Image quality and radiation dose at coronary CT angiography
Images




Dr. Collins is currently a third-year Radiology Resident at the University of California-San Francisco, San Francisco, CA. He received his medical degree from The Northwestern University Feinberg School of Medicine in Chicago, IL, and completed his Internship at The Queen's Medical Center, Honolulu, HI. He plans to pursue a Cardiovascular Imaging Fellowship followed by an Interventional Radiology Fellowship.
Noninvasive, accurate morphologic imaging of the coronary arteries is the Holy Grail of emergency medicine, cardiology, and radiology. The coronary arteries are small vessels requiring submillimeter isotropic resolution to resolve distal branches. High temporal resolution is a priori to freeze cardiac motion. Dose-reducing algorithms and low-dose protocols have reduced the coronary CT angiography (CTA) dose to one that is comparable to that of catheter angiography. This article will discuss coronary CTA technique, components of image quality, radiation dosimetry, and exposure.
Morphologic imaging of the coronary arteries was first performed on electron-beam computed tomography (EBCT) scanners in 1984. 1,2 Advantages included a temporal resolution of 50 to 100 msec. 3 Poor resolution in the Z-axis, limited volume coverage, and low power limited its use for coronary CT angiography (CTA). 4
The introduction of helical multidetector CT (MDCT) in the 1990s revolutionized imaging of the coronary arteries. Improvements in volume coverage were achieved through simultaneous acquisition of 4 slices per gantry rotation, reducing acquisition time to 40 seconds. An effective temporal resolution of 250 msec limited image quality. Multisegment reconstruction algorithms were developed to overcome this limitation. 5 Despite improvements in axial collimation to 1 mm, partial volume artifacts remained problematic. 6,7
Sixteen-slice MDCT scanners with simultaneous acquisition of 12 to 16 slices further reduced acquisition time to 15 to 20 seconds. Gantry rotation increased to 375 msec. Spatial resolution improved with axial collimation to 0.75 mm. Investigators reported fewer nondiagnostic segments with improvement in accuracy compared with 4-slice scanners. 8,9 Poor temporal resolution necessitated pharmacologic heart rate reduction and the use of multisegment reconstruction algorithms. Partial volume artifacts remained challenging in patients with stents and high Agatson scores. 10-
Sixty-four slice MDCT scanners were introduced in 2004. The latest-generation 64-slice scanner (SOMATOM Sensation 64, Siemens Medical Solutions, Forchheim, Germany) enables simultaneous acquisition of 64 overlapping slices through advanced z-sampling techniques, improving temporal resolution to isotropic voxels of 0.4 mm. 13 Greater volume coverage per gantry rotation with an acquisition time of 5 to 14 seconds is within a comfortable breath-hold for all but the most dyspneic patients. An effective temporal resolution of 165 msec is achieved with partial scan algorithms. Many investigators recommend routine pharmacologic heart-rate- reducing medications. 14,15
The recent introduction of a dual-source MDCT scanner (SOMATOM Definition, Siemens Medical Solutions, Forchheim, Germany) presented a solution to the limited temporal resolution achievable with single-source systems. Combining multiple X-ray sources to improve temporal resolution at CT was initially described in 1979 by Robb and Ritman. 16,17 The incorporation of a second X-ray source and detector array into the gantry offset 90˚ enables the acquisition of half-scan data in a quarter gantry rotation. An effective temporal resolution of 83 msec is achieved without employing multisegment reconstruction algorithms. Improved temporal resolution enables heart-rate-independent imaging and clear visualization of intra-cardiac structures. 4,18-20
A 256-slice scanner has been introduced by Toshiba Medical Systems (Ohtawara, Japan). This scanner employs a cone-beam design with a Z-axis coverage of 12.8 cm per rotation, enabling whole-heart coverage without table movement. 21-23 Stationary volume acquisition eliminates interpolation artifacts. Spatial resolution is improved with 0.5-mm detector elements. Gantry rotation speeds on the alpha prototype are as fast as 350 msec. 24 The relatively high radiation dose associated with the cone-beam X-ray source is a limitation of this scanner design. 22 Investigators have proposed prospectively gated acquisitions and improved detector design as solutions to this problem. 22,25
CTA technique
Several aspects of coronary CTA technique are pertinent to this discussion of image quality and radiation dose.
