Osteoid Osteoma Treatment: Image-guided Resection Vs Image-guided Ablation
By Wirth A, Towbin RB, Schaefer CM, Towbin AJ

Case Summary
A child who plays catcher in baseball had a 2-month history of left hip pain, more severe at night, that improved with ibuprofen. Physical examination demonstrated decreased left hip range of motion and guarding when rotating the hip.
Imaging Features
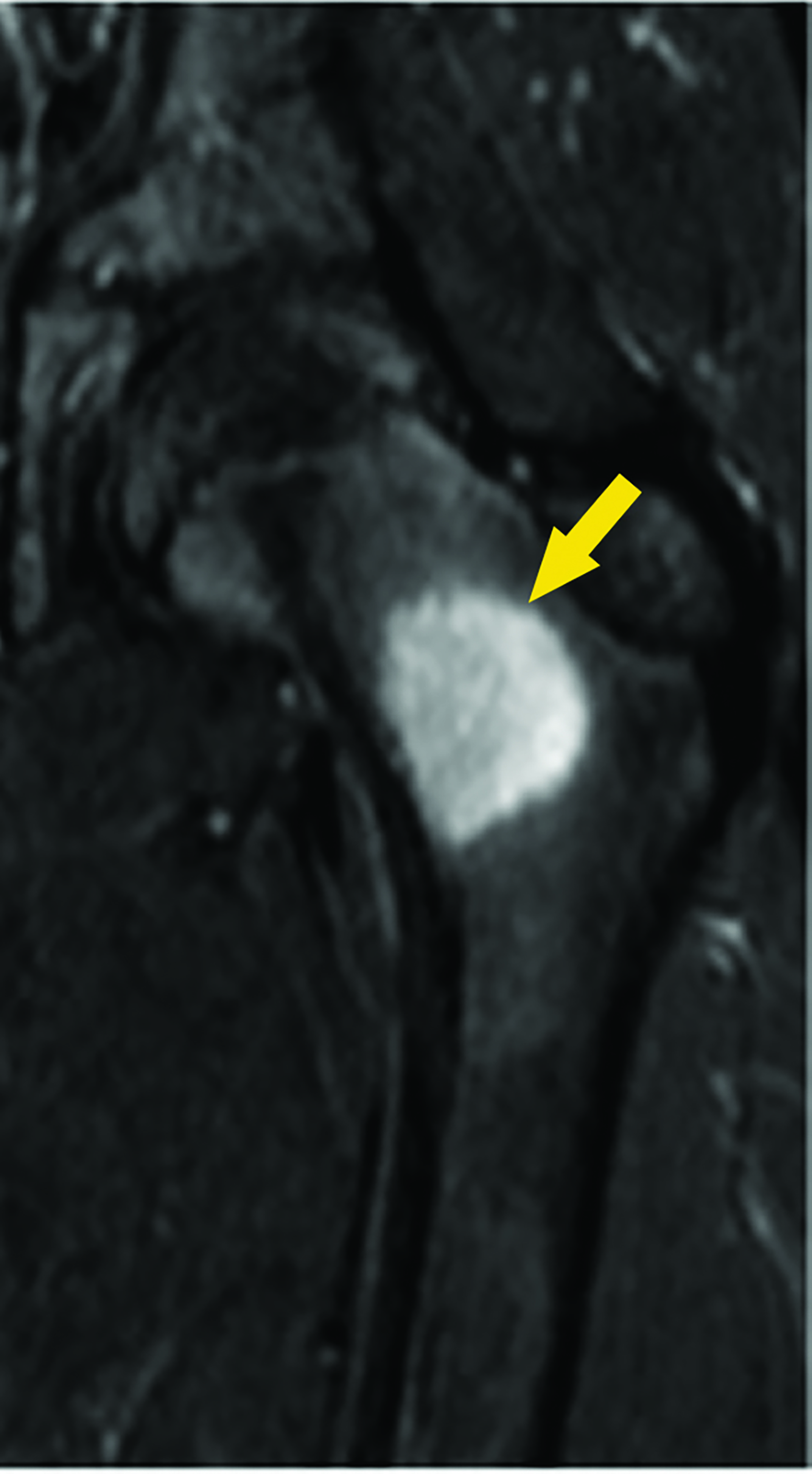
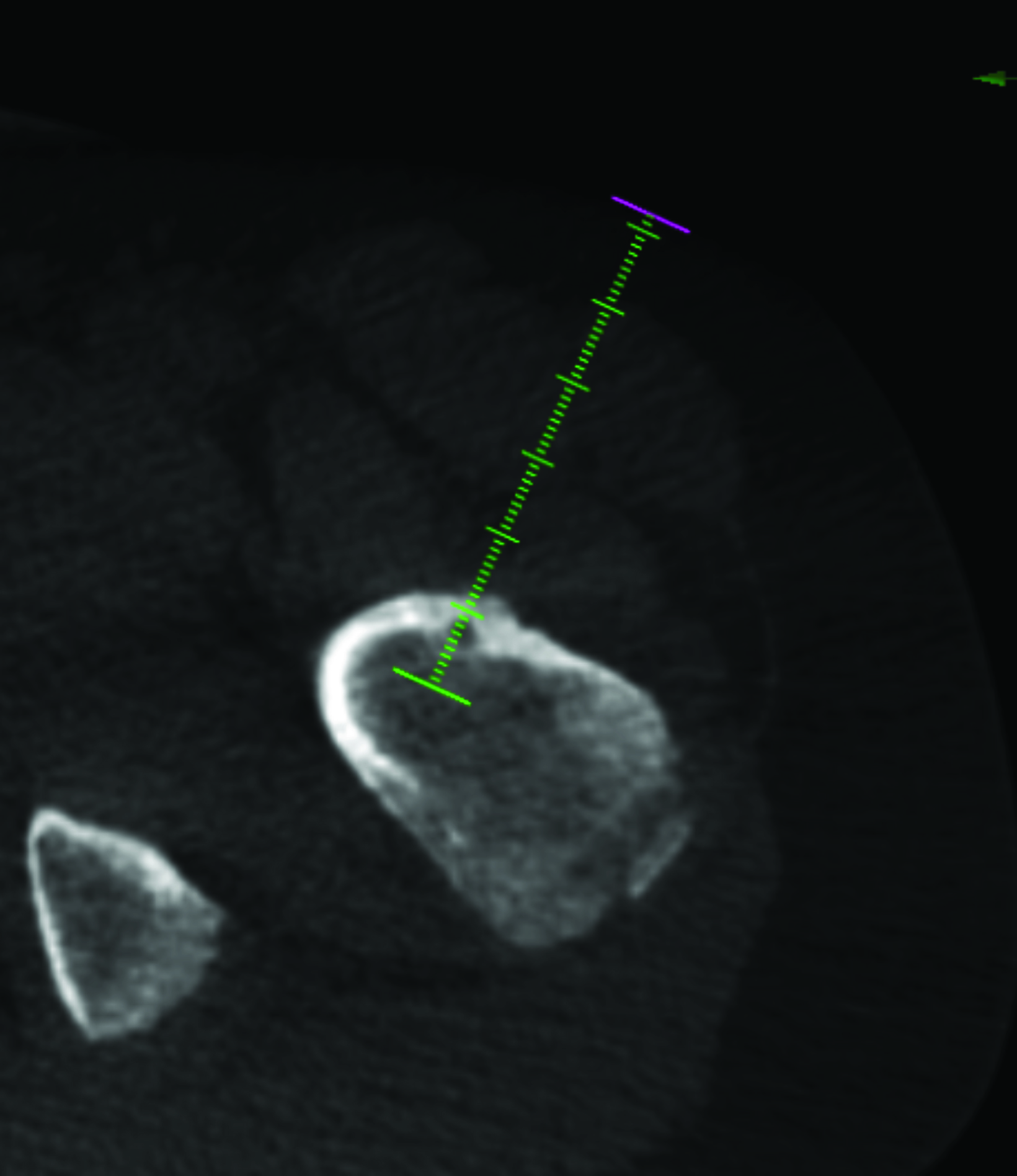
Magnetic resonance imaging (MRI) showed a focal area of marrow edema (Figure 1) seen along the medial central aspect of the junction of the left femoral neck and proximal shaft. Anteriorly a small focal nidus involved the cortex. Sagittal STIR images showed mild edema overlying the osseous, cortically-based nidus with mild local periosteal reaction. All findings were consistent with a diagnosis of osteoid osteoma (OO).
