) and 90Y. 7-10
Once it was shown that antibodies produced in animals (so-called polyclonal antibodies) could distinguish tumors growing in rats from their normal tissues, the challenge was to translate this to use in cancer patients. Before embarking on clinical studies, it was necessary to first reproduce these findings in an animal model growing human cancer transplants, using antibodies against a human tumor antigen that could be localized in these tumors. At that time, none of the immunodeficient mouse models that are currently available for use by researchers were available. In 1972, we were fortunate to have observed that a human intestinal tumor growing in the cheek pouches of hamsters expressed carcinoembryonic antigen (CEA). 11 Carcinoembryonic antigen was first described in 1965 by Phil Gold, MD, PhD, and Sam Freedman, DSc, MD, at McGill University in Montreal, as an intestinal-specific human cancer antigen. 12 We reasoned that this would be a selective target for delivering radioactivity to intestinal tumors when antibodies against CEA were tagged with radionuclides. In 1973, with F. James Primus, PhD (Hoffman-LaRoche, Nutley, NJ and University of Kentucky, Lexington, KY) and Hans J. Hansen, PhD (Hoffman-LaRoche), we showed, for the first time, that human tumors grown in hamsters could, in fact, localize radioactive antibodies. 13,14 This set the stage for clinical studies and--after developing and purifying clinical-grade antibodies to CEA, and labeling them with 131 I (an isotope commonly used in humans to detect and treat thyroid tumors that selectively absorb iodine)--we embarked to prove that cancers in patients could be detected and actually localized with radioactive antibodies. At this time, one concern was that CEA, which also circulates in the blood of these cancer patients, would bind the injected antibodies and prevent it from reaching the tumors. We disproved this dogma when, in the first patients reported with this new technology in 1978, we visualized CEA-producing tumors and failed where there were tumors devoid of CEA. 15 Equally important was the observation that cancers other than intestinal tumors (including breast, uterine, and lung tumors) also produced sufficient CEA to be revealed by these radioactive antibodies. This observation gave birth to the use of radioactive antibodies for disease detection, which the authors termed radioimmunodetection (Figure 5), or for therapy (termed radioimmunotherapy ). 16
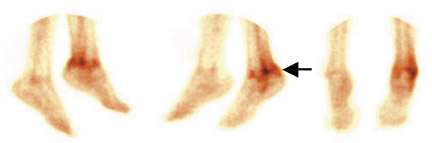
In the ensuing years, more refined monoclonal antibodies that could be mass-produced evolved, isotopes other than 131 I were attached to antibodies, and antibody forms were changed to make them smaller and more compatible for use in humans. 17,18 Once this principle for locating and visualizing CEA-expressing cancers was proven, radioimmunodetection was applied to other cancers and diseases, even targeting antigens expressed in clots, heart infarcts, and infections. In fact, we and others showed that small antibody fragments against a receptor on activated white blood cells (neutrophils) would accumulate at sites of infection and could be tagged with a common imaging isotope, 99m Tc. This could then reveal sites of infection (such as bone infection), osteomyelitis (Figure 6), or appendicitis (Figure 1), thus providing rapid and more selective diagnostic information. 1,19-21
We also showed that immunocompromised patients with a rare infection, Pneumocystis carinii pneumonia, could have their infection visualized with radioactive antibodies developed by our colleague Peter Walzer, MD (University of Cincinnati, OH) against this organism (Figure 2). 2 More recently, Arturo Casadevall, MD, PhD, and Ekaterina Dadachova, PhD, at the Albert Einstein College of Medicine (Bronx, NY) have shown that they could localize and treat fungal infections in mice, such as Crytopoccus and Histoplasma , with radioactive antibodies, thus anticipating a new method of treating serious and resistant infections. 22,23 Several antibody-based imaging products have been commercialized, based on either intact murine immunoglobulin or Fab' fragments, which are much smaller and, therefore, less immunogenic and more rapid targeting agents. 24,25 Nevertheless, the adoption of these nuclear imaging methods was slow, since the low contrast of the images required considerable experience by the nuclear physician, and other imaging agents, although less disease-specific, were being introduced.