Prospective triggering
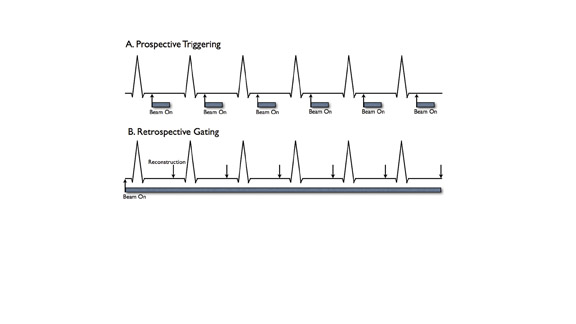
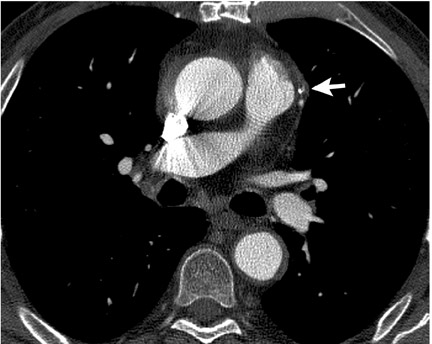
Prospective triggering initiates CT scanning at a specified point in the cardiac cycle (Figure 1A). This is based on the length of a recent RR interval on the electrocardiogram (ECG) (ie, 60% of the R-R interval or 500 msec after the R wave). Data acquisition is continued for a chosen length of time (ie, time corresponding to 20% of the R-R interval). Lower radiation dose is an advantage of prospective triggering. Disadvantages include susceptibility to heart rate variability, inability to edit data to compensate for arrhythmias, and limited cardiac phases for data reconstruction. Functional parameters can be determined by including end systole and end diastole in the triggered acquisitions. 25
Retrospective gating
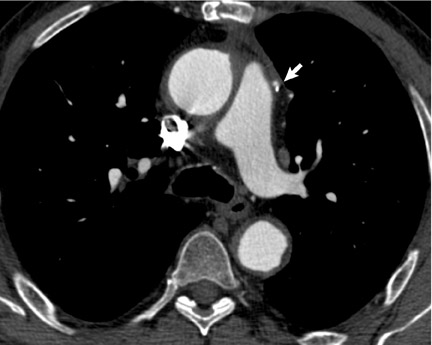
Retrospectively gated acquisitions record the ECG simultaneously with CT scanning. Imaging data are acquired throughout the entire cardiac cycle (Figure 1B). Volume data can be reconstructed at any point in systole or diastole. An advantage of this technique is the ability to edit the ECG tracing to exclude data acquired following ectopic beats. Cardiac functional parameters such as ejection fraction, wall motion, and myocardial thickening can also be assessed.