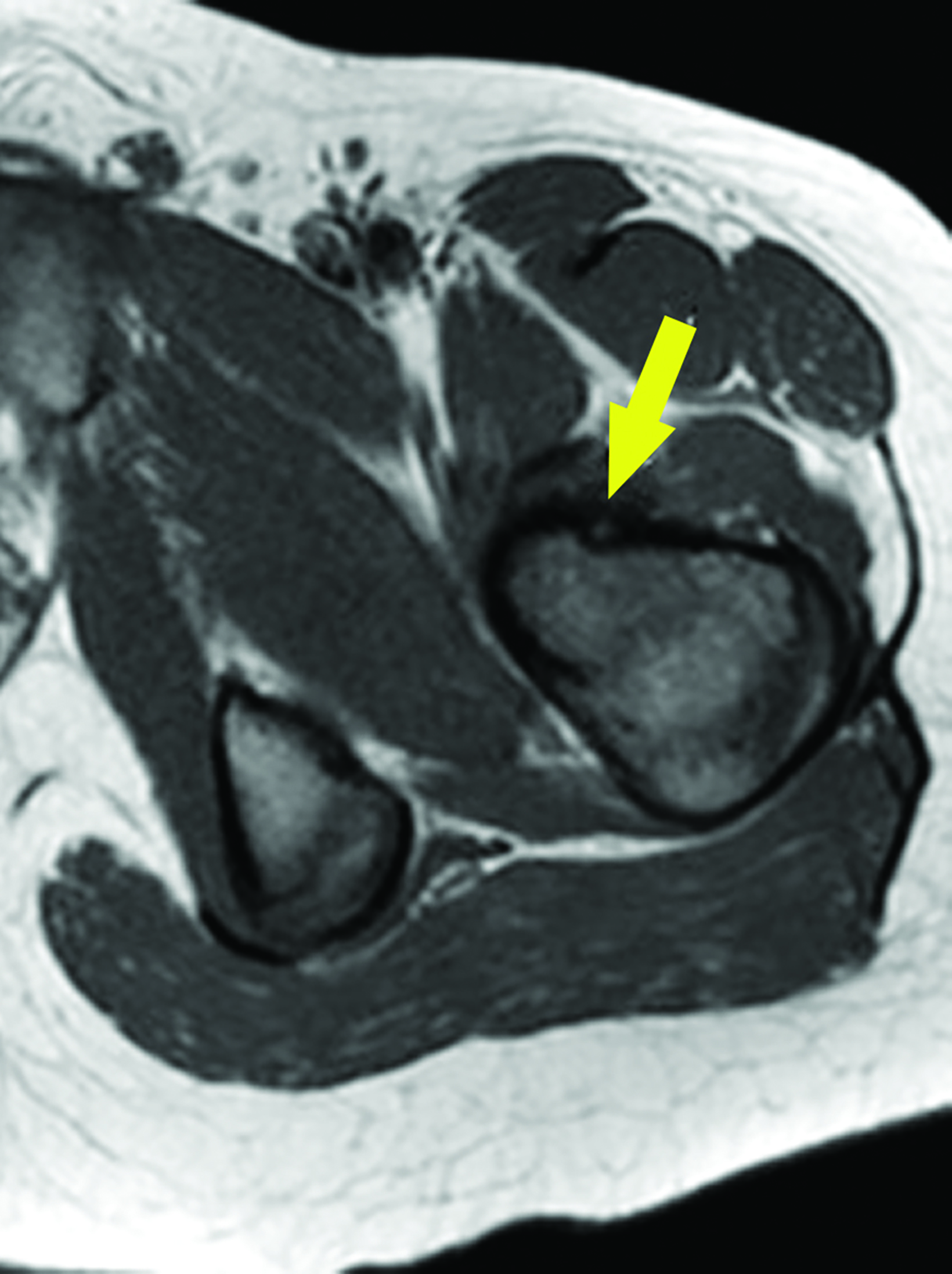
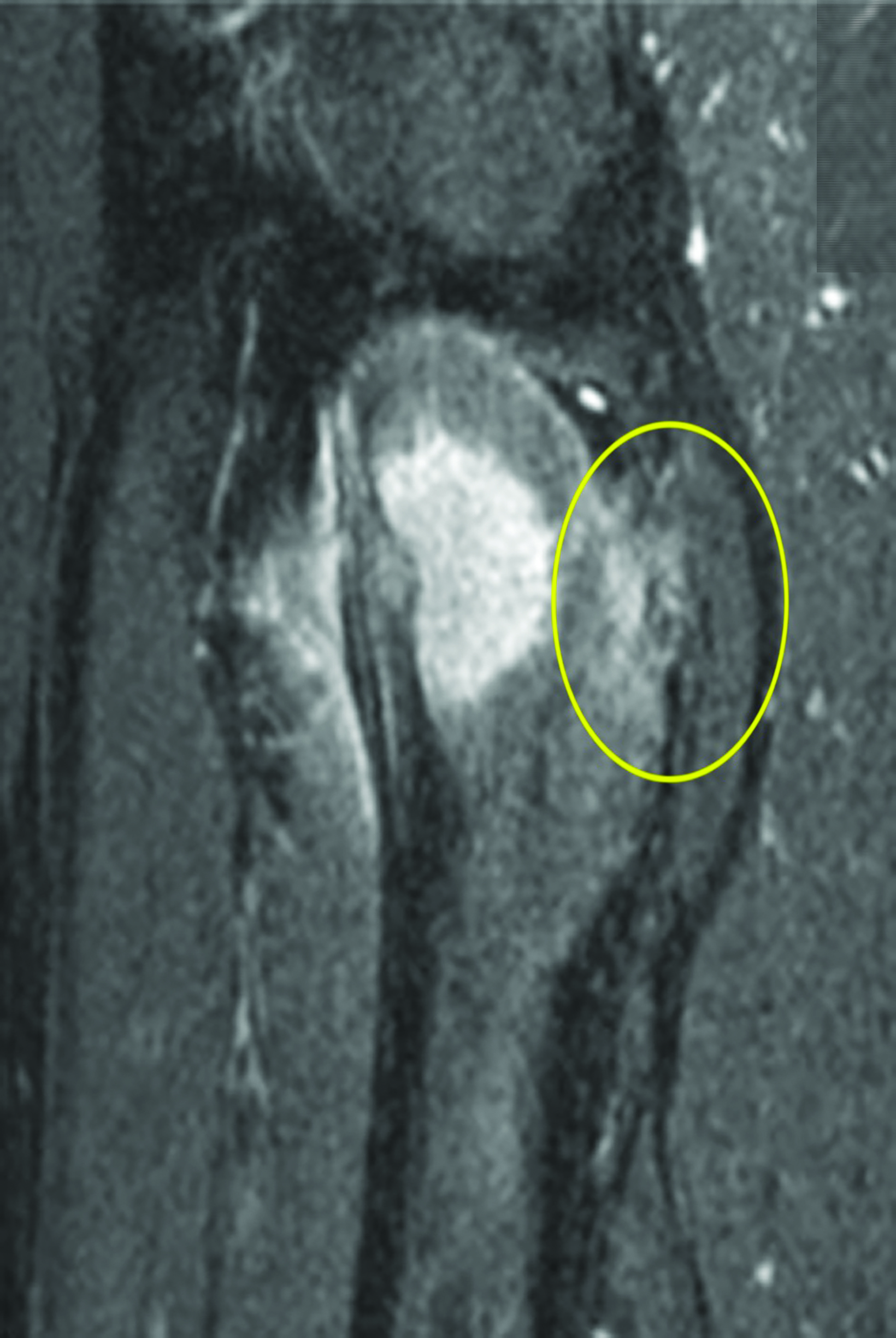
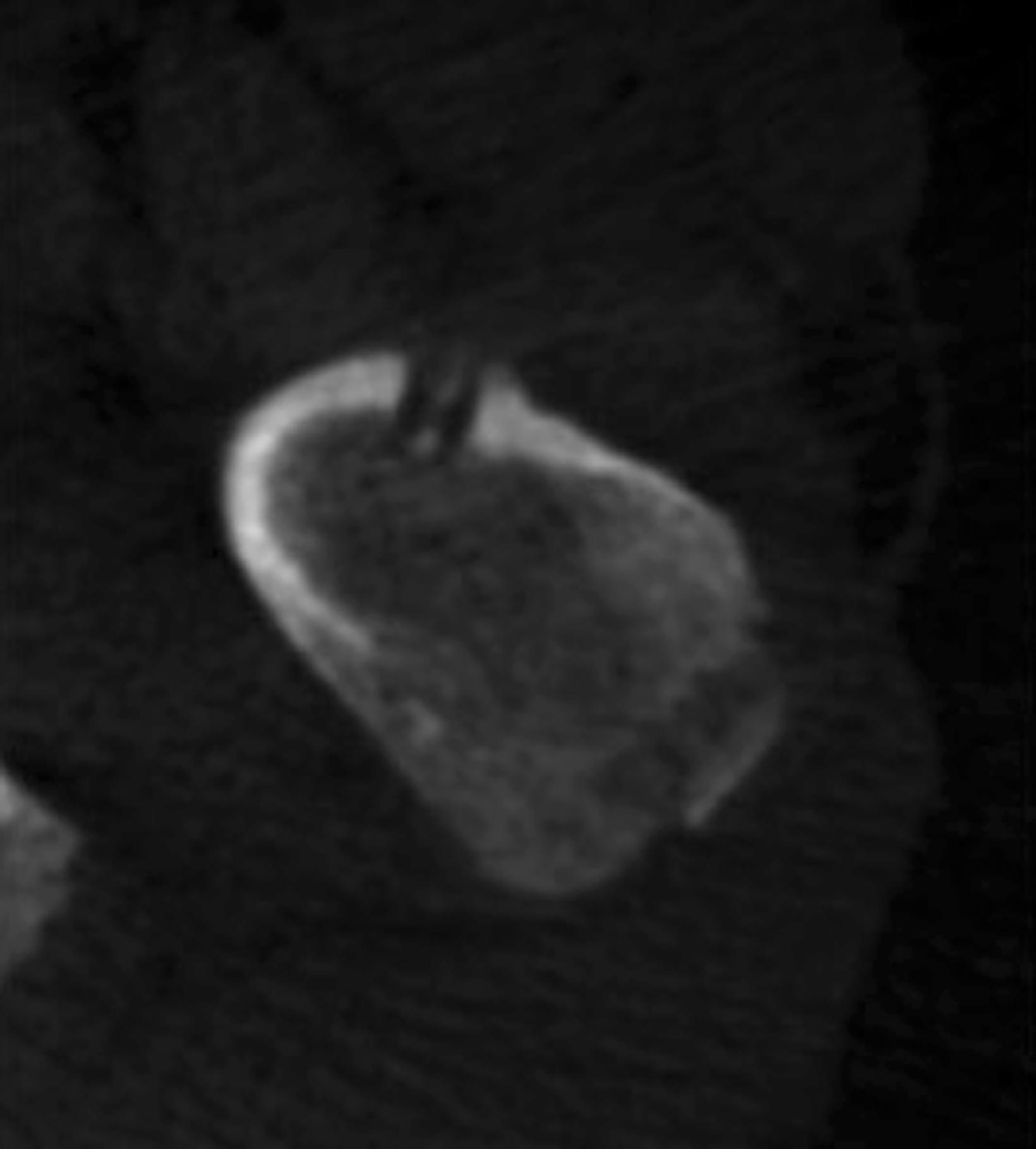
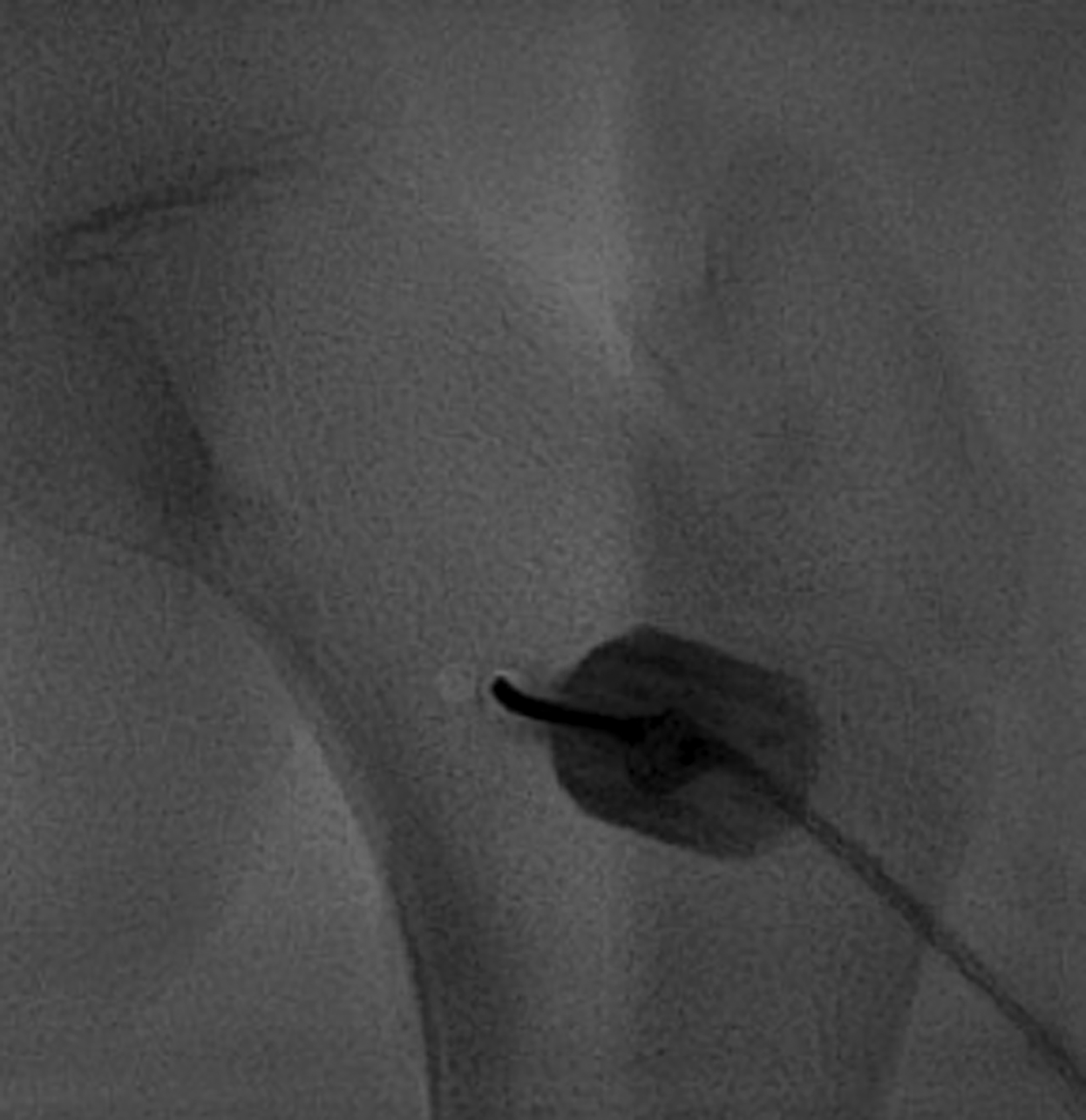
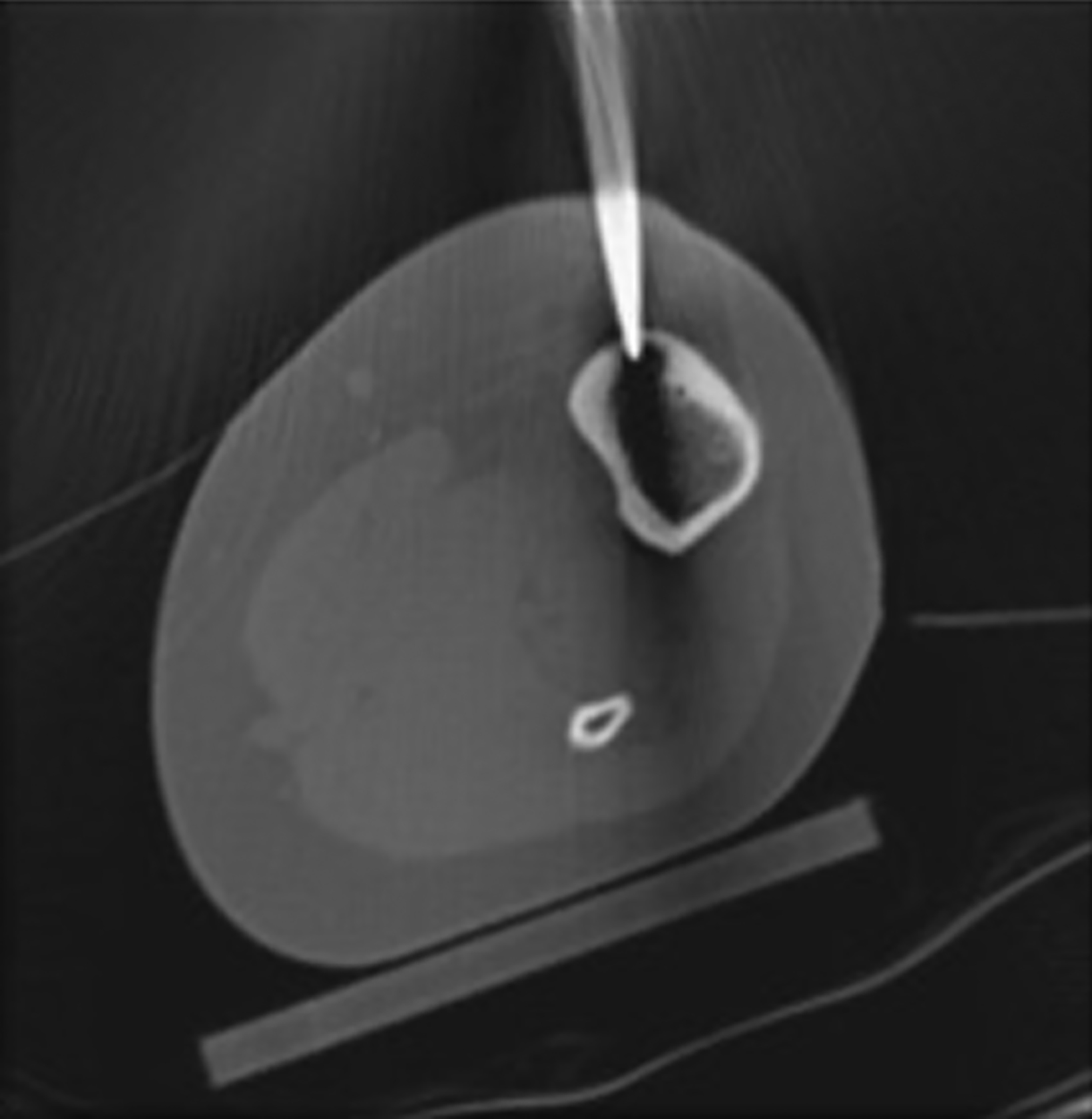
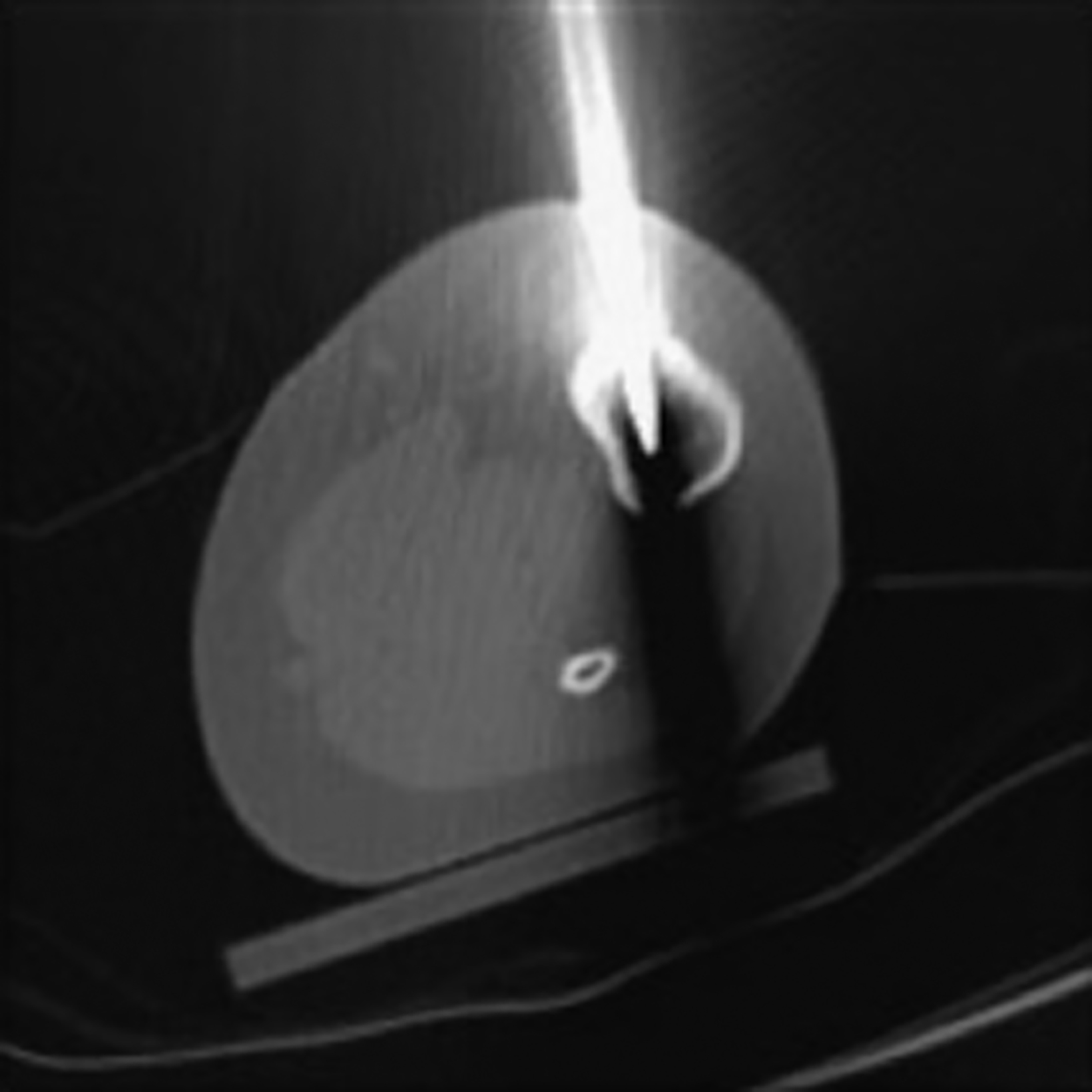
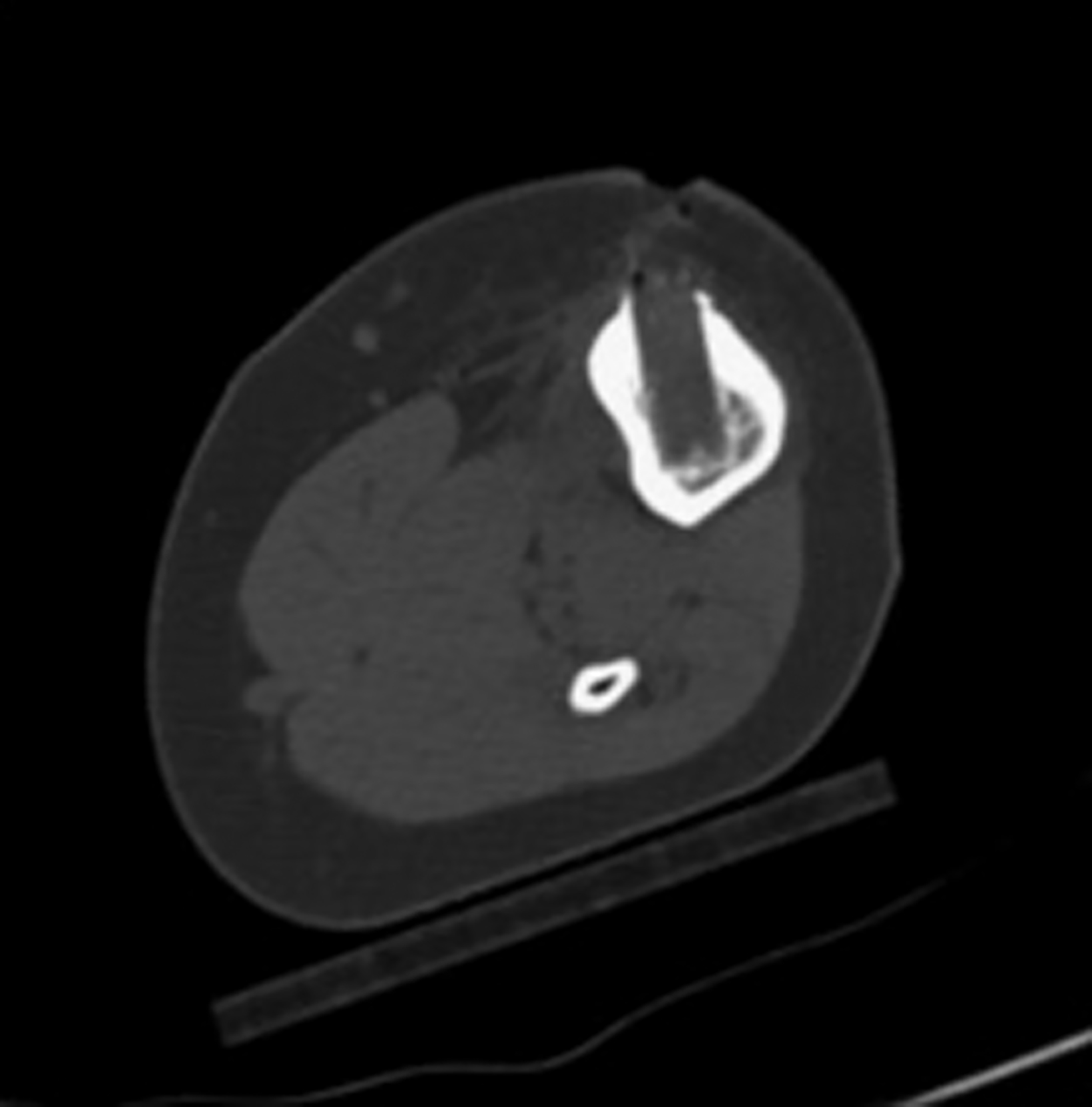
After treatment planning (Figure 2) the osteoid osteoma was treated percutaneously using a combination of percutaneous resection and radiofrequency ablation (Figure 3).
Discussion
Osteoid osteoma is a benign, bone-forming neoplasm that accounts for 12% of all skeletal neoplasms .1 Osteoid osteomas are highly vascularized tumors consisting of osteoid and woven bone that typically do not exceed 1.5 cm in