New classes of imaging agents
Antibodies, and smaller fragments thereof, have been developed against many different antigens associated with disease in order to specifically image these diseases. 25,26 These have included antibodies for imaging colorectal cancer (eg, satumomab pendetide, 27 arcitumomab 24 ), prostate cancer (eg, capromab pendetide 28-30 ) NHL, 31 liver cancer, 32 myocardial infarcts (imciromab penlefate 33-35 ), infection and inflammation (eg, sulesomab, fanolesomab 36-38 ), specific infections, 2 and clots, 39 to mention a few that are now available or in development. Either the intact immunoglobulin, its Fab' fragment, or even smaller antigen-binding Fv units can be used when radiolabeled. The most popular imaging isotopes are 99m Tc and 111 In, although recent intensive research has included 124 I (iodine-124), 68 Ga (gallium-68), 64 Cu (copper-64), and 18 F (fluorine-18) as PET agents for development of ImmunoPET, which provides the specificity of antibodies and the sensitivity, or improved image resolution, of PET isotopes. 40 Steven M. Larson, MD, Chief of Nuclear Medicine at the Memorial Sloan-Kettering Cancer Center (New York, NY) was one of the first to show the targeting of tumors with antibodies labeled with the positron emitter 124 I. 41
With the data showing that radioactive antibodies could selectively target and image diseases that express the specific antigens, we and other researchers focused on applying this technology to radioimmunotherapy (RAIT) of cancer.
Radioimmunotherapy of cancer
Cancer treatment is dominated by 3 therapeutic procedures--surgery, radiation, and chemotherapy. If cancer is detected early, when it is localized, surgery and/or radiation therapy could be adequate to control or eradicate disease. Radiation therapy typically involves focusing an external beam of radiation to the specific area of the body that has the tumor mass (Figure 7). Alternatively, radioactive "seeds," which will emit local radiation within the region, can be implanted into the tumor area (brachytherapy). Unfortunately, cancer is frequently detected after it has spread beyond the original tumor site to other tissues and organs (metastasis), so that a systemic therapy is then required. Chemotherapy is the primary option for systemic therapy, although antibodies have recently been introduced as another treatment modality (immunotherapy), especially used in combination with anticancer drugs. The same kind of antibodies, however, can be used to carry radioactivity throughout the body and to deliver toxic radiation more selectively to cancer cells (Figure 7).
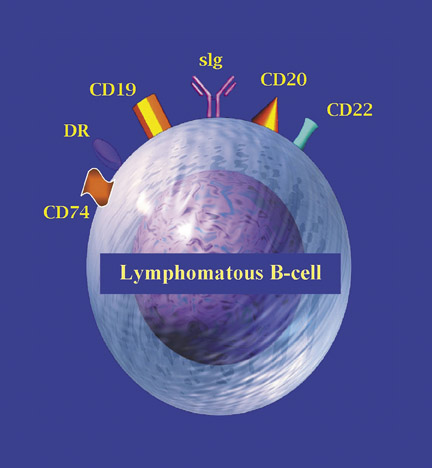
Although considerable research was undertaken in solid cancers (such as intestinal, breast, ovarian, and other carcinomas), the first true evidence of success came from the use of radiolabeled antibodies against marker antigens of hematologic cancers, such as NHL, a cancer of the lymphatic system. In the past 30 years, the U.S. incidence of NHL has increased significantly, becoming the fifth most common and sixth most fatal cancer in the United States, with approximately 270,000 Americans currently affected. An estimated 60,000 new U.S. cases of NHL will be diagnosed this year, and only approximately half of these cases will be cured. Normal and malignant lymphocytes express various receptors that are targeted by monoclonal antibodies, which are sometimes referred to as cluster designation (CD) types (eg, CD19, CD20, CD22; Figure 8), as well as human leukocyte transplantation antigen, HLA-DR.