Multisegmentation
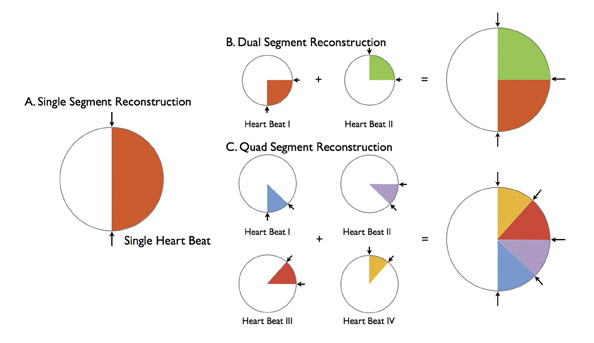
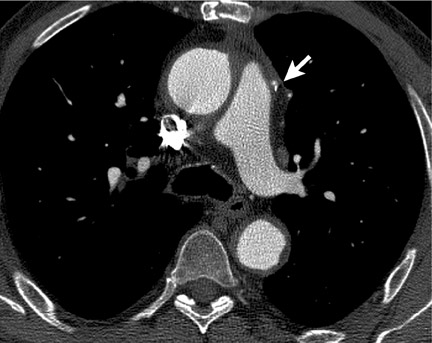
Multisegmentation refers to acquiring cardiac cycle data at a point along the Z-axis from ≥2 adjacent heartbeats (Figure 2). Although an effective solution to limited temporal resolution, this technique is particularly susceptible to beat-to-beat variability and ectopic beats. Pitch is necessarily reduced, increasing scan time and radiation dose.
Convolution kernel
CT kernels are filters designed to enhance particular spatial frequencies. Sharp filters are applied when high-density objects such as stents, bones, and dense vascular calcifications are imaged. Sharp filters enhance edge definition at high-contrast interfaces at the expense of increased noise and reduced signal-to-noise ratio (SNR). Coronary CTA is usually performed with a medium-sharp filter, which balances edge definition with acceptable noise (Figure 3). In patients with high Agatson scores and stents, sharp filters result in subjective improvements in image quality. 26
Image quality
Spatial resolution
Spatial resolution is the most fundamental aspect of image quality and is described in voxel dimensions. CT data are reconstructed on a 512 × 512 pixel matrix. Spatial resolution is dependent on the field of view, slice collimation, and detector element size. Utilizing clinical protocols and a sharp convolution kernel, a maximum in-plane resolution of 0.5 mm × 0.5 mm and through-plane resolution of 0.4 mm can be achieved. 4
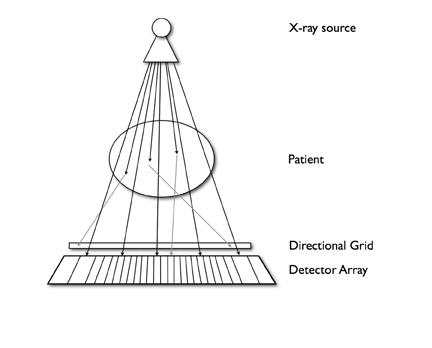
The SNR is a practical measure of image quality associated with spatial resolution. Reducing the reconstructed field of view or utilizing thinner collimation while keeping kVp and mAs constant results in fewer X-rays traversing each voxel relative to a constant noise background. Consequently, SNR decreases, and the image appears grainy. The tube current must be increased in order to maintain an acceptable SNR. Similarly, tube current must be in-creased to maintain an adequate SNR with smaller detector elements. Gains in SNR are not directly proportional to tube current, however. A portion of X-rays scattered within the patient are deflected out of a straight path and are absorbed by peripheral detector elements, thus increasing noise (Figure 4). Doubling the tube current results in an increase in SNR of 1.41.
These considerations have practical implications regarding imaging the coronary arteries. Most investigators administer vasodilator medications to increase vessel size. 27 Current scanner spatial resolution is adequate for the evaluation of the epicardial coronary arteries and major branches. High spatial resolution reduces beam-hardening artifacts from stents and calcified vessels and improves accurate assessment of stenoses (Figure 5). 28 Techniques to reduce beam-hardening artifacts are currently under investigation (Sven Prevrhal, University of California, San Francisco, CA, personal communication). Further improvement in spatial resolution to 0.2 to 0.25 mm is necessary for accurate imaging of in-stent restenosis and plaque characterization.
Future gains in spatial resolution can be achieved with smaller detector elements. Flat-panel detectors have a high spatial resolution of up to 0.1 mm. A prototype CT scanner utilizing flat-panel detectors has been developed. 29 This scanner is capable of a spatial resolution of 0.25 mm. 3 Although this is an exciting development, to achieve acceptable radiation doses and temporal resolution, further improvements in flat-panel detector efficiency and flat-panel data read-out are necessary.