diameter.1,2 While OOs can occur anywhere in the skeleton, they are most commonly found in the cortex of long bones, with a higher predominance in the lower extremities.1, 2, 3 These lesions are commonly seen in patients between 10 and 30 years old; they occur more often in males at about a 2:1 ratio.4 Patients with OOs present with pain, classically occurring at night and relieved with nonsteroidal anti-inflammatory drugs (NSAIDs).5 Their nonspecific symptoms and poorly localized pain may delay diagnosis and treatment.
Osteoid osteomas appear as round to oval, radiolucent lesions with a central nidus surrounded by a well-circumscribed sclerotic rim on radiographs. Intramedullary and intra-articular lesions usually lack the sclerotic margin as well as periosteal reaction.4 If radiographs are inconclusive, computed tomography (CT) is valuable for diagnostic confirmation and treatment planning. 1,3,5
The use of MRI to diagnose OOs is controversial, as the nidus cannot be clearly detected in up to 35% of cases.2 On MRI the nidus can best be visualized on T1 contrast-enhanced images as low-to-intermediate signal intensity that enhances with gadolinium-based contrast.2 Additionally, T2 fat-suppressed images may be used to identify the nidus.
Dynamic contrast-enhanced MRI (DCE-MRI) is more sensitive than traditional MRI in detecting OO. In DCE-MRI, OOs are visualized as a peak in signal enhancement during the arterial phase.2 This technique is utilized to distinguish OO from Brodie abscess, where Brodie abscess has a central, nonenhanced area and OO has diffuse enhancement.2