Gerald DeNardo, MD, and Sally DeNardo, MD, at the University of California at Davis, were among the first to show successful therapy of NHL with radioactive antibodies against HLA-DR. 41 Oliver Press, MD, PhD, at the Fred Hutchinson Cancer Research Center (Seattle, WA) pioneered the use of very high doses of radioiodinated anti-CD20 antibodies under protection of bone marrow transplantation (to overcome the radiation toxicity to the bone marrow) for very effective NHL treatment. He and his collegues have even shown the feasibility and advantage of combining RAIT with chemotherapy. 42-48 Indeed, targeting NHL with murine antibodies against CD19 and CD20 (not conjugated with isotopes) was the subject of clinical studies for more than 20 years, but the development of a human/mouse chimeric antibody against CD20, called rituximab , revolutionized the therapy of NHL and spurred further developments in monoclonal antibodies for other cancers. 49 Rituximab induces remissions in roughly half of patients with the indolent form of NHL, and, although it is not curative by itself, it can improve the effects of standard chemotherapy and increase the number of cases of complete disappearance of tumor in many NHL types. Particularly intriguing is its relative lack of major toxicities, in contrast to standard anticancer drugs that often cause suppression of blood cells, mouth ulcers, and hair loss.
Thomas Witzig, MD, and Gregory Wiseman, MD (Mayo Clinic, Rochester, MN), Susan Knox, MD, PhD (Stanford University, Stanford, CA), and Christine White, MD (IDEC Pharmaceutical, Biogen Idec, Inc., Cambridge, MA) were instrumental in the clinical investigation that showed the superiority of a radiolabeled antibody treatment compared with the unlabeled antibody, which led the U.S. Food and Drug Administration (FDA) to approve the first radioimmunotherapeutic agent for cancer ( 90 Y-ibritumomab tiuxetan). 50,51 A combination of rituximab with a therapeutic radiometal, 90 Y-ibritumomab tiuxetan is 90 Y attached to the mouse antibody that was commercialized in 2002 and is used to treat the indolent form of NHL. 52
A second radioimmunotherapeutic against CD20, 131 I-tositumomab, for the treatment of NHL, was also approved for U.S. use in 2003, and it involves a different radioisotope, iodine-131 ( 131 I). 53,54 Although this is a mouse antibody (which has a different form of radioactivity), both radiolabeled products appear to have similar efficacy results, which clearly show a significant improvement in shrinking tumors as compared with their non-radioactive forms. 50,55 While clinical data showing that these treatments significantly increase survival are lacking, highly durable, complete responses have been reported. 56-58 Although their applications to date have been restricted to patients who have failed to respond to other forms of therapy, results reported by Mark S. Kaminski, MD, and Richard Wahl, MD, of the University of Michigan showed impressive responses as a first treatment in patients with the indolent form of NHL. 54 One concern raised in the use of these types of treatment has been the possibility of future development of myelodysplastic syndrome/acute myeloid leukemia. 52 While a low incidence was reported in patients previously treated with chemotherapy before receiving a radiolabeled antibody, an evaluation has shown that none of the 76 patients given 131 I-tositumomab as a first-line treatment has developed these conditions during a 5-year follow-up. 59 In addition, studies have shown that patients are able to receive subsequent chemotherapy or a second radioantibody treatment successfully after receiving radioimmunotherapy, which further supports integrating these treatments into a standard therapeutic regimen. 60,61
In addition to targeting CD20, we are studying a humanized antibody (less mouse protein) against CD22, another marker for NHL, labeled with 90 Y, as a fractionated, repeated therapy in indolent and aggressive forms of NHL (Figures 3 and 9). Patients who may become resistant to a CD20 antibody therapy could have this second therapeutic option. 62 In addition to the successful treatment of lymphoma, David A. Scheinberg, MD, PhD, and Joseph G. Jurcic, MD (Memorial Sloan-Kettering Cancer Center, NY, NY) have the lead in treating acute myeloid leukemia with radiolabeled antibodies. 63,64
Numerous antibodies against solid tumors have been evaluated for therapeutic applications. 65 Although antibodies to CEA were among the first to be studied in a variety of cancers, Jeffrey Schlom, PhD, and David Colcher, PhD (National Cancer Institute, Bethesda, MD) developed the B72.3 against a tumor-associated glycoprotein and its subsequent second-generation monoclonal antibodies (CC49) and other molecularly engineered products that have been widely studied in colorectal, breast, and other cancers, not only for imaging and therapy, 66-69 but also for use with an intraoperative probe. 70 In contrast to the experience in NHL and other tumors of the blood system, radiolabeled antibodies have been less effective when used to treat solid tumors, such as colorectal, breast, lung, ovarian, and other carcinomas. 