Temporal resolution
Slice acquisition time must be less than an isovolumetric portion of the cardiac cycle to effectively freeze cardiac motion. Using partial scan algorithms, nominal slice acquisition time can be reduced to 165 msec in scanners with 330-msec gantry rotation time. At a heart rate of 72 beats per minute (bpm), this nominal acquisition time corresponds to approximately 20% of the R-R interval.
Pharmacologic heart rate control is routine at single X-ray source coronary CTA. Target heart rates of <70 and <65 bpm are reported in the literature. 12-15 Reduction of temporal resolution to <100 msec is necessary for heart-rate-independent imaging across most resting heart rates. 4 Early studies with dual-source technology demonstrated excellent image quality across a wide range of heart rates, obviating the need for pharmacologic heart rate reduction. 4,28
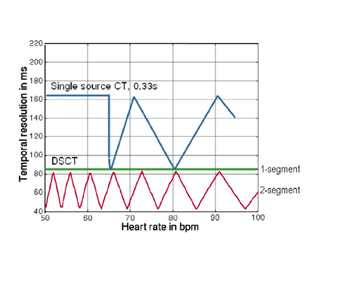
Multisegmentation reduces the effective temporal resolution by acquiring slice-specific, isophasic data from 2 to 4 consecutive heart beats. Effective temporal resolution can be reduced to 43 msec. 30,31 Improvements in temporal resolution with this technique are related to heart rate; 83 msec temporal resolution is achievable only at heart rates of 68 and 82 bpm 32 (Figure 6). These "sweet spots" occur when there is optimal desynchronization between the gantry rotation and heart rate. Effective temporal resolution with dual-segment reconstruction at other heart rates averages 124 msec. A recent study investigated the accuracy of single- versus dual-segment reconstruction at 64slice coronary CTA. 32 Although dual-segment reconstruction resulted in improved image quality, there was no difference in diagnostic accuracy.
Increases in gantry rotation speeds have occurred with each generation of MDCT scanners, improving temporal resolution. Higher gantry rotation rates would generate significant centrifugal forces and necessitate increases in processing power to accommodate the high rate of data transmission. 4 The dual-source scanner design provides an efficient solution to improve temporal resolution. A temporal resolution of 83 msec is achieved with single-segment reconstruction; an average temporal resolution of 60 msec is achieved with dual-segment reconstruction (Figure 6). Several authors reported motion-free coronary CTA without pharmacologic agents using single-segment reconstruction. 4,19,20 Flohr and colleagues 4 discourage the routine use of dual-segment algorithms, citing radiation considerations.
Retrospectively gated acquisitions enable the user to specify the cardiac phase for reconstruction. Although a single-phasic reconstruction may not be motion-free, an analysis of multiple reconstructions may yield a complete evaluation of the coronary tree. Reconstructions at end diastole and end systole are preferable for low and high heart rates, respectively. 4
Contrast resolution
Contrast resolution refers to the ability of an imaging system to resolve 2 objects of similar size but different attenuation. This is simply demonstrated by concentric solid squares. As the shading pattern of the center square closely approximates that of the outer square, they blend together and cannot be distinguished, although slight differences in shading density do exist (Figure 7). Contrast resolution is important in differentiating fatty, fibrofatty, and calcific plaque as well as calcifications from luminal contrast. An important limitation of coronary CTA contrast resolution is the small size of luminal plaque components. Small structures require greater differences in attenuation for differentiation at coronary CT. Ap-propriate selection of window and level settings is necessary to observe the minimum contrast during image review.
Dual-source CT scanners may be able to utilize differential energy spectra subtraction to improve contrast resolution. Applications at coronary CTA are the focus of ongoing investigations.
