Right-to-left shunt associated with microscopic pulmonary arterial venous malformations (PAVMs) secondary to hepatopulmonary syn
Images
Prepared by Bao To, MD , Department of Radiology, University of California San Francisco; and Marcia McCowin, MD and Stephen K. Gerard, MD, PhD , San Francisco VA Medical Center and Department of Radiology, University of California San Francisco, San Francisco, CA.
Case summary
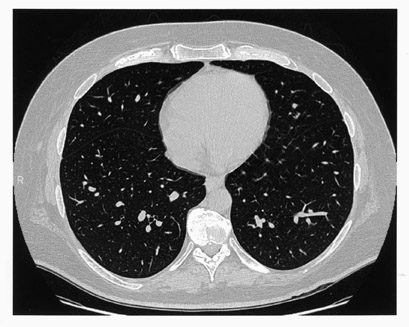
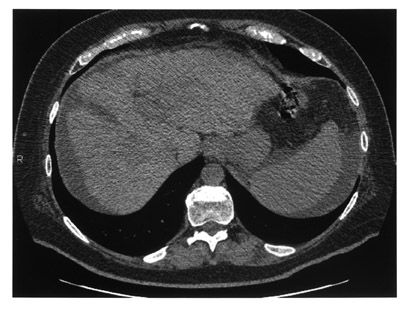
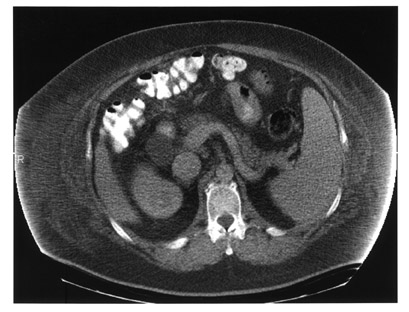
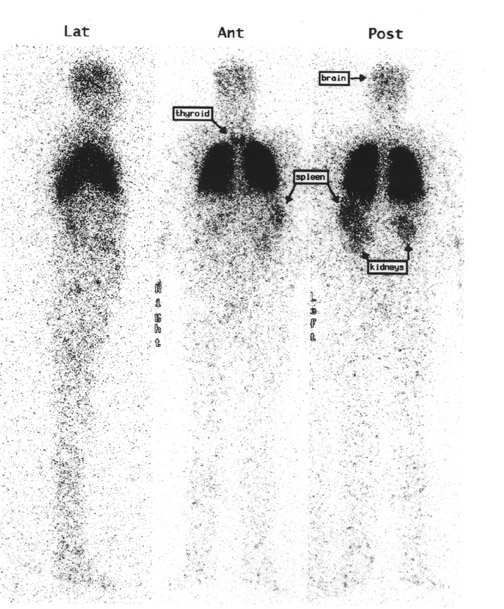
A 45-year-old man with hepatitis C was admitted with extreme volume overload, platypnea, and hypoxia. An echocardiogram showed evidence of pulmonary hypertension, no signs of heart failure, and a bubble study suggesting an extracardiac shunt. Arterial blood gas measurements repeatedly revealed hypoxemia with oxygen saturation in the range of 80% to 90%. A ventilation/perfusion (V/Q) scan showed a low probability for pulmonary embolism (not shown), and a chest computed tomography (CT) scan was negative for significant pulmonary disease (Figure 1). The patient's volume overload and hypoxia were believed to be associated with worsening liver failure. He was heavily diuresed and discharged. His hypoxia became worse over time, requiring increased home oxygen support. Several years later, a repeat echocardiogram showed resolution of pulmonary hypertension. A liver biopsy confirmed end-stage liver disease and hepatic cirrhosis and he was placed on the liver transplant list. A repeat CT scan and a shunt study with intravenous (IV) technetium-99m (Tc99m) macroaggregated albumin (MAA) (Figure 2) were performed to evaluate his hepatopulmonary status prior to liver transplantation.
Diagnosis
Right-to-left shunt associated with microscopic pulmonary arterial venous malformations (PAVMs) secondary to hepatopulmonary syndrome (HPS).
