Thymic carcinoma
Images
CASE SUMMARY
A 57-year-old male singer with no significant past medical history, who presented to the emergency room with pleuritic chest pain with extreme deep breathing, first noticed while performing on stage. Physical examination was unremarkable. Patient vitals were within normal limits with a pulse oximetry of 93 percent on room air. Labs were as follows: WBC 39.1, H/H 9.9/30.8, Platelets 395. All other lab values including electrolytes were within normal limits.
IMAGING FINDINGS
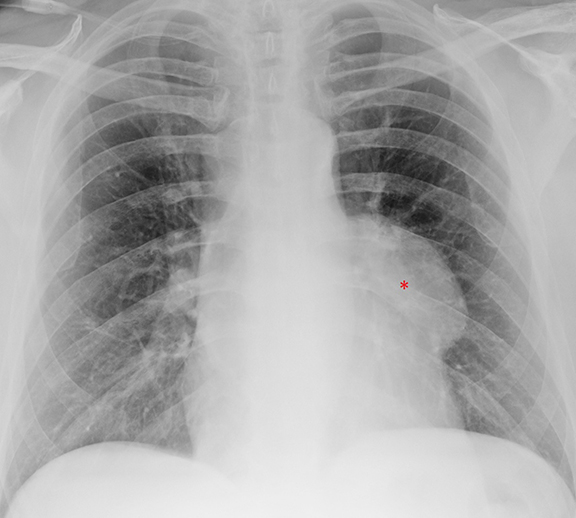
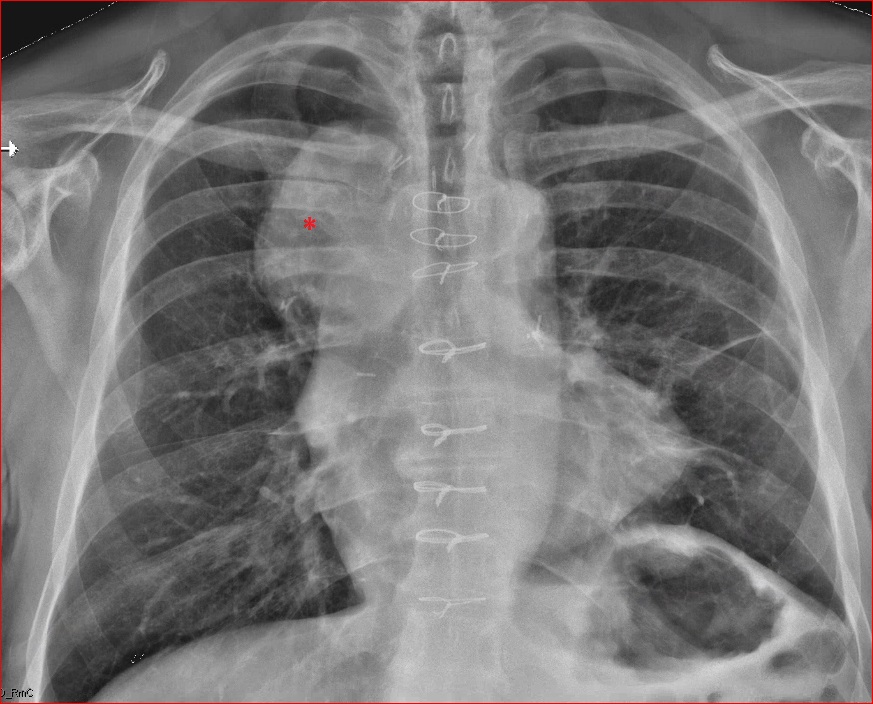
A chest radiograph (Figure 1) demonstrated an abnormal left sided mediastinal contour suggestive of a mediastinal mass (*). The lungs were unremarkable.