65 The same radiation dose that shrinks lymphoma is less effective in these other cancers, so it is concluded that lymphomas are more sensitive to radiation, but the basis for this radiosensitivity is poorly understood. Such factors as increased vascularization, less fibrous connective tissue, and higher number of tumor cells more diffusely spread in lymphomas are just some of the reasons speculated for their responding so well. It could also be that the direct effects of radiation are supplemented by antitumor effects of the antibodies themselves in these lymphomas, or that patients more readily invoke immune responses to lymphoma than they do to solid tumors. Further, the dose delivered by giving the highest tolerated amount of radioactivity attached to an antibody is less likely to show tumor shrinkage in larger tumors (eg, >5 cm) than in smaller ones, especially when there are small clusters of cancer cells. For example, giving RAIT to animals during an early phase of metastasis can affect survival significantly, even resulting in cures (Figure 10). 71 Initial clinical studies reported by Thomas M. Behr, MD, and Wolfgang Becker, MD (University of Göttingen, Germany) support this preclinical finding. 72 More recently, a clinical trial performed under the direction of Torsten Liersch, MD, with the other investigators from the University of Göttingen, reported that colorectal cancer patients having resection of their liver metastasis who received postoperative radioimmunotherapy with 131 I-anti-CEA humanized antibodies did significantly better, with doubling survival time, than did a contemporaneous control group of patients who did not receive radioimmunotherapy. 73
Also, improved methods for increasing the radiation dose delivered selectively to tumors need to be developed. One obvious approach is to deliver the radioactivity locally, such as in regional or intratumoral applications. Indeed, both of these options have shown encouraging results clinically, particularly when delivering the radiolabeled antibody into surgical reservoirs for treating malignant brain tumors, as has been described by Pietro Riva, MD, in Cesena, Italy, Giovanni Paganelli, MD, at the European Institute of Cancer in Milan, Italy, and by Darrell D. Bigner, MD, PhD, Henry S. Friedman, MD, and Michael R. Zalutsky, PhD, at Duke University (Durham, NC). Others, such as Ruby M. Meredith, MD, PhD, at the University of Alabama, in Birmingham, have examined the delivery of radioactive antibodies directly into the abdominal cavity. 74-80
Table 1 summarizes many of the radio-labeled antibodies in clinical research, showing that radioimmunotherapy is being actively investigated in a wide variety of cancers using antibodies to different tumor-associated antigens. While there are currently only 2 therapeutic agents approved for human use, it is important to understand the complexity and diversity involved in these types of investigations. Selection of appropriate markers for specific targeting is a major challenge, while selecting the appropriate radionuclide, method for radiolabeling, and even the form of antibody used in the targeting procedure are all variables that need to be carefully considered for the particular clinical situation.
Table 2 is a partial listing of some of the radionuclides used for radioimmunotherapy and highlights just some of issues that investigators must consider while evaluating prospective radionuclides. Most are beta-emitters, and some of these travel several millimeters in tissues, a distance that extends the range where the radionuclide could potentially damage cells at a length equal to the diameter of as many as 50 to even 200 cells. Beta-emitters are generally preferred for killing visible tumors, but their long range can also cause collateral damage to adjacent normal tissues. Antibodies radiolabeled with beta-emitters that stay in the blood for several days will damage the blood-forming cells found in the bone marrow, which lowers blood counts in treated patients and limits the amount of radioactivity that can be given. Alpha-emitters, while they have a relatively short range (eg, up to 10 cell diameters), are much more potent cell-killers than beta-emitters. Because of their short range, alpha-emitters have most commonly been used to treat blood cancers, such as leukemia, because they frequently are found as single cells or small cell clusters. These cancers are readily accessible to the radiolabeled antibody injected in the bloodstream. This is important, since many of the alpha-emitters decay in a short period of time and, therefore, must be quick to selectively localize the tumor cells. Alpha-emitters are also being used to treat cancers regionally, such as ascites and peritoneal implants in ovarian cancer, and brain cancers. Low-energy electron emitters have an even smaller range than alpha-emitters and are capable of killing mostly single cells, which reduces damage to neighboring normal cells. However, a highly efficient means of delivering these types of radionuclides is required in order to increase the likelihood of specific cell killing. By targeting radionuclides with different path lengths, it may be possible to effectively kill visible, yet small, solid tumors, while following with a radionuclide of shorter path-length to seek out microscopic deposits of tumors that might not be as effectively killed by a targeted beta-emitter.