Patient factors
Patient-specific factors impact image quality. Image noise is directly related to the thickness of tissue that the X-ray photon traverses. In patients with high body mass indices, increasing the tube current and slice collimation is necessary to maintain SNR. Similarly, in women with large breasts, positioning the breasts out of the field of the X-ray beam reduces beam attenuation and maintains SNR. Scan times with state-of-the-art 64-slice scanners average 5 to 9 seconds for coronary CTA; whole thoracic gated acquisitions can take 20 seconds. 33 Patient preparation for the length of the breath-hold as well as the sensation of warmth from intravenous (IV) contrast injection improves cooperation. Respiratory motion causes interpolation and misregistration artifacts, which can mimic stenoses. 34 If patients cannot complete the breath-hold, they should be instructed to slowly exhale. Shortening the scan time by increasing the pitch or reducing the scan volume reduces respiratory motion artifacts. Finally, a regular heart rhythm is necessary. The ECG tracing can be edited in retrospectively gated acquisitions to reduce artifacts from ectopic beats. Frequent ectopy or irregular atrial fibrillation rhythms may result in nondiagnostic studies (Figure 8). Tachycardic patients who are not candidates for pharmacologic heart rate reduction can be imaged with dual-source scanners. 4,32
Other technical factors
Optimal opacification of the coronary arteries is critical to CTA. A large-bore antecubital IV line is the preferred access site. Review of prior imaging is prudent to exclude central venous stenosis or occlusion. A test bolus is preferred by most authors to determine transit time to the aortic root. 35 Contrast media with an iodine concentration >350 mg/mL is preferred. The volume of contrast is equal to the scan time multiplied by the injection rate. Contrast is injected, followed by a saline flush at 5 to 6 mL/sec. This dual-injection technique clears contrast from the central veins and right heart, preventing beam-hardening artifacts from obscuring the mid portion of the right coronary artery. 36
Radiation dose considerations
All imaging studies utilizing ionizing radiation should adhere to the "as low as reasonably achievable" (ALARA) principle. Reported doses at coronary CTA vary significantly because of inconsistent application of dose-reducing methodology and varied coronary CTA protocols (Table 1). Multiple measures of radiation dose have been reported in the literature, adding to the confusion (Table 2).
Radiation dose at coronary CTA is dependent on multiple technical factors. Dose is directly proportional to the square of the kVp and varies linearly with mAs. 43 All cardiac CTA protocols are acquired helically, where pitch setting determines the degree of overscanning. Pitch is defined by the length of table translation per gantry rotation divided by the total detector width. Pitch should be maximized to reduce the radiation dose. 4 The scanning volume should be confined to the cardiac anatomy with the minimal necessary z-overscanning for slice interpolation at the edges of the volume. Exposure time should be minimized for all patients.
Reducing dose at coronary CTA
It is estimated that 20% of the radiation dose is used to reconstruct a single phase. The remainder of the dose enables functional evaluation, unless additional phasic reconstructions are required for analysis. 44 Multiple strategies have been developed to reduce radiation dose. As discussed under spatial resolution above, with smaller detector elements, increased tube current is necessary to maintain SNR. Without the advent of dose-saving strategies, a significant increase in exposure would have been realized with higher slice MDCT. 39 However, a steady decline in radiation exposure has occurred from 4-slice to 64-slice MDCT because of the implementation of dose reduction strategies (Table 1).