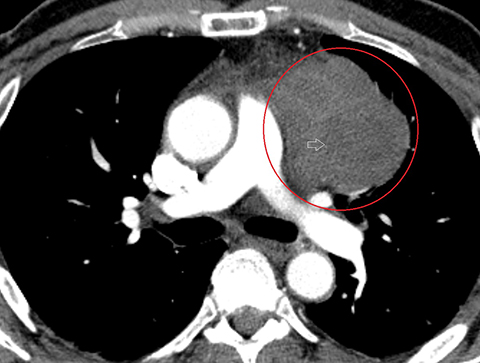
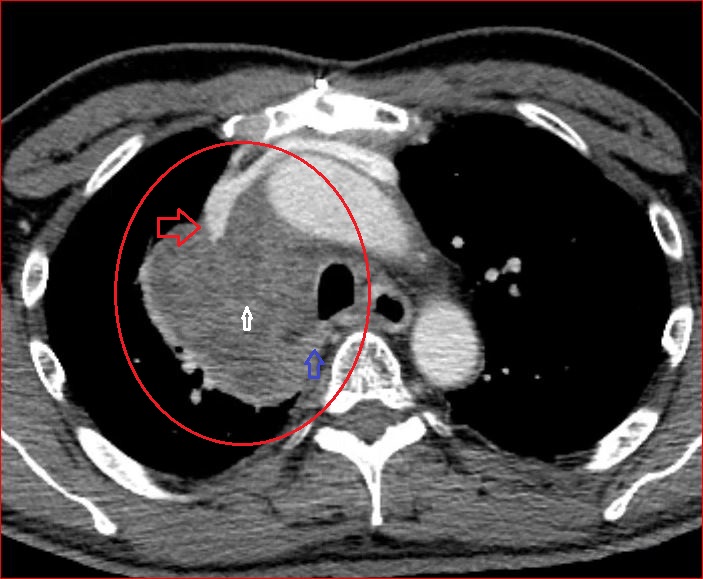
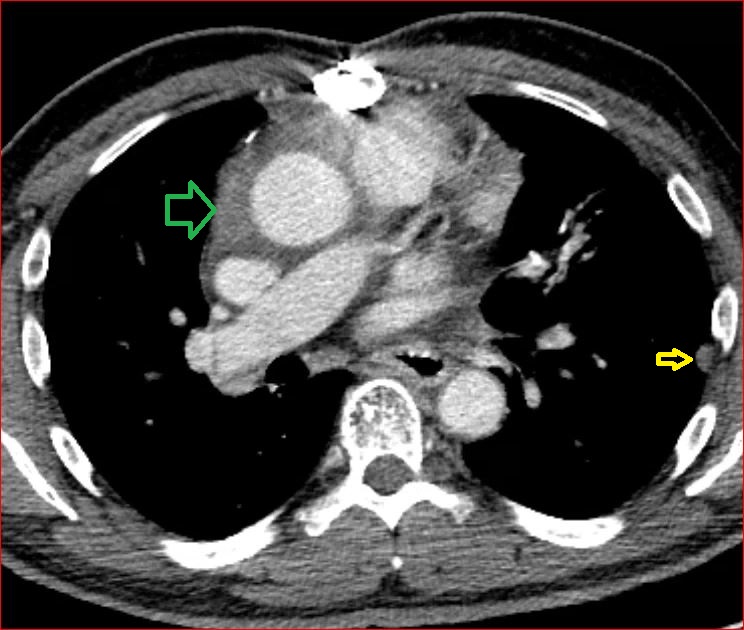
A CTA of the chest (Figure 2) demonstrated a large anterior mediastinal mass (O) with internal heterogeneity suggestive of necrosis (white arrow). The left anterior descending artery was seen to traverse the mass without resultant arterial narrowing (yellow arrow). The remainder of the examination was unremarkable.
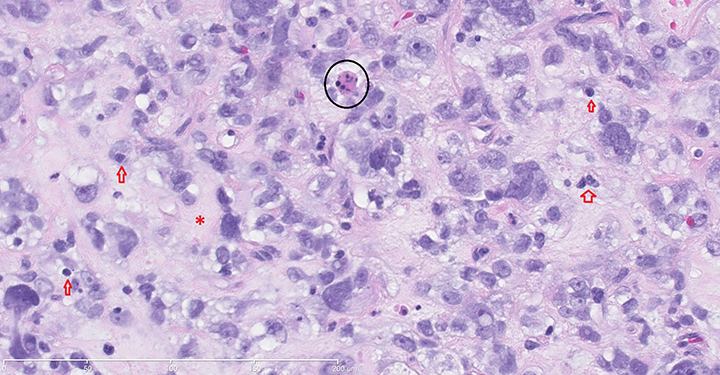
Pathologic samples (Figure 3) showed polygonal pleomorphic poorly differentiated myxoid cytoplasm with apoptosis and fibrous bands interwoven between tumor cells with extensive hyalinization positive for keratin, vimentin and p63.