Treatment Approaches
Observation with pain control may be considered in patients with OOs, as they may heal without intervention.6 Unfortunately, symptom resolution may take 2-3 years or longer, making this a rarely chosen and impractical approach.6 Treatment should be considered in patients who do not respond to NSAIDs, who are at risk for renal and gastrointestinal complications of long-term NSAID use, and in skeletally immature patients due to risk of growth disturbance.6,7 The goal of intervention is excising or destroying the entire nidus to cure the lesion and provide symptomatic relief.
Historically, open en bloc excision with cortical shaving and curettage of the nidus cavity was standard of treatment.6,7 However, this approach comes with challenges in localizing the tumor, which may lead to incomplete removal and an increased risk of recurrence.6,7 Additionally, resecting weight-bearing bone often necessitates instrumentation with fixation devices, casting, and longer periods of restriction on weight bearing and return to activities. 6,7
Despite intraoperative CT guidance and/or nuclear imaging with radiotracers to guide open en bloc excision, minimally invasive procedures have become the mainstay of treatment. The image-guided approaches are more precise and have lower complication rates and recovery times. They are also less costly and usually can be performed on an outpatient basis.
Currently, the preferred minimally invasive approaches to treatment consist of CT-guided percutaneous excision, CT-guided radiofrequency ablation, and CT-guided cryoablation.
