Radiological Case: Nonsurgical pneumoperitoneum
Images




CASE SUMMARY
A 56-year-old Caucasian female with multiple falls and ataxia was admitted for exacerbation of multiple sclerosis and placed on high-dose steroid therapy. The patient’s past medical history was significant for polycystic kidney disease and a kidney transplant, for which she took cyclosporine as an immunosuppressant. There was no recent history of surgery or endoscopic examination.
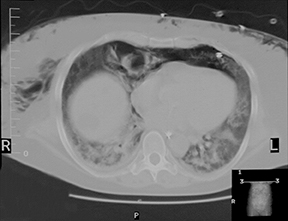
Within 12 hours of admission, the patient developed severe hypoxemia requiring mechanical ventilation. Standard ventilatory therapy was inadequate and the patient required high levels of positive end-expiratory pressure with peak airway pressures exceeding 50 mmHg. On the fifth day of admission, the patient developed crepitus over the anterior chest wall. Chest x-ray confirmed bilateral pneumothoraces, pneumomediastinum, and subcutaneous emphysema (Figure 1). Bilateral chest tubes were placed. The patient’s clinical signs began to deteriorate with radiographic signs of acute respiratory distress syndrome (ARDS).
The following day, a physical exam revealed abdominal distention, with no rigidity. Pneumoperitoneum and an unknown source of sepsis prompted an exploratory laparotomy. Upon entering the peritoneum a large egress of air was noted. There was no evidence of intraperitoneal fluid. Exploration of all of the hollow organs showed no signs of perforation or signs of inflammatory reaction. Examination of the diaphragm showed no obvious defects. The patient tolerated the procedure well and was returned to the intensive care unit. Over the following days, the patient’s clinical course declined until elective termination of care secondary to cardiorespiratory collapse.
IMAGING FINDINGS
Radiologic supine film of the abdomen demonstrated free intraperitoneal air. A computed tomography (CT) scan of the abdomen and pelvis with oral and intravenous contrast revealed a large amount of intraperitoneal air with extensive subcutaneous air of the abdominal and chest wall. There was no intra-abdominal fluid or extravasation of contrast from the gastrointestinal tract. There was no evidence of retroperitoneal air or pneumotosis coli (Figure 2).
DIAGNOSIS
Pneumoperitoneum.
Classic surgical and emergency teaching suggests that pneumoperitoneum is diagnostic for a perforated hollow viscus and is therefore a surgical emergency. This dictum, however, is accurate in only 85% to 95% of cases.1 Although infrequent, there are nonsurgical causes of pneumoperitoneum (Table 1).. Presented here is a case of nonsurgical pneumoperitoneum that resulted from mechanical ventilation-induced barotrauma.
DISCUSSION
Pneumoperitoneum from cardiopulmonary resuscitation, mechanical ventilation, and tracheal rupture has been recognized in literature since the early 1940’s.1 Current retrospective analysis estimates the incidence of mechanical ventilation-induced pneumoperitoneum to be as high as 7%.2 The mechanism by which air in the thorax traverses the abdominal cavity is still under investigation. However, there are 3 proposed pathways (Table 2).
In a nonacute setting, when signs of sepsis or peritonitis are absent and when clinical suspicion for a ruptured viscus is low, management strategies for pneumoperitoneum can be conservative. Repeat follow-up radiologic examination can be performed and the patient monitored until the pneumoperitoneum resolves. However, as the largest cause of nonsurgical pneumoperitoneum is mechanical ventilation;1 the majority of patients are not only intubated but are usually sedated. Furthermore, they frequently have confounding factors, such as high-dose steroid use or immunosuppression, which can obscure both the laboratory and clinical signs of perforation leading to peritonitis and sepsis.3 These critically ill patients are the least tolerant of surgical intervention. Therefore, given the possibility of inaccuracy that comes with the clinical exam in a mechanically ventilated patient, alternative means of detecting the surgical emergency of pneumoperitoneum must be considered.