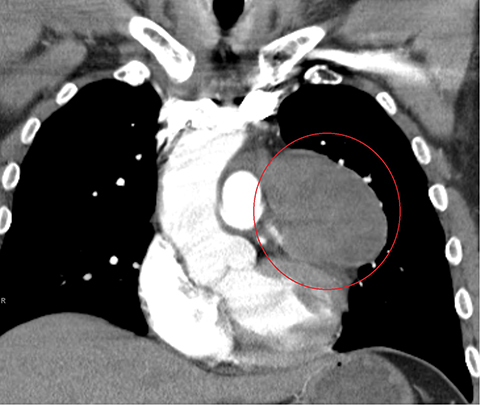
The patient underwent resection and radiation therapy and follow-up CT imaging three months later was negative for recurrence. Follow-up imaging 9 months postresection was obtained. Follow-up posteroanterior chest radiograph showed an abnormal right-
sided mediastinal contour suggestive of a mediastinal mass (*, Figure 4).
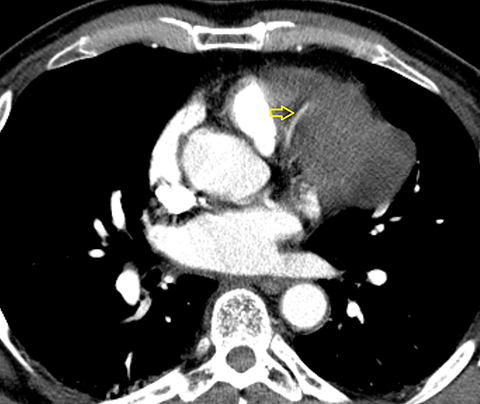
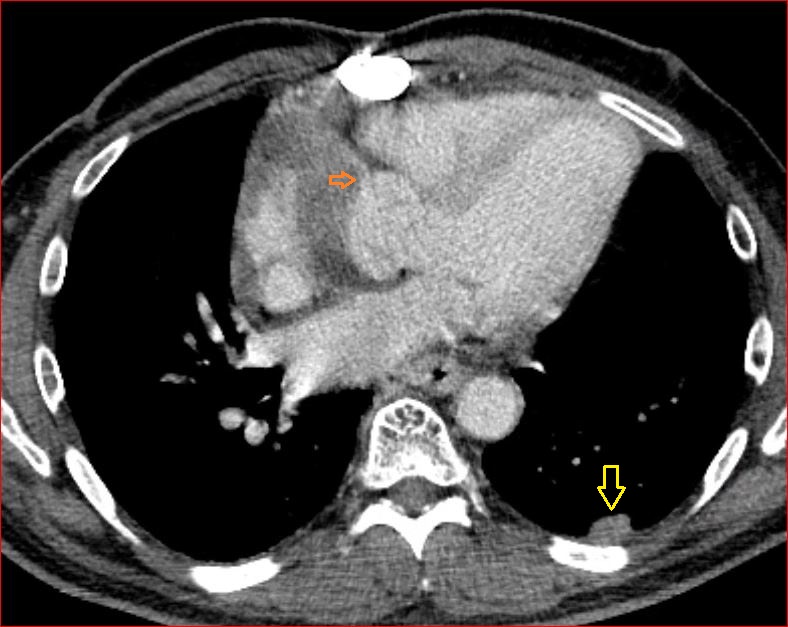
A CTA of the chest (Figure 5 demonstrated a large mediastinal mass (O) with extension into the posterior (blue arrow) mediastinal compartment, and internal heterogeneity suggestive of necrosis (white arrow). The mass partially encased the superior vena cava (red arrow) and extended into the epicardial fat (green arrow) with a large component involving the right atrioventricular groove surrounding the right coronary artery (orange arrow). Pleural nodules on the left, which was the side of the initial tumor, were also present (yellow arrows).