David A. Scheinberg, MD, PhD, at Memorial Sloan-Kettering Cancer Center and M. Jules Mattes, PhD, at the Garden State Cancer Center at the Center for Molecular Medicine and Immunology (Belleville, NJ), have found that very short-range and potent alpha-emitters and low-energy electrons can be used with antibodies to selectively kill small clusters of human cancer cells in animal models, thus expanding the choice of therapeutic radionuclides beyond the more conventional beta-emitters ( 131 I and 90 Y) with longer path lengths. 63,81,82
Restructuring antibodies by molecuar engineering
The art of antibody preparation has been revolutionized by molecular biology. The development of monoclonal antibodies from mouse cells by Georges Köhler, PhD, and Cesar Milstein, PhD (the University of Cambridge, England) can be credited as one of the most important developments in molecular immunology. In 1984, they shared the Nobel Prize for Physiology and Medicine just 9 years after publishing their seminal paper on hybridoma technology. 83 Just as polyclonal antibodies were quickly replaced when techniques for monoclonal antibody production were developed, murine monoclonal antibodies are being superseded with other molecular constructs that more closely mimic human antibodies. Initially limited to only 3 forms of antibodies (eg, intact IgG, F(ab') 2 , and Fab'), investigators now have an expanding repertoire of antibody forms with variable numbers of antigen-binding sites and with different molecular sizes and shapes. Even their binding affinity, immune effector activity, and the manner in which they are cleared from the blood can be modified. As their basis, many of these constructs use molecules known as single-chain variable fragments (scFv), which consist of the variable-light (V L ) and variable-heavy (V H ) chain regions of an antibody that contain the 3 complementarity-determining regions (CDRs) responsible for antigen binding. Brought together by the insertion of a variable-length amino acid linker, these 2 chains fold to form a 25,000 dalton (25 kD) molecular size molecule with 1 binding site (about one sixth the size of an intact, natural, IgG antibody with 2 binding arms. But, as the amino acid linker length is changed, these molecules can self-anneal to form diabodies, triabodies, and even tetrabodies (2, 3, and 4 binding sites, respectively). Still other forms, such as sc(Fv) 2 , [sc(Fv) 2 ] 2 , scFv-Fc, and minibodies, have been described (Figure 11).
Targeting studies with these new molecular antibody forms have suggested that some are preferable over others for either imaging or therapy. The smallest clear very rapidly from the blood, which improves tumor/blood ratios as compared with those observed with an intact radiolabeled IgG. However, the blood clearance of these molecules can be so fast that the tumor uptake is also correspondingly much lower than that achieved with the larger constructs. In addition, the selection of a particular form might also affect the choice of radionuclide used. Antibody versions that are smaller than ~60,000 are directly filtered from the blood into the kidneys. However, in contrast to conjugates with radiometals for which kidney uptake can be high, renal retention by radioiodinated constructs is reduced considerably. While the choice of an antibody form and a radionuclide may depend on the intended application, there have been several advances, primarily in preclinical testing, of improved targeting and therapy with several of these reengineered constructs, such as has been reported by Anna Wu, PhD, 84 from the University of California at Los Angeles, and Richard H. Begent, MD, 85 at the Royal Free Hospital and University College Medical School in London, England.