Although pneumoperitoneum is most commonly discovered on radiography, CT has a higher sensitivity for demonstrating the presence of free intraperitoneal gas versus conventional radiography, 92% vs. 74% respectively.4 By monitoring for CT findings specific for intrabdominal hollow-organ perforation as the cause of free intraperitoneal air (Table 3), CT contrast examination has been shown to be 100% sensitive in the detection of perforation of the stomach, duodenum, and colon.4
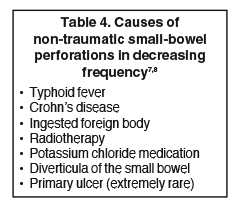
However, there still remains great debate over the accuracy of CT for identifying small bowel perforations. Numerous case reports describe the difficulty in the radiographic diagnosis of small bowel perforations with studies showing the accuracy of CT-detected small bowel perforation to range from 33% to 94%.5,6 Physiologically, this discrepancy can be explained as the small bowel does not normally contain much air or liquid and is collapsed upon itself, making CT evaluation difficult. However, excluding obstruction as the etiology, isolated small-bowel perforation is very rare (Table 4).7,8 Crohn’s disease is the most common cause in the Western world of free perforation of the small intestine with an incidence of approximately 1% of Crohn’s patients.9,10 If CT evaluation fails to demonstrate adequate signs of perforation, and suspicion remains high for small bowel perforation, a proper history and further laboratory evaluation of the patient can aid in differentiating the possible causes.7,8
Once pneumoperitoneum is discovered on CT, the search for a source of perforation ensues. The most accurate evidence of an oral-contrast CT for perforation of a hollow abdominal organ requiring surgery is the extravasation of contrast and/or free intra-abdominal fluid, not the presence of intraperitoneal air.4 In cases of pneumoperitoneum with CT findings of intra-peritoneal fluid or extravasation of contrast, the decision for operative intervention is clear. As seen in this case, however, only intraperitoneal air without additional evidence of perforation should lead the physician to consider nonsurgical causes of pneumoperitoneum. Factors such as patient history, previous surgeries, concurrent medical conditions, and course of events must be evaluated prior to determining the cause of free air and subsequently the need for operative intervention versus observation and supportive care. In the sole presence of intraperitoneal air, concurrent CT findings of ventilation-induced barotrauma, such as pneumothoraces, pneumomediastinum, or subcutaneous emphysema, strengthens the argument for observation.3 Although there is currently no predictive correlation between the incidence of computed tomography findings of barotrauma and the occurrence of nonsurgical pneumoperitoneum, these findings should be used as a retrospective tool to direct the physician to consider a nonsurgical cause of the pneumoperitoneum.
CONCLUSION
As demonstrated in this case, CT-detected pneumoperitoneum is not pathognomonic of bowel perforation. In patients where the clinical exam is limited due to mechanical ventilation, oral contrast CT evaluation can be used as the primary diagnostic tool in assessing the surgical nature of pneumoperitoneum. This information may save a patient from an unnecessary laparotomy. Observation in the setting of pneumoperitoneum and sepsis should not be considered a conservative approach; rather it is an aggressive approach that must be used with discretion.
REFERENCES
- Mularski RA, Sippel JM, Osborne ML. Pneumoperitoneum: A review of nonsurgical causes. Crit Care Med. 2000;28:2638-2644.
- Henry RE, Ali N, Banks T, Dais KA, Gooray DA. Pneumoperitoneum associated with mechanical ventilation. J Natl Med Assoc. 1986;78: 539-541.
- Balthazar EJ, Moore SL. CT evaluation of infra-diaphragmatic air in patients treated with mechanically assisted ventilation: A potential source of error. AJR Am J Roentgenol. 1996;167:731-734.
- Catalano O. Computed tomography in the study of gastrointestinal perforation. Radiolo Med (Torino). 1996;91:247-252.
- Grassi R, Pinto A, Rossi G, Rotondo A. Conventional plain-film radiology, ultrasonography and CT in jejuno-ileal perforation. Acta Radiol.1998;39:52-56.
- Scherck J, Shatney C, Sensaki K, Selivanov V. The accuracy of computed tomography in the diagnosis of blunt small-bowel perforation. Am J Surg. 1994;168:670-675.
- Leijonmarck CE, Fenyö G, Räf L. Nontraumatic perforation of the small intestine. Acta Chir Scand. 1984;150:405-411.
- Jung F, Reiss D, Sibilly A. Acute primary ulcer of the small intestine. Present concepts. Sem Hop. 1982;58:1349-1353. [in French]
- Greenstein AJ, Mann D, Sachar DB, Aufses AH Jr. Free perforation in Crohn’s disease: A survey of 99 cases. Am J. Gastroenterol. 1985;80: 682-689.
- Greenstein AJ, Sachar DB, Mann D, et al. Spontaneous free perforation and perforated abscess in 30 patients with Crohn’s disease. Ann Surg. 1987;205:72-76.
Citation
Radiological Case: Nonsurgical pneumoperitoneum. Appl Radiol.
September 5, 2014