CT-guided Percutaneous Excision
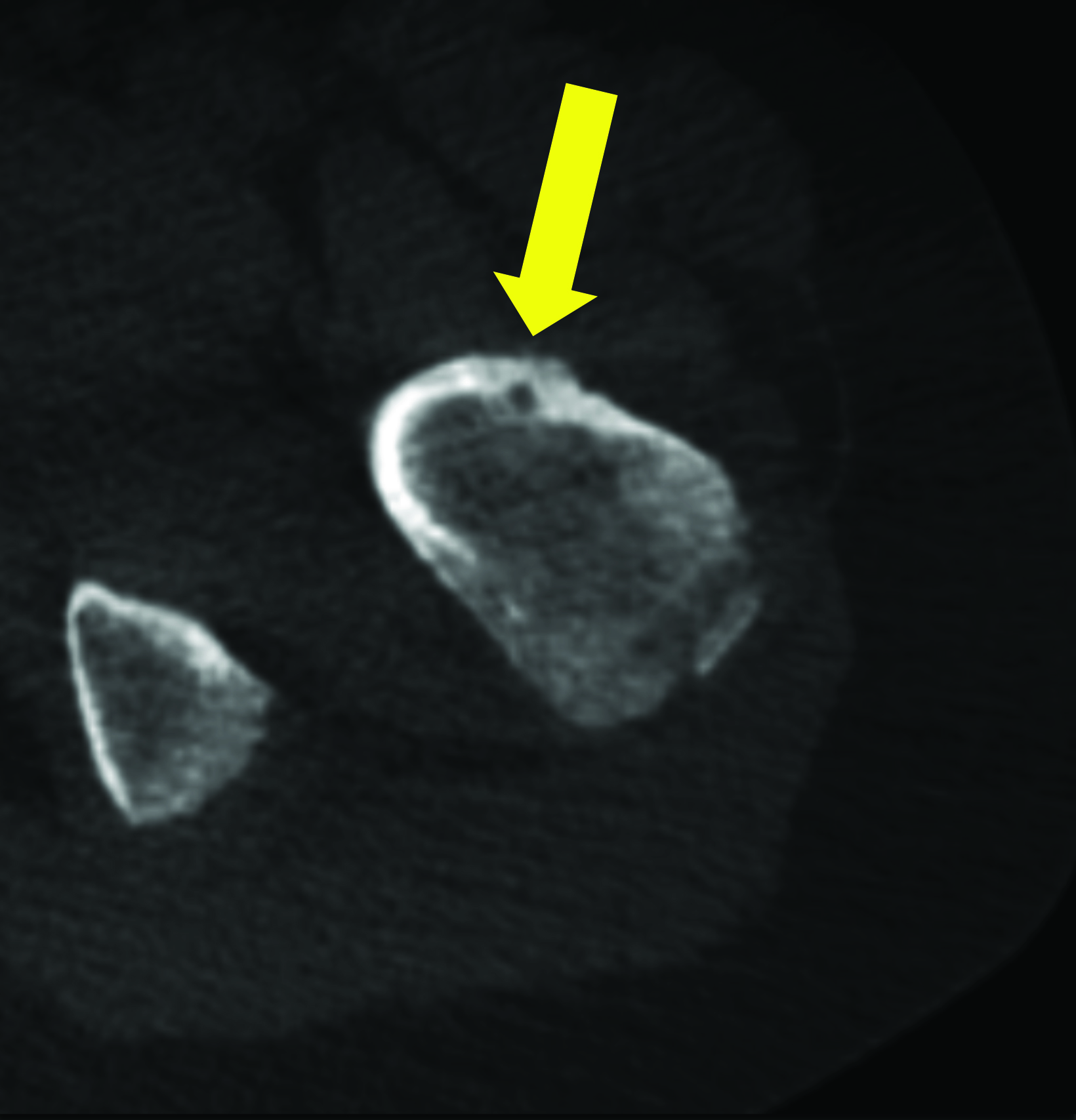
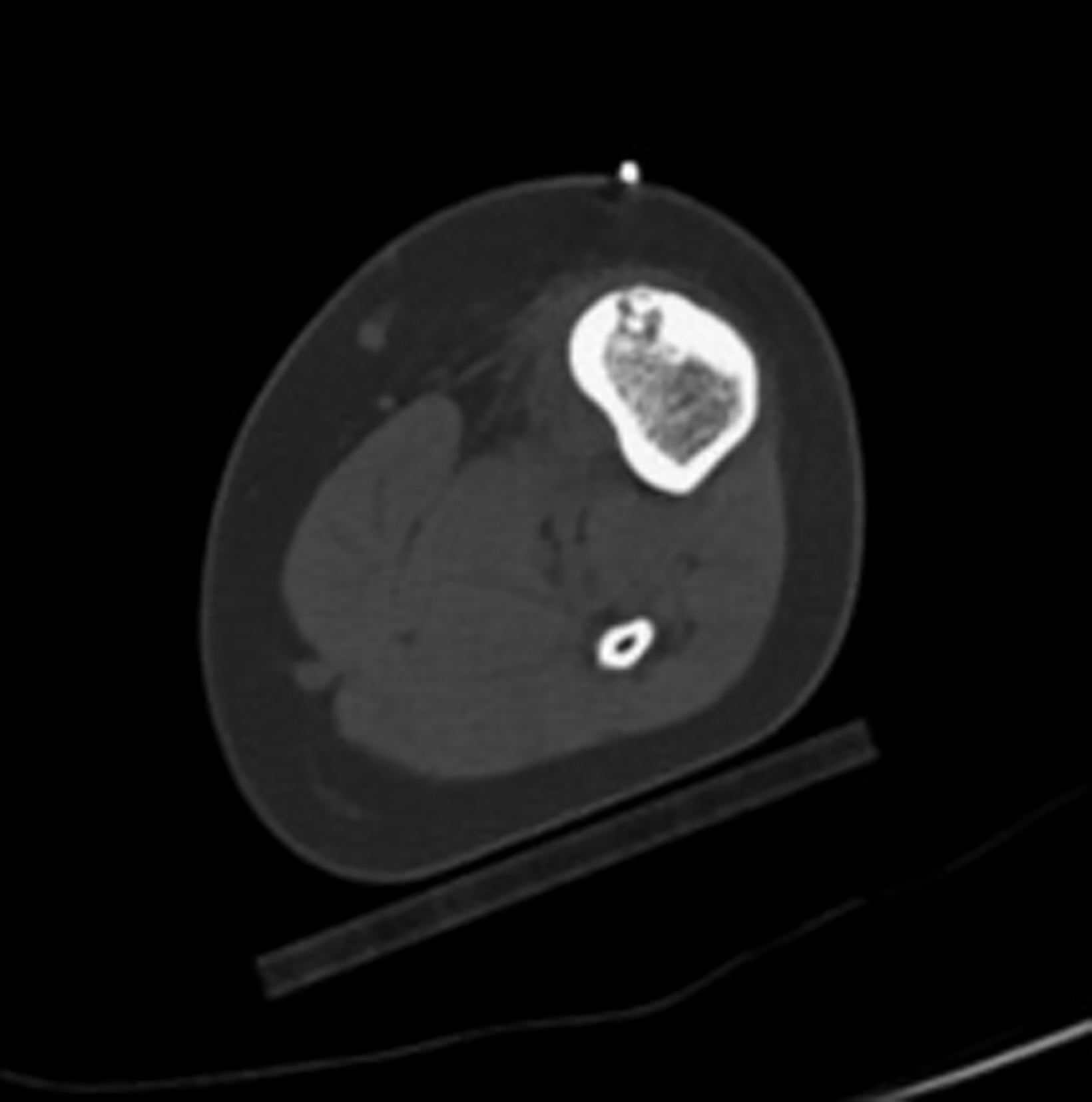
In this method, a trephine or cannulated drill and a wire are used to excise the lesion under CT guidance.7-9 This approach is highly precise and is correlated with a high clinical success rate and minimal complications (Figure 4).7-9 This approach offers the benefit of a surgical specimen to confirm complete lesion removal. On rare occasions, a self-limiting nerve injury or infection may be associated with this approach. Fenichel, et al,9 noted mild femoral neuropraxia in one patient after removal of a lesion from the acetabular roof. Towbin, et al,8 noted sensory loss over the proximal thigh after removal of a lesion of the femoral neck.