Toward new imaging and therapy methods: Pretargeting
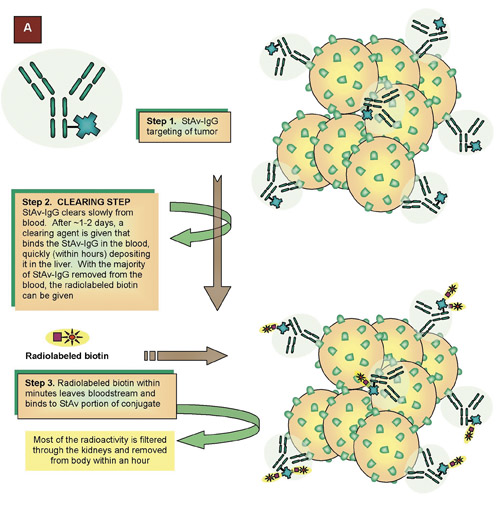
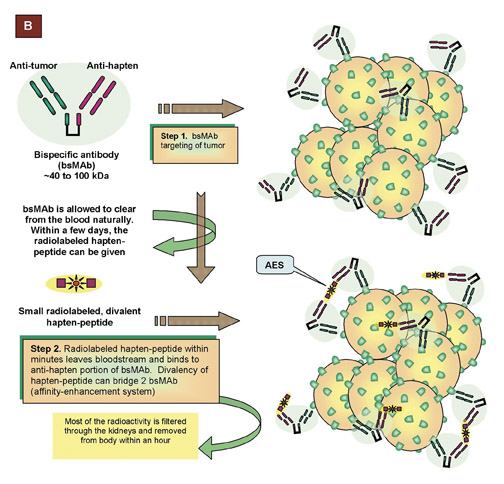
An alternative to using directly radiolabeled antibodies is a procedure known as pretargeting, a multistep process by which the radionuclide is attached to a small compound that is cleared very rapidly (in minutes) from the blood. The antibody has dual specificity, with 1 arm binding to the target antigen and the other to a recognition unit on a small radiolabeled compound. David Goodwin, MD, of Stanford University and VA Hospital, Palo Alto, CA introduced this concept more than 15 years ago. 86,87 The 2 most common pretargeting procedures utilize a strepta-vidin-biotin recognition system (advanced by Donald Hnatowich, PhD, of the University of Massachusetts, Worcester; Giovanni Paganelli, MD, European Institute of Cancer, Milan, Italy; and NeoRx's John Reno, PhD, and Alan Fritzberg, PhD, Seattle WA), or a bispecific monoclonal antibody (bsMAb)-hapten model (Figure 12), as being pursued by our group and that of Otto Boerman, PhD, and Frans Corstens, MD, and their colleagues at the University Nijmegen (Netherlands) after the initial studies of Michael Delaage, PhD, and co-workers in Marseilles, France. 88-95 In our view, pretargeting methods utilizing bsMAb should have an adantage over those that involve streptavidin, since the immunogenicity of bsMAbs can be reduced or eliminated by the use of humanized bsMAbs, whereas host antibodies against streptavidin have occurred at high frequency. Bispecific antibodies also can be generated as small, antigen-binding variable domains (Fv) that improve the pharmacokinetic properties (localization and clearance) of the primary targeting agent, while also binding to a versatile, bivalent, peptide-hapten carrying an imaging or therapeutic radionuclide. 93-95
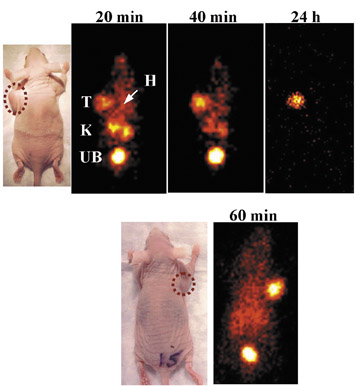
This pretargeting strategy (Figure 12B) also appears to be promising for improved disease imaging, since it relies on the principle of reduced background radioactivity by allowing time for clearance of nontargeted bsMAb from the body before the peptide bearing a radioactive signal is given for disease localization. In the case of cancer, we are finding that human colonic cancer grown in immunedeficient mice can be visualized within 20 minutes of injecting the imag-ing signal (Figure 13) and that at 24 hours, ratios of uptake in tumor versus blood can exceed 100, which is 40-fold better then any directly labeled antibody or antibody fragment. 96 This pretargeting procedure, when used in combination with an 124 Ilabeled hapten-peptide for ImmunoPET imaging, provides a durable, strong signal in the tumor with much lower uptake in the normal tissues than 18 FDG, thereby affording less ambiguous tumor targeting. 97 Recently, our group has shown the localization of a human colon cancer growing as discrete ≤0.3-mm colonies in the lungs of nude mice with the same pretargeting ImmunoPET procedure (Figure 14). 18 FDG was unable to disclose these very small lesions, which further illustrates how the enhanced sensitivity of this pretargeting procedure, along with its high specificity, could assist in the detection of cancer. Since antibodies exist for targeting various cancers, infections, myocardial damage, clots, emboli, and atherosclerotic plaques, a new generation of imaging and diagnostic agents for cancer and diverse diseases may now be developed using these antibodies combined with radioisotopes, along with the signal from various contrast agents for magnetic resonance imaging and ultrasonography. The challenge is to choose the most specific antibodies for creating bispecific constructs and to load the signal-generating contrast agent in sufficient quantity for image resolution with current cameras. The signal-bearing peptides can be devised to bear many end-groups as attachment sites and can be of virtually any size, so that such loads may be feasible, possibly also for drug delivery.