Imaging findings
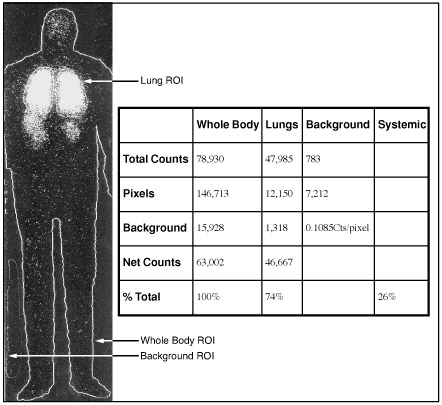
High-resolution CT images showed evidence of cirrhosis and end-stage liver disease, including a small liver with nodular contour, abdominal ascites, and splenomegaly (Figure 1). There was no evidence of macroscopic PAVMs or parenchymal lung abnormalities. A shunt study with IV Tc99m-labeled MAA, using a low-particle preparation (see below), shows activity in the kidneys and brain, in addition to normal lung activity (Figure 2). Also noteworthy is visualization of the spleen, consistent with splenomegaly, evident on the CT scan (Figure 1). The thyroid gland is also seen in the anterior view (Figure 2). Diffuse soft-tissue uptake labeling the whole body is also best appreciated in the lateral projection. These findings are con- sistent with a significant right-to-left shunt. Quantitative analysis estimated a 26% shunt fraction (Figure 3). Aside from microscopic PAVMs associated with HPS in advanced liver disease, right-to-left shunt can also be seen with macroscopic PAVMs most frequently associated with hereditary hemorrhagic telangiectasia (Osler-Rendu-Weber disease) and with congenital intracardiac anomalies, including Eisenmenger's complex, tetralogy of Fallot, Ebstein's anomaly of the tri- cuspid valve, and pulmonic stenosis with atrial septal defect.
Discussion
Whole-body nuclear imaging following IV injection of Tc99m MAA is useful to document and quantify right-to-left shunt. 1-11 Hepatopulmonary syndrome consists of arterial hypoxemia secondary to abnormal gas exchange in the setting of associated hepatic dysfunction and in the absence of intrinsic cardiopulmonary disease. Patients with HPS develop widespread vasodilation of peripheral branches of the pulmonary vasculature, at both the precapillary and capillary levels (15 to 150 µ in diameter), near the gas exchange area. 12 The dilated capillary has an expanded diameter, limiting oxygen diffusion capacity from adjacent alveoli for adequate red blood cell oxygenation. 4 These pulmonary arteriovenous communications are microscopic and are rarely visible, as opposed to the macroscopic PAVMs seen in hereditary hemorrhagic telangiectasia. The exact mechanism underlying HPS and associated PAVMs remains unclear. Postulates have included: failure of the damaged liver to clear circulating pulmonary vasodilators; production of a circulating vasodilator or inhibition of a circulating vasoconstrictor by the damaged liver; blunted hypoxic pulmonary vasoconstriction; release of a substance from the diseased liver that promotes fistula formation; and inability of the diseased liver to metabolize various substances present in the portal venous blood. 13 Other causes of HPS include trauma, schistosomiasis, metastatic carcinoma, and actinomycosis. 2
The Tc99m MAA perfusion tracer used in this shunt study had a measured radiochemical purity of 99.6%. Factors contributing to radiochemical impurity include unbound Tc99m in the form of free pertechnetate and albumin particles smaller than the optimal 10 to 50 µ size that could pass through the normal pulmonary arteriolar-capillary bed. Both of these sources of impurity can falsely elevate the measured shunt fraction, since a component of the total Tc99m label will pass through into the systemic circulation unrelated to a right-to-left shunt. 4
When performing a pulmonary perfusion study with Tc99m MAA in a patient with a known or suspected right-to-left shunt, it has been recommended that a low- particle preparation be used to minimize the extent of systemic arterial embolization. 14 Normally, ~1 million particles 10 to 40 µ in size are injected IV for a standard lung perfusion dose of Tc99m MAA. It is recommended that this be reduced to 100,000 to 200,000 particles in the setting of right-to-left shunt, as was done in this case.
Estimation of the pulmonary to systemic shunt fraction is calculated using the equation 1 : 100 × (TBC - TLC)/(TBC) where TBC is total body count and TLC is total lung count. Total body and lung counts are obtained by placing regions of interest
(ROIs) over the posterior whole-body image that is acquired 2 minutes post-tracer injection for 15 minutes.
An alternative method of right-to-left shunt quantification derives an estimate of total body counts from the measurement of renal and/or cerebral activity, assuming a given fixed proportion of total body blood flow. 6,11 While this approach may simplify determination of the total body counts, interpatient variability of the (renal and/or cerebral) to systemic blood flow ratios could introduce some imprecision. Quantification of the whole-body counts directly, as was done in this case, avoids this potential source of error.
Conclusion
Right-to-left shunt due to microscopic PAVMs is a manifestation of hepatopulmonary syndrome in ad-vanced liver disease. Although PAVMs are not readily evident radiographically, findings associated with chronic liver failure on CT along with evidence of right-to-left shunt determined by whole-body Tc99m MAA imaging in the appropriate clinical setting can confirm this diagnosis noninvasively.