ECG pulsing
Electrocardiographic tube current modulation was initially described in 1999. 45,46 This strategy is used with retrospectively gated acquisitions, where an 80% to 90% reduction in the nominal tube current is applied during systole and early diastole (Figure 9). The authors recommend a 400-msec interval of nominal tube current applied during diastole or during late systole and diastole. Phases reconstructed within the pulsing interval have low signal-to-noise ratio, which is adequate for measurements of functional parameters but may be insufficient for the evaluation of coronary stenoses (Figure 10). Hence, appropriate selection of the pulsing interval is mandatory. Dose savings from 37% to 56% are reported with this technique. 46-49
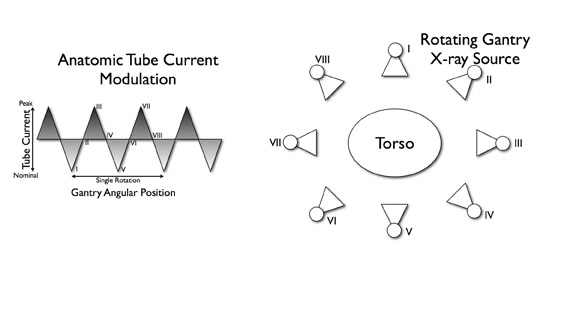
Anatomic tube current modulation
Anatomic tube current modulation is a recently developed dose-saving technique. Several approaches to estimate or measure patient X-ray attenuation have been described in the literature. Studies that compared angular tube current modulation based on anthropomorphic phantoms with modulation from angular projection-specific attenuation reported greater dose savings with in-line modulation (Figure 11). This results in an average dose savings of 44% in children and 53% in adults. 50 The addition of automated exposure control to 16-slice coronary CTA performed with ECG modulation resulted in a further 43% reduction in radiation dose. 40
Prospective triggering
Prospectively triggered acquisitions impart less radiation dose than retrospective acquisitions (see Figure 1). Radiation in a prospectively triggered acquisition occurs only during a specified portion of the cardiac cycle. This reduces exposure time and the radiation dose compared with retrospectively gated acquisitions. Dose reductions of 70% to 80% have been reported with this technique. 51 The use of a prospectively triggered acquisition has been proposed as a means of reducing dose at 256-slice MDCT, reducing the dose from 14.2 to 2.4 mSv. 25
Low-dose protocols
Low kVp and mAs protocols have been promulgated in smaller patients and children for coronary CTA and extracoronary CT. 52 An approach pioneered by Paul and Abada 44 measures image noise on a low-dose CT scan with 120 kVp and 20 mAs at the inferior margin of the heart. Scan parameters are subsequently adjusted based on control tables. A recent study by Hausleiter and colleagues 39 compared doses at 16- and 64-slice coronary CTA using 100 kVp and 120 kVp protocols with ECG modulation. They reported 53% and 64% dose reduction at 16- and 64-slice coronary MDCT, respectively, without a difference in evaluable coronary segments. A recent study by Delhaye et al 41 evaluated the feasibility of coronary segment analysis on 64-slice MDCT gated thoracic imaging with a low-dose protocol (120 kVp and 200 mAs). Although the study was not tailored for coronary imaging, 75% of coronary segments were evaluable. The average dose for low-dose gated thoracic evaluation was 5 mSv. The accuracy of low-dose protocols is the subject of ongoing research.