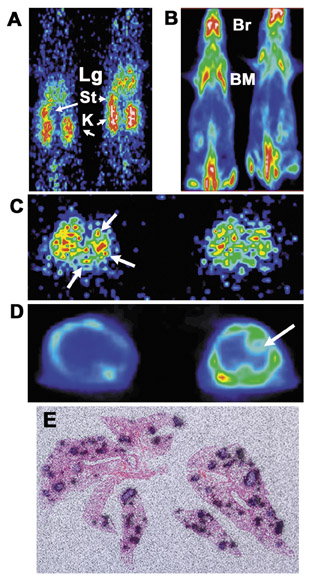
Pretargeting methods have also been shown to be more effective in selectively delivering radionuclides to tumors for therapy. 98-100 Using a bispecific antibody construct composed of an antibody fragment against CEA and another antibody fragment against a metallic compound that is bound to radioactive iodine ( 131 I), clinical trials that are being performed by us in France are showing excellent tumor imaging (Figure 15) and initial evidence that the radiation dose to the tumors, as compared with a directly radiolabeled antibody, can be improved. 101 Studies are in progress to assess whether these doses are sufficient to control tumor growth, but the advantage of using totally humanized, nonimmunogenic reagents is to permit repeated dosing, which is likely to be needed. Early results in patients with lung, colorectal, and medullary thyroid cancers that produce CEA indicate a slowing of tumor growth. For example, a retrospective analysis of medullary thyroid cancer patients treated with an anti-CEA bispecific antibody pretargeting procedure and an 131 I-labeled hapten-peptide has revealed a subset of patients in whom a significant survival advantage occurs. 102 These encouraging findings suggest that more laboratory research should be conducted to determine the best reagents, appropropriate treatment regimens, and specific tumor types that may be the best candidates for this form of radioimmunotherapy.
We have found that having an antibody with 2 binding arms against the cancer antigen CEA and 1 binding arm against the peptide that carries the therapeutic isotope, 90 Y, appears to be optimal. 103,104 In a study that compared the efficacy of a CEA antibody directly labeled with 90 Y with the 2-step pretargeting involving 90 Y attached to the peptide, a clear advantage of pretargeting was shown; 30% of the animals that had initially had well-developed tumors were cured, while none were cured in the control group receiving directly radiolabeled antibody. 98,99 This advantage was again confirmed in another model of human NHL growing in immunodeficient mice, where pretargeting involving antibodies against CD20 receptors on the tumor cells again showed better results in controlling tumor growth than did a directly radiolabeled antibody, which is similar to the two CD20 antibody products currently approved for NHL radioimmunotherapy. 104
Bispecific antibody pretargeting-either for improved cancer imaging or therapy-is still in the early phase of development, despite already showing evidence of safety and efficacy in animal and human studies, because many questions still need to be resolved: What is the most effective form of the bispecific antibody construct? How high does it need to be dosed and how often? Which peptide and isotope are preferred? Are there any long-term toxicities when high and/or repeated therapeutic doses are given? Other important matters involve stability and production properties, if these are to become new candidates for commercial use. Since antibodies can be made against other markers of disease, as discussed above, what will be the most important disease targets for applying this new technology? Could this also be expanded to the more selective delivery of other agents, such as drugs, toxins, hormones, enzymes, vaccines, or even genes as a next generation for gene therapy or diagnostic imaging agents? We believe that these studies with radioactive antibodies have set the stage for asking such questions and for stimulating these other applications.