DIAGNOSIS
Thymic carcinoma
Differential Diagnosis: Thymic germ cell tumor, lymphoma, invasive thymoma, benign mature teratoma
DISCUSSION
The overall incidence of thymomas is approximately 0.15 cases per 100,000, with thymic carcinomas accounting for only 0.06 percent of these neoplasms; making this an extremely rare entity.1 Literature is very limited owing to the paucity of reported cases. The neoplasm shows a preponderance in the 40through 60-year-old population with a male to female ratio of 1.9:1.2
Most patients with this tumor are generally asymptomatic. The most common presenting symptom is chest pain. Other signs including cough, dyspnea, and shoulder pain have been reported.2 Symptomatology is generally secondary to compression or invasion of contiguous mediastinal structures as well as distant intrathoracic spread, which are distinguishing characteristics of this entity helping differentiate it from other masses.4
Abnormal laboratory values can include leukocytosis and anemia secondary to tumor production of granulocyte colony-stimulating factor (G-CSF) and interleukin-6, respectively.3,4
On CT, thymic carcinomas are generally poorly marginated and invariably located within the anterior mediastinal region. Internal areas of heterogeneity may represent necrosis, hemorrhage, calcifications and sometimes even cyst formation.5 Benign mature teratomas in this region can also demonstrate internal calcifications; however, these are usually well defined rounded masses which may sometimes have a multicystic appearance with internal fat.5 Internal foci of fat excludes thymic carcinoma from the differential and is diagnostic of a thymolipoma or teratoma.6
Pleural implants otherwise known as “drop metastases” can be seen in association with thymic carcinomas. Diaphragmatic elevation secondary to phrenic nerve palsy has also been reported.7Unfortunately, these findings can also be seen with thymomas, making histologic analysis pivotal to diagnosis.
Pathologic staining for TTF-1, Keratin, p63, CD70, CD5, and CD1a helps distinguish thymic carcinoma from other thymic and pulmonary neoplasms.8
The invasive nature of this mass makes it unresectable in most cases; however, surgical resection is still attempted when deemed feasible. Multimodality treatment with radiation and chemotherapy is generally used due to the aggressive nature of this mass.9 Five-year survival ranges from 30-50 percent.9 Due to a very high relapse rate, lifelong surveillance for recurrence is recommended.9
CONCLUSION
Thymic carcinomas are extremely rare tumors seldom seen in clinical practice. They are relentlessly progressive and invasive and thus recognition of the imaging and pathologic characteristics of these tumors is critical for diagnosis. Their highly aggressive nature and tendency for distant intrathoracic spread helps distinguish this entity from other masses. Given the limited number of cases in the literature, optimal management has yet to be determined. Multifaceted treatment with surgery, chemotherapy and radiation is the treatment of choice given the aggressiveness of this disease.
REFERENCES
- Engels EA, Pfeiffer RM. Malignant thymoma in the United States: Demographic patterns in incidence and associations with subsequent malignancies. Int J Cancer. 2003;105(4):54651.
- Maghbool, Maryam, et al. Primary adenocarcinoma of the thymus: an immunohistochemical and molecular study with review of the literature. BMC Clinical Pathology. 2013;13(1):17.
- Tada Y, et al. A case of thymic carcinoma producing granulocyte colony-stimulating factor. Nihon Kokyuki Gakkai Zasshi. 1998;36(12):1043-1047.
- Suzuki, et al. Interleukin-6producing undifferentiated thymic carcinoma with neuroendocrine features. Lung Cancer. 2009;63(3):425-429.
- Bogot NR, Quint LE. Imaging of thymic disorders. Cancer Imaging. 2005;5(1):139-149.
- Juanpere S, Canete N, Ortuno P, et al A diagnostic approach to the mediastinal masses. Insights Into Imaging. 2013;4(1):29-52.
- Jung KJ, Lee KS, Han J, et al. Malignant thymic epithelial tumors: CT-pathologic correlation. AJR. 2001;176:433-439.
- Pomplun S, Wotherspoon AC, Shah G, et al. Immunohistochemical markers in the differentiation of thymic and pulmonary neoplasms. Histopathology. 2002;40:152-158.
- Eng TY, Fuller CD, Jagirdar J, et al. Thymic carcinoma: state of the art review. Int J Radiat Oncol Biol Phys. 2004;59(3):654-664.
Citation
C S, J R, T F, J F. Thymic carcinoma. Appl Radiol. 2016;(2):38-40.
February 3, 2016