Radiation risks
X-rays are classified as carcinogens by the National Institute of Environmental Health Sciences, the World Health Organization, and the Centers for Disease Control. 53-55 The effects of ionizing radiation are classified as deterministic or stochastic (Table 3). Patient doses at coronary CTA are well below the thresholds for deterministic effects. Stochastic effects are theoretically possible with the dose from a single coronary CTA study. Limited data is available to determine the risk of carcinogenesis from effective doses <50 mSv. Above this level, population data from the Hiroshima nuclear fallout found a statistically significant increased cancer incidence. 57,58 Wide variation in individual radiation exposure in this population limits extrapolation to patients exposed to doses common in diagnostic imaging. 59 Additional studied populations include
U.S. X-ray technologists, Nordic airline pilots, populations living in high background exposure regions of China, nuclear radiation workers, patients exposed to Thorotrast contrast, radium dial painters, and postpartum mastitis and ankylosing spondylitis radiotherapy patients. 59-70 Radiation exposure in these cohorts is similar to doses at coronary CTA. Study results are inconsistent, although a statistically significant incidence or positive trend for thyroid cancer, leukemia, melanoma, and nonmelanoma skin cancer, and colorectal cancer was noted in multiple studies. Data on breast carcinoma induction is limited, as available study cohorts were predominantly male. Based on available data, the lifetime risk of fatal cancer is estimated at 5% to 7.9% per sievert of exposure. 59,71 This corresponds 1/1300 to 1/2000 risk of fatal cancer following an exposure of 10 mSv. 58,72
The lifetime attributable risk (LAR) of cancer incidence from coronary CTA was recently analyzed by Einstein and colleagues. 73 Monte Carlo simulations and data from the National Academies' Biological Effects of Ionizing Radiation 7th report formed the basis for their work. The authors highlighted stochastic risk associated with coronary CTA in younger patients, particularly women. The LAR for a 20-year-old woman undergoing 35% ECG modulated coronary CTA was estimated at 0.46% or 1 in 219. This compared to an LAR of 0.1% or 1 in 1000 in a 20-year-old man. The LAR declined progressively with age. A 20-year-old man had 5 times the relative risk (RR) of an 80-year-old man. However, a 20-year-old woman had 23 times the RR of an 80-year-old man. Gender-specific differences in LAR were attributed to increased lung parenchyma and breast doses in women.
Ionizing radiation is a relatively weak carcinogen. Differentiating ionizing-radiation-induced cancers from sporadic and other carcinogen-induced cancers is challenging, as approximately 40% of the U.S. population will develop cancer. 74 Investigators have turned to population-based data to determine the scope of medical imaging exposure. In 1980, the collective dose from medical imaging was 0.55 mSv per person. This quantity has increased approximately 6-fold to 3.3 mSv per person in 2006. 58 The U.S. population dose from medical imaging in 2006 totaled 450,000 person-Sv. This compares with 600,000 person-Sv total exposure from the Chernobyl nuclear accident. The total annual U.S. population exposure is comparable to that in high background radiation regions of China. 70 Gonzalez and colleagues 75 estimate a 0.6% to 1.8% lifetime cancer risk from radiation exposure at medical imaging in developed countries.
The European Commission has developed guidelines for exposure limits for CT. 76 Currently, no federal guidelines exist for CT dosimetry. The American College of Radiology (ACR) recently published a white paper on radiation dose, 77 which makes recommendations on imaging utilization but does not specify CT dosimetry guidelines.
Relative effective dose at coronary CTA
Effective doses at coronary CTA, positron emission tomography, diagnostic angiography, and nuclear imaging are listed in Table 4. Effective doses at coronary CTA vary significantly based on imaging protocol and patient factors, but range from 4.95 to 40 mSv. Using dose reduction techniques, radiation doses at coronary CTA are similar to diagnostic catheter angiography and technetium-99m nuclear perfusion studies.
Radiation considerations in special populations
When imaging children, women, and patients with low body mass indices, special care should be taken to implement dose-reducing measures at coronary CTA. Pediatric patients are more sensitive to radiation than older patients. 72,86,89 Manufacturers have developed pediatric protocols which utilize low-dose algorithms. Investigators have validated low-dose protocols in whole thoracic and focused coronary CT imag- 39,41,44,90 Paul and Abada 44 noted excellent image quality at coronary CTA utilizing 80 kVp protocols as long as mAs was adapted to patient weight. In women undergoing coronary CTA, the breasts receive the highest effective dose. When possible, breasts should be positioned out of the beam. An alternative approach is the use of bismuth breast shields. 44
Conclusion
Innovations in scanner design have yielded consistent improvements in image quality since coronary CTA was first described with EBCT. Novel dose-reduction strategies realized reduced effective doses with later-generation CT scanners. Rapid, accurate, noninvasive coronary imaging is now possible at CTA with radiation doses comparable to a diagnostic catheter angiogram.
Related Articles
Citation
Image quality and radiation dose at coronary CT angiography. Appl Radiol.
December 17, 2007