In both instances, symptoms were self-limiting and resolved in less than 2 months.8,9 This complication was also noted with CT-guided radiofrequency ablation.10 Another consideration for this approach is the concern that excision of the nidus and surrounding cortex may predispose the patient to a pathological fracture. Fortunately, this occurs in less than 1% of cases.
CT-guided Radiofrequency Ablation
Computed tomography-guided radiofrequency ablation is the most commonly used technique to treat OO. In this procedure, a bone-cutting needle creates a defect in the adjacent cortex. A radiofrequency probe is then introduced into the lesion through a guide needle. The tip of the probe is heated to 70-90°C for 5-6 minutes.7 This technique is associated with excellent pain relief, reduced operative morbidity, and low recurrence rates.4 While tissue sampling can be performed with this technique, histologic diagnosis is less successful, occurring in less than 50% of cases, when compared to CT percutaneous excision, which removes the entire nidus.7
CT-guided Cryoablation
Computed tomography-guided cryoablation utilizes multiple freeze and thaw cycles at -40°C to induce tissue necrosis.10 Lindquester, et al,10 found the technique to have a success rate statistically similar to CT-guided radiofrequency ablation, with the added benefit of significantly less pain, a decreased need for post-operative analgesia, and faster patient discharge. As a newer technique, CT-guided cryoablation has a smaller body of evidence than CT-guided radiofrequency ablation.
Conclusion
Osteoid osteoma is a benign tumor that most commonly presents in the long bones of adolescent males. Diagnosis is based on clinical symptoms of night pain relieved by NSAIDs and the presence of round or oval radiolucent nidus on skeletal radiographs and/or CT. Treatment includes NSAIDs and removal of the lesion.
CT-guided ablation and CT-guided percutaneous excision have replaced open en block excision as treatments of choice owing to their superior precision, reduced complication rates, and shorter recovery time. Emerging research into CT-guided cryoablation shows the procedure has similar success rates as CT-guided radiofrequency ablation with decreased pain and faster recovery, factors that in time may lead it to be preferred over radiofrequency ablation.
References
- Hakim DN, Pelly T, Kulendran M, Caris JA. Benign tumours of the bone: A review. J Bone Oncol. 2015;4(2):37-41. Published 2015 Mar 2. doi:10.1016/j. jbo.2015.02.001
- Malghem J, Lecouvet F, Kirchgesner T, Acid S, Vande Berg B. Osteoid osteoma of the hip: imaging features. Skeletal Radiol. 2020;49(11):1709-1718.
- Healey JH, Ghelman B. Osteoid osteoma and osteoblastoma. Current concepts and recent advances. Clin Orthop Relat Res. 1986 Mar;(204):76-85. PMID: 3956019.
- Goldblum JR, Lamps LW, McKenney JK, Myers JL. Rosai and Ackerman’s Surgical Pathology E-Book. 11th ed. Elsevier; 2017.
- May CJ, Bixby SD, Anderson ME, et al. Osteoid osteoma about the hip in children and adolescents. J Bone Joint Surg Am. 2019;101(6):486-493. doi:10.2106/JBJS.18.00888
- Noordin S, Allana S, Hilal K, et al. Osteoid osteoma: contemporary management. Orthop Rev (Pavia). 2018;10(3):7496. Published 2018 Sep 25. doi:10.4081/or.2018.7496
- Atesok KI, Alman BA, Schemitsch EH, Peyser A, Mankin H. Osteoid osteoma and osteoblastoma. J Am Acad Orthop Surg. 2011;19(11):678-689. doi:10.5435/001 24635-201111000-00004
- Towbin R, Kaye R, Meza MP, Pollock AN, Yaw K, Moreland M. Osteoid osteoma: percutaneous excision using a CT-guided coaxial technique. AJR Am J Roentgenol. 1995;164(4):945-949. doi:10.2214/ajr.164.4.7726054
- Fenichel I, Garniack A, Morag B, Palti R, Salai M. Percutaneous CT-guided curettage of osteoid osteoma with histological confirmation: a retrospective study and review of the literature. Int Orthop. 2006;30(2):139-142. doi:10.1007/s00264-005-0051-1
- Lindquester WS, Crowley J, Hawkins CM. Percutaneous thermal ablation for treatment of osteoid osteoma: a systematic review and analysis. Skeletal Radiol. 2020;49(9):1403-1411. doi:10.1007/s00256-020-03435-7
University of Arizona College of Medicine-Phoenix (Ms Wirth), Phoenix Children's Hospital, Department of Radiology (Drs Schaefer, R Towbin), Children’s Hospital Medical Center, and University of Cincinnati, Cincinnati, Ohio (Dr Alexander